Abstract
Urban areas attract birds during the winter when cities provide a predictable source of food and relatively stable weather conditions. However, many other factors determine the occurrence of birds in cities. This study analysed the relationship between corvids wintering in the city of Poznań, in western Poland and habitat features. Each of 32 research site was investigated three times. The most abundant species was the rook (mean = 17.4 individuals/site); the most frequent was the Eurasian magpie (88% of all sites). To determine variables that influence the abundance of birds, a set of generalised linear models was created, with model averaging being selected for each species. The abundance of hooded crows was positively influenced by surface of parks, water and by the length of tram tracks. The number of rooks was positively linked with the number of walnut trees, garbage cans and the surface of the water and negatively linked with the length of watercourses. Jackdaw was affected positively by the number of garbage cans, the surface of the water and negatively by the cover of trees. The number of magpies was associated with the number of walnut trees, garbage cans and negatively influenced by the surface of trees. None of the investigated habitat variables significantly affected the abundance of European jay. We also found a positive correlation between the abundances of hooded crow, rook, jackdaw and magpie. The results indicate that corvids wintering in urban ecosystems exhibit selectivity toward specific habitat features and that preferences differ among the studied species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation is proceeding at an ever-growing rate, an increasing part of the globe surface is occupied by human settlements with 55% of the world’s human population living in town and cities nowadays (United Nations 2018). Such transformed areas create an entirely new environment, which animals need to either adapt to or abandon (Angold et al. 2006; Nielsen et al. 2014; McKinney 2008; Møller 2009). The presence of humans in the immediate vicinity, anthropogenic barriers, noise, and artificial light are only a few of the many inconveniences that animals inhabiting cities need to cope with (Fernández-Juricic et al. 2001; Slabbekoorn and Peet 2003; de Molenaar et al. 2006; Møller 2008; Benítez-López et al. 2010; Van Doren et al. 2017). Diamond (1986) has suggested that the formation of such challenging habitats and the attempts of certain species to colonise them were, in the past, an example of rapid evolution, resulting in the creation of new groups of species commensal with humans.
Many taxa (e.g. birds, butterflies and carabid beetles) usually decrease in the number of species in the urbanization gradient (Kowarik 2011). Moreover, intensive management, combined with small surfaces and the spatial isolation of individual parks, greatly reduces the biological richness of urban greenery in terms of, e.g. birds and pollinators (Kowarik 2011; Banaszak-Cibicka et al. 2016; Müller et al. 2018).
On the other side, green urban areas, together with the blue infrastructure, are indisputably the city areas characterised by the highest biodiversity (Chamberlain et al. 2007). However, due to variation in the vegetation composition and landscape structure and management, urban habitats may have a various effect on urban flora and fauna create local hot-spot and cold-spots in the cityscape (Vilisics and Hornung 2009; Tonietto et al. 2011). The high degree of heterogeneity of the urban landscape, which favoured the co-occurrence of species with different habitat requirements, has led to an increased number of specific taxa, e.g. bees (Banaszak-Cibicka et al. 2016), butterflies (Hogsden and Hutchinson 2004), birds (Blair 1999), mammals (Racey and Euler 1982). Moreover, urban green areas provide refuges for rare and threatened species (Ives et al. 2016).
In addition, supplementary provision of food in cities, both intentional (bird feeders) and incidental (e.g. garbage cans, places where leftover food is dumped), reduces starvation and facilitates the survival of birds, particularly during the winter (Brittingham and Temple 1988; Robb et al. 2008). Another advantage of living in the city is the higher air temperature in the urban areas compared to surrounding areas (“urban heat island”). During the winter, higher temperature resulting in the thinner snow cower in city is favourable for foraging birds and help them to save energy (Møller 1983).
The compositions of avian communities change along the urbanisation gradient. The number of species decreases toward the city centre, where the avifauna is composed of several abundant synanthropic species and without habitat specialists (Marzluff 2001; Chace and Walsh 2006; Clergeau et al. 2006). Urban environments require great environmental tolerance (Møller 2009) and behavioural plasticity (Slabbekoorn and Peet 2003) from animals. Omnivores are in a favourable position (Chace and Walsh 2006). Corvids, with large brains compared to their body size and high behavioural plasticity, are assumed to possess a relatively high level of cognitive abilities (comparable even to those demonstrated by great apes) and exhibit a high degree of dietary diversity (Seed et al. 2009). This, along with the ability to discover and use new food resources, is extremely advantageous in cities, where garbage cans with plenty of food leftovers are common.
Although there have been numerous papers about the habitats and foraging preferences of corvids during the breeding season, the literature concerning the selectivity of wintering birds is scanty and usually concerns farmlands (e.g. Wilson et al. 1996; Jadczyk 2009). Very little research of relatively recent date has been conducted in urban landscapes during the winter (e.g. Jokimäki et al. 1996; Jokimäki and Suhonen 1998; Tryjanowski et al. 2015). As well, there have been few papers about interspecific relationships in winter assemblages of corvids (e.g. Waite 1984a, b). Therefore, this study aimed at a comparison of the abundances of corvids wintering in an urban ecosystem with habitat features in order to analyse birds’ habitat preferences.
Methods
Data collection
The study was conducted in the city of Poznań [52° 24′ 30.85″ N,
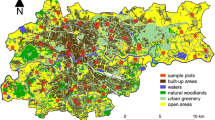
16° 56′ 2.44″ E] located in western Poland. With a population of approx. 540,000 citizens and an area of 262 km2 it is fifth the biggest city in Poland. Mean human population density is 2063 citizens per 1 km2 (Central Statistical Office 2017). The urban greenery is composed of three concentric rings and four connecting them green corridors (wedges) diverging from the centre to the boundaries of the city. There are 42 municipal parks (approx. 2% of the city territory), urban forests (covering 13.7% of the territory) and abundant blue infrastructure: Warta river with numerous smaller watercourses and several natural and artificial water reservoirs (covering 1.9% of the territory) (Klimko et al. 2009; Majkowska et al. 2017). Research sites (Fig. 1) were randomly selected in QGIS (QGIS Development Team 2016). A vector grid of polygons (squares of 500 × 500 m), limited by the boundaries of the city, was formed, with 32 polygons randomly selected using the ‘random extract’ tool. Research sites differed in terms of the land use structure (see Data analysis).
Fieldwork was carried out in the winter 2016/2017. The mean daily ambient temperature was −0.5 °C (range: −10 to 9 °C). The sum of precipitation during the trimester was 60.68 mm (data from Weather Underground https://www.wunderground.com, Poznań-Ławica Airport meteorological station (EPPO), accessed 15 December 2017). Thin snow cover (approx. 1–2 cm of thickness) was present during 12 of 36 days of the fieldwork, mainly in the second part of January and the beginning of February. Study sites were visited once a month. Observations were always performed by the same observer (KS). All countings were done between 8 a.m. and 2 p.m., to meet the daily activity of corvids.
Each field investigation consisted of a zigzag walk with binoculars aimed at surveying every part of the research sites (Supplementary material: Fig. A1). During each visit, the track was recorded with a smartphone with the GPS localisation application, the Geo Tracker, version.3.3.0.1338 (Bogdanovich 2016). The mean time spent on one visit was 45 min (range: 13–96). The minimal time applied to time spent in open areas (crop fields) with very few barriers (e.g. trees, buildings) obstructing the view. Consequently, observation with binoculars from a number of locations in the middle of the research site was entirely sufficient to determine the presence and abundance, or absence, of birds. This is a reliable method used in counting medium and large birds in an open area (Jankowiak et al. 2015). Contrastingly, the maximal periods applied to areas with trees, houses, tenement houses, and blocks of flats, where every part of the research site needed to be checked during a field visit in order to detect all birds. In purpose to avoid double-counting of the same individuals, the census was conducted quite quickly (mean = 18 min per 10 ha) (Jokimäki and Suhonen 1998). Backyards of houses could not be checked. Field visits were not conducted in adverse weather conditions, e.g. snow or heavy rain (Sliwinski et al. 2016).
In our study, we considered the following species of corvids: the hooded crow Corvus cornix, the rook Corvus frugilegus, the jackdaw Corvus monedula, the magpie Pica pica, the jay Garrulus glandarius, and the raven Corvus corax. Due to the low frequency of the raven during the research (5 individuals), only the first five species were taken under consideration in subsequent data analysis. We counted all birds that were observed foraging, sitting on the ground, perches, or roofs, or flying low while visibly linked with the ground (Fontana et al. 2011). Individuals that were heard only or that overflew the research site were not counted unless they were seen landing within the boundaries of the site.
Data analysis
Each research site was transformed into a vector layer (shapefile) in QGIS. Each different type of landscape was assigned to one of the following categories: blocks of flats, garden plots, houses, industrial areas, open areas, parks, wastelands, tenement houses, trees, or water, based on the definitions presented in Table 1. The areas of each category at each research site were measured in QGIS. The mean, minimum, and maximum values and standard deviation for each landscape category for each research site are presented in Supplementary material (Table A1). The length of watercourses and tram tracks (with broken stone and wooden or concrete railway ties – rails on concrete were not counted) and the distance from the research site to the Warta River were noted. Walnut trees Juglans regia and garbage cans were counted in the field.
The mean values of abundance for each species were taken from three counts for statistical analyses. A Z-score scale transformation was used to standardise independent variables prior to the analyses. To avoid multicollinearity in models, the variance inflation factor (VIF) was computed. Variables with a VIF > 4 were excluded from all models: houses, open areas, and tenement houses. The multicollinearity of the remaining explanatory variables in all models was not excessive (VIF < 3). We used generalised linear models (GLM’s) with a negative binomial distribution to determine factors affecting the abundance of hooded crow, rook, jackdaw, European jay and magpie. In order to best determine factors explaining the abundance of corvids, we used the model averaging function, which created a set of various models from all variables. We employed the information-theoretic approach (Burnham and Anderson 2002) to identify the most parsimonious models explaining variation in all dependent variables. Based on the full model, in each analysis, we constructed a set of candidate models that included different combinations of the predictors. We ranked all possible model combinations according to their ΔAICc values. We used models with the lowest AICc together with associated weight values (the probability that a given model is the best) as those best describing the data. We considered candidate models differing by less than 2 AICc units (ΔAICc <2.0) to be equally informative and subject to possible model averaging. For averaging, we used models with weights which had ΔAICc values lower than 4 (Burnham and Anderson 2002). The best models explaining the abundance of corvids are presented in Supplementary material (Table A2).
We used the canonical correspondence analysis (CCA) to correlate the corvids community composition with environmental factors. Based on the variance inflation factor, we excluded all multicollinearity variables from the model. Finally, we used following environmental variable: surface of tree, surface of water, surface of parks, surface of wasteland, surface of industrial, surface of tenement house, surface of open space area, number of garbage cans, number of walnut, length of tram tracks, distance to river, length of watercourses. We forward the selection of explanatory variables to choose the model, including only significant variables (p ≤ 0.05). We also calculated Spearman’s rank correlation coefficient between mean abundances of studied species.
All analyses were carried out in R (R Development Core Team 2016) and CANOCO 5 (Braak and Šmilauer 2012; Šmilauer and Lepš 2014). The VIF was calculated in the ‘usdm’ package (Naimi et al. 2014); the visualisation was performed in the ‘ggplot2’ package (Wickham 2009). The interspecific correlation was carried out in the ‘Hmisc’ package (Harrell Jr 2016), the model selection in the ‘glmulti’ package (Calcagno 2013).
Results
All counted species (except ravens) were recorded together at 28% of all study sites. The mean number of individuals per research site was 39.0 (SD = 53.8, range 0–315). The most frequent species was the magpie (88% of all study sites), the least frequent was the jackdaw (47% of all study sites). The most abundant species was the rook (mean = 17.4 individuals, max. = 197) with the highest standard deviation of abundance; the least abundant was the European jay (mean = 0.8 individuals) with the lowest standard deviation of abundance (details in Table 2).
The analysis revealed that each investigated species showed selectivity for a different set of habitat features (Table 3). The abundance of hooded crows was positively influenced by surface of the parks (p < 0.001), the surface of the water (p < 0.001) and by the length of tram tracks (p < 0.001, Fig. 2). The number of rooks was positively linked with the number of walnut trees (p < 0.001), the number of garbage cans (p < 0.001) and the surface of the water (p < 0.01) and negatively linked with the length of watercourses (p < 0.05, Fig. 3). Jackdaws were positively influenced by the number of garbage cans (p < 0.001), by the surface of the water (p < 0.05) and negatively influenced by the surface of trees (p < 0.05, Fig. 4). The number of magpies was associated with the number of walnut trees (p < 0.05), the number of garbage cans (p < 0.05) and negatively affected by the surface of trees (p < 0.05, Fig. 5). None of the investigated habitat variables significantly affected the abundance of European jay.
Environmental factors significantly predicted the corvids community in the urban environment in the winter period (test for all axes, pseudo-F = 2.6, p = 0.002). The environmental variables explain of the 54.76% variation in corvids data. The eigenvalues (importance measures of ordination axes) and explained cumulative variance for the selected CCA axes 1 and 2 were 0.23 and 41.05%, and 0.05 and 50.07%, respectively. The surface of trees and number of garbage cans were the most important variables predicting the corvids community in winter (Fig. 6, Supplementary material: Table A3).
Results of canonical correspondence analysis (CCA) results – environmental factors that significantly predicted the corvids community in the urban environment in the winter period (variables: number of garbage cans, surface of tenement houses, surface of trees, distance from the research site to Warta river, surface of open areas and length of watercourses)
We also found statistically significant positive correlations between the abundances of hooded crows, rooks, jackdaws, and magpies, as summarised in Table 4. Selected relationships between abundances of birds are presented in Fig. 7.
Discussion
In this paper, we showed that different species of birds from one family use different habitats within the urban ecosystem during a crucial time for survival, i.e. the winter. Although their prime habitat preferences differ significantly, they share the same environments in urban conditions. Urban greenery, together with the blue infrastructure are areas in cities with the highest avian species richness (Chamberlain et al. 2007). It is consistent with the results of our study since we found a positive relationship between the surface of the water bodies and the abundance of hooded crow, rook and jackdaw and between the surface of parks and the abundance of a hooded crow. River valleys are considered a natural habitat of the hooded crow (Zduniak and Kuczynski 2003). Crustaceans and molluscs constitute an essential part of its diet (Gołdyn et al. 2016); thus, the greater abundance of this species in proximity to water is not surprising. However, we found no relationship between the abundance of this species and distance from the river valley in the urban environment. On the other hand, we found a negative relationship between small watercourses and rook abundance. Watercourses were especially abundant in research sites covered by forest, in open areas more distant from the city centre and housing estates areas. As shown in CCA, the rook is strongly linked with habitats transformed by humans. Thus, the negative link between rook and watercourses may be a side effect of rook avoiding forests and open areas. Since it is assumed that populations of the hooded crow are relatively sedentary (Holyoak 1971), our results are consistent with data from other European cities showing that municipal parks create the most important habitats for breeding individuals (Vuorisalo et al. 2003; Zduniak 2005; Kövér et al. 2015). Many people visiting parks feed birds with leftover food (mostly bread), especially during the cold part of the year (Chosińska et al. 2012), which may be one of the factors attracting hooded crow to parks.
Corvids, as omnivorous with the ability to use new food resources supplied in the urban landscape like food leftovers in garbage cans or walnut trees profit from human proximity. Abundant garbage cans with leftover food, along with easily accessible bird feeders (Chosińska et al. 2012), may constitute factor inviting birds to forage near buildings. In this study, we found that rooks, jackdaws, and magpies were mainly affected by garbage cans. The supplementary food source is particularly important in the winter season when snow cover prevents effective foraging (Brittingham and Temple 1988; Robb et al. 2008). The phenomenon of the urban heat island may be additionally advantageous for wintering birds. Higher ambient temperatures cause thinner snow cover, which makes foraging easier and therefore birds may save energy (Møller 1983). Both latitude and longitude negatively influence the abundance of birds, but this relationship is less visible in highly urbanized areas which buffer the effect of severe winters on the north and east (Jokimäki et al. 2002; Tryjanowski et al. 2015). One of the significant predictors of the occurrence of rook and magpie was the number of walnut trees in a given plot. These trees are quite abundant in backyard gardens in housing estates, but also wastelands. As shown by Lenda et al. (2012), not only do corvids profit from caloric seeds such as walnuts but, by caching them, they contribute to the spread of walnut trees in abandoned agricultural areas. Corvids are smart enough to crack the hard shells of walnuts by dropping them from the air to the ground.
Although it is usually assumed that anthropogenic barriers such as railways have an adverse effect on animal populations (Santos et al. 2017), it turns out that some species can benefit from them (Li et al. 2010; Morelli et al. 2014). For example, several species of birds use railways as attractive foraging areas, sources of gastroliths, or sand-bathing locations where they can clean their feathers (Morelli et al. 2014; Lucas et al. 2017). In this study, we found a positive relationship between the length of tram tracks and the abundance of a hooded crow. Despite numerous papers showing the influence of railways on birds, to the best of our knowledge, there have been no studies concerning the impact of tram tracks in an urban landscape, which makes it more difficult to predict how a tram network may affect birds. Tram tracks with broken stone may constitute an attractive foraging area, especially during severe winters when crop fields in the farmlands and lawns in the cities are covered with layers of snow that impedes the foraging process. Dark stone aggregate beneath tracks quickly absorbs the rays of the sun. Also, the heat of passing trams and friction between tram wheels and rails raise the temperature of the substratum, resulting in thinner snow cover. Kaczmarski and Kaczmarek (2016) observed that tram tracks constitute a humid and prey-rich autumn and winter habitat for an urban population of smooth newts Lissotriton vulgaris. Therefore, corvids, as generalist foragers, may prey on small animals hiding or hibernating under rails. Since corvids living in an urban environment commonly forage on roadkill (Schwartz et al. 2018), they can profit equally from the carcasses of animals killed in collisions with trams. Corvids are known to cache seeds for use when food availability is low (Waite 1985; Cristol 2005; Castro et al. 2017). They can use tram tracks or railways with broken stones to hide, e.g. walnuts, to be retrieved when needed (Waite 1985). Further investigations are needed in order to understand better whether – and if so, how – tram tracks can influence the behaviour of wintering corvids and birds in general.
Canonical correspondence analysis revealed that the most important habitat variables predicting the winter corvids community in the city are: the surface of tree and number of garbage cans. Species differed in response to habitat features with European jay as the most outstanding species. According to Mazgajski et al. (2008) and Tzortzakaki et al. (2018), European jays occur in the woods on the peripheries of cities. Currently, in the phase of colonising urban habitats, they are encountered more and more frequently in urban green areas. However, further statistical analysis showed no statistically significant relationship between European jay occurrence and the surface of trees nor the surface of parks. On the other hand, there are hooded crow, rook and jackdaw that are more linked with the landscape transformed by humans and magpie connected with open areas and small watercourses. The similarity of the variables influencing the abundances of these four species is in accordance with statistically significant correlations between them.
During the winter, hooded crows, rooks, jackdaws, and magpies often forage in mixed flocks in abundant aggregations. According to Waite (1984b), although their foraging areas overlap, competition, because of dietary diversification, is not intense. While the prey of (in this case) carrion crow and rooks in grasslands consists mostly of earthworms and invertebrates from within the soil, jackdaws and magpies forage on these animals on the surface. Rooks may even benefit from foraging in dense flocks, as other birds dislodge earthworms that are then easier to catch. Conversely, larger earthworms eaten by carrion crows became unavailable when disturbed, then they tend to avoid foraging with large flocks of other birds. Individual magpies usually forage separately on the edges of corvids assemblages (Waite 1984b) and apparently do not join their flocks (e.g. do not fly away with the other birds). Since magpies do not migrate for winter, adults are strongly bonded to their territory. At the same time, non-breeders (birds in the first and second year of life and adults that have lost their territories) forage in monospecific flocks within a larger area in which they also spend the night (Busse 1963; Eden 1989). Wintering rooks, jackdaws and hooded crows do not hold territories but make daily flights between roosts where they spend the night and their foraging areas, using regular air corridors (Jadczyk 1994). Moreover, interspecific correlations may result from similar habitat preferences. As shown in this paper, hooded crows, rooks and jackdaws are linked with the water, garbage cans attract both rooks and jackdaws, and both rooks and magpies exhibit selectivity in favour of walnuts.
In the end, we need to highlight that since our fieldwork was performed only during one season (in the winter), then the results of this study are limited. However, the weather during three months of the fieldwork was rather typical for Polish winter.
Conclusion
Cities constitute attractive winter habitats for corvids. However, different species use different elements of the urban landscape. Therefore, a kind of mosaic of green areas (grasslands and ploughed lands) interspersed with aggregations of trees may be an attractive combination for wintering corvids. Jokimäki and Suhonen (1998) suggested that knowledge of the habitat features that influence the occurrence of birds is essential for urban planners, who could thus make cities more attractive for birds in order to better conserve declining species. Although corvids are generalists, with a high degree of behavioural plasticity, enabling them to easily adapt to changes in the environment (Seed et al. 2009), they will benefit from urban greening. Plans for the cityscape should account for these areas as elements of sustainable development.
References
Angold PG, Sadler JP, Hill MO, Pullin A, Rushton S, Austin K, Small E, Wood B, Wadsworth R, Sanderson R, Thompson K (2006) Biodiversity in urban habitat patches. Sci Total Environ 360:196–204. https://doi.org/10.1016/j.scitotenv.2005.08.035
Banaszak-Cibicka W, Ratyńska H, Dylewski Ł (2016) Features of urban green space favourable for large and diverse bee populations (Hymenoptera: Apoidea: Apiformes). Urban For Urban Gree 20:448–452. https://doi.org/10.1016/j.ufug.2016.10.015
Benítez-López A, Alkemade R, Verweij PA (2010) The impacts of roads and other infrastructure on mammal and bird populations: a meta-analysis. Biol Conserv 143:1307–1316. https://doi.org/10.1016/j.biocon.2010.02.009
Blair RB (1999) Birds and butterflies along an urban gradient: surrogate taxa for assessing biodiversity? Ecol Appl 9(1):164–170
Bogdanovich I (2016) Geo Tracker – GPS tracker (version 3.3.0.1338). Android application Retrieved from https://geo-tracker.org
Braak CJF, Šmilauer P (2012) CANOCO reference manual and user's guide: software for ordination (version 5.0). Biometris
Brittingham MC, Temple SA (1988) Impacts of supplemental feeding on survival rates of black-capped chickadees. Ecology 69(3):581–589. https://doi.org/10.2307/1941007
Burnham KP, Anderson DR (eds) (2002) Model selection and inference: a practical information-theoretic approach, 2nd edn. New York, Springer-Verlag. https://doi.org/10.1007/b97636
Busse P (1963) Wyniki obrączkowania ptaków w Polsce Rodzina Corvidae. Acta Ornithol 7:189–220
Calcagno V (2013) Glmulti: model selection and multimodel inference made easy. R package version 107. https://www.CRANR-projectorg/package=glmulti
Castro J, Molina-Morales M, Leverkus AB, Martínez-Baroja L, Pérez-Camacho L, Villar-Salvador P, Robello S, Rey-Benayas JM (2017) Effective nut dispersal by magpies (Pica pica L) in a Mediterranean agroecosystem. Oecologia 184(1):183–192. https://doi.org/10.1007/s00442-017-3848-x
Central Statistical Office (2017) Area and population in the territorial profile in 2017. Statistical Publishing Establishment, Warszawa [in Polish and English]
Chace JF, Walsh JJ (2006) Urban effects on native avifauna: a review. Landscape Urban Plan 74:46–69. https://doi.org/10.1016/j.landurbplan.2004.08.007
Chamberlain DE, Gough S, Vaughan H, Vickery JA, Appleton GF (2007) Determinants of bird species richness in public green spaces. Bird Study 54(1):87–97. https://doi.org/10.1080/00063650709461460
Chosińska K, Duduś L, Jakubiec Z (2012) Supplemental feeding of birds in human settlements of Western Poland. Intern Stud Sparrows 36:95–102. https://doi.org/10.1515/isspar-2015-0018
Clergeau P, Croci S, Jokimäki J, Kaisanlahti-Jokimäki ML, Dinetti M (2006) Avifauna homogenisation by urbanisation: analysis at different European latitudes. Biol Conserv 127:336–344. https://doi.org/10.1016/j.biocon.2005.06.035
Cristol DA (2005) Walnut-caching behavior of American crows. J Field Ornithol 76(1):27–32. https://doi.org/10.1648/0273-8570-76.1.27
de Molenaar JG, Sanders ME, Jonkers DA (2006) Road lighting and grassland birds: local influence of road lighting on a black-tailed godwit population in: rich C, Longcore T (eds) ecological consequences of artificial night lighting. Island Press, Washington DC, pp 114–136
Diamond JM (1986) Natural selection: rapid evolution of urban birds. Nature 324:107–108. https://doi.org/10.1038/324107a0
Eden SF (1989) The social organization of non-breeding magpies Pica pica. Ibis 131:141–153. https://doi.org/10.1111/j.1474-919X.1989.tb02753.x
Fernández-Juricic E, Jimenez MD, Lucas E (2001) Bird tolerance to human disturbance in urban parks of Madrid (Spain): management implications. In: Marzluff JM, Bowman R, Donelly R (eds) Avian ecology and conservation in an urbanizing world. Springer, Boston MA, pp 259–271. https://doi.org/10.1007/978-1-4615-1531-9_12
Fontana S, Sattler T, Bontadina F, Moretti M (2011) How to manage the urban green to improve bird diversity and community structure. Landscape Urban Plan 101:278–285. https://doi.org/10.1016/j.landurbplan.2011.02.033
Gołdyn B, Książkiewicz-Parulska Z, Zduniak P (2016) Freshwater molluscs in diet of hooded crow (Corvus cornix). Wilson J Ornithol 128:459–462. https://doi.org/10.1676/1559-4491-128.2.459
Harrell Jr FE. With contributions from Charles Dupont and many others (2016) Hmisc: Harrell Miscellaneous R package version 40–2. https://www.CRANR-projectorg/package=Hmisc
Hogsden KL, Hutchinson TC (2004) Butterfly assemblages along a human disturbance gradient in Ontario, Canada. Can J Zool 82:739–748. https://doi.org/10.1139/z04-048
Holyoak D (1971) Movements and mortality of Corvidae. Bird Study 18(2):97–106. https://doi.org/10.1080/00063657109476300
Ives CD, Lentini PE, Threlfall CG, Ikin K, Shanahan DF, Garrard GE, Bekessy AA, Fuller RA, Mumaw L, Rayner L, Rowe R, Valentine LE, Kendal D (2016) Cities are hotspots for threatened species. Glob Ecol Biogeogr 25:117–126. https://doi.org/10.1111/geb.12404
Jadczyk P (1994) Winter roosting of rooks Corvus frugilegus in Wrocław. Acta Ornithol 29:39–47
Jadczyk P (2009) Foraging strategies of wintering corvids Corvidae in suburban agrocenoses. Ann Univ Mariae Curie-Sklodowska Sectio C 64:49–65. https://doi.org/10.2478/v10067-010-0014-9
Jankowiak Ł, Antczak M, Kwiecinski Z, Szymanski P, Tobolka M, Tryjanowski P (2015) Diurnal raptor community wintering in an extensively used farmland. Ornis Fennica 92:76–87
Jokimäki J, Suhonen J (1998) Distribution and habitat selection of wintering birds in urban environments. Landscape Urban Plan 39:253–263. https://doi.org/10.1016/S0169-2046(97)00089-3
Jokimäki J, Suhonen J, Inki K, Jokinen S (1996) Biogeographical comparison of winter bird assemblages in urban environments in Finland. J Biogeogr 23(3):379–386. https://doi.org/10.1046/j.1365-2699.1996.00033.x
Jokimäki J, Clergeau P, Kaisanlahti-Jokimäki ML (2002) Winter bird communities in urban habitats: a comparative study between central and northern Europe. J Biogeogr 29(1):69–79. https://doi.org/10.1046/j.1365-2699.2002.00649.x
Kaczmarski M, Kaczmarek JM (2016) Heavy traffic low mortality – tram tracks as terrestrial habitat of newts. Acta Herpetol 11:227–231. https://doi.org/10.13128/Acta_Herpetol-17922
Klimko M, Bałuka B, Klimko W (2009) Przyroda miasta Poznania. Wydział Ochrony Środowiska Urzędu Miasta Poznania, Poznań
Kövér L, Gyüre P, Balogh P, Huettmann F, Lengyel S, Juhász L (2015) Recent colonization and nest site selection of the hooded crow (Corvus corone cornix L.) in an urban environment. Landscape Urban Plan 133:78–86. https://doi.org/10.1016/j.landurbplan.2014.09.008
Kowarik I (2011) Novel urban ecosystems biodiversity and conservation. Environ Pollut 159:1974–1983. https://doi.org/10.1016/j.envpol.2011.02.022
Lenda M, Skórka P, Knops JMH, Moroń D, Tworek S, Woyciechowski M (2012) Plant establishment and invasions: an increase in a seed disperser combined with land abandonment causes an invasion of the non-native walnut in Europe. P R Soc B 279:1491–1497. https://doi.org/10.1098/rspb.2011.2153
Li Z, Ge C, Li J, Li Y, Xu A, Zhou K, Xue D (2010) Ground-dwelling birds near the Qinghai-Tibet highway and railway. Transport Res D – Tr E 15:525–528. https://doi.org/10.1016/j.trd.2010.07.004
Lucas PS, de Carvalho RG, Grilo C (2017) Railway disturbances on wildlife: types effects and mitigation measures. In: Borda-de-Água L, Barrientos R, Beja P, Pereira HM (eds) Railway ecology. Springer, Cham, pp 81–102
Majkowska A, Kolendowicz L, Półrolniczak M, Hauke J, Czernecki B (2017) The urban heat island in the city of Poznań as derived from Landsat 5 TM. Theor Appl Climatol 128:769–783. https://doi.org/10.1007/s00704-016-1737-6
Marzluff JM (2001) Worldwide urbanization and its effects on birds. In: Marzluff JM, Bowman R, Donnelly R (eds) Avian ecology and conservation in an urbanizing world. Springer, Boston MA, pp 19–47. https://doi.org/10.1007/978-1-4615-1531-9_2
Mazgajski TD, Żmihorski M, Halba R, Woźniak A (2008) Long-term population trends of corvids wintering in urban parks in Central Poland. Pol J Ecol 56:521–526
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11(2):161–176. https://doi.org/10.1007/s11252-007-0045-4
Møller AP (1983) Habitat selection and feeding activity in the magpie Pica pica. J Ornithol 124:147–161. https://doi.org/10.1007/BF01640161
Møller AP (2008) Flight distance of urban birds predation and selection for urban life. Behav Ecol Sociobiol 63:63–75. https://doi.org/10.1007/s00265-008-0636-y
Møller AP (2009) Successful city dwellers: a comparative study of the ecological characteristics of urban birds in the Western Palearctic. Oecologia 159:849–858. https://doi.org/10.1007/s00442-008-1259-8
Morelli F, Beim M, Jerzak L, Jones D, Tryjanowski P (2014) Can roads railways and related structures have positive effects on birds? - a review. Transport Res D – Tr E 30:21–31. https://doi.org/10.1016/j.trd.2014.05.006
Müller A, Bøcher PK, Fischer C, Svenning J-C (2018) ‘Wild’ in the city context: do relative wild areas offer opportunities for urban biodiversity? Landscape Urban Plan 170:256–265. https://doi.org/10.1016/j.landurbplan.2017.09.027
Naimi B, Hamm N, Groen TA, Skidmore AK, Toxopeus AG (2014) Where is positional uncertainty a problem for species distribution modelling. Ecography 37:191–203. https://doi.org/10.1111/j.1600-0587.2013.00205.x
Nielsen AB, van den Bosch M, Maruthaveeran S, van den Bosch CK (2014) Species richness in urban parks and its drivers: a review of empirical evidence. Urban Ecosyst 17(1):305–327. https://doi.org/10.1007/s11252-013-0316-1
QGIS Development Team (2016) QGIS geographic information system, version 2160. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-projectorg/
Racey GD, Euler DL (1982) Small mammal and habitat response to shoreline cottage development in Central Ontario, Canada. Can J Zool 60:865–880. https://doi.org/10.1139/z82-119
Robb GN, McDonald RA, Chamberlain DE, Bearhop S (2008) Food for thought: supplementary feeding as a driver of ecological change in avian populations. Front Ecol Environ 6(9):476–484. https://doi.org/10.1890/060152
Santos SM, Carvalho F, Mira A (2017) Current knowledge on wildlife mortality in railways. In: Borda-de-Água L, Barrientos R, Beja P, Pereira H (eds) Railway ecology. Springer, Cham, pp 11–22
Schwartz ALW, Williams HF, Chadwick E, Thomas RJ, Perkins SE (2018) Roadkill scavenging behaviour in an urban environment. J Urban Ecol 4(1):1–7. https://doi.org/10.1093/jue/juy006
Seed A, Emery N, Clayton N (2009) Intelligence in corvids and apes: a case of convergent evolution? Ethology 115:401–420. https://doi.org/10.1111/j.1439-0310.2009.01644.x
Slabbekoorn H, Peet M (2003) Ecology: birds sing at a higher pitch in urban noise. Nature 424:267–267. https://doi.org/10.1038/424267a
Sliwinski M, Powell L, Koper N, Giovanni M, Schacht W (2016) Research design considerations to ensure detection of all species in an avian community. Methods Ecol Evol 7:456–462. https://doi.org/10.1111/2041-210X.12506
Šmilauer P, Lepš J (2014) Multivariate analysis of ecological data using CANOCO 5. Cambridge University Press, Cambridge
Tonietto R, Fant J, Ascher J, Ellis K, Larkin D (2011) A comparison of bee communities of Chicago green roofs, parks and prairies. Landsc Urban Plan 103:102–108. https://doi.org/10.1016/j.landurbplan.2011.07.004
Tryjanowski P, Sparks TH, Biaduń W, Brauze T, Hetmański T, Martyka R, Skórka P, Indykiewicz P, Myczko Ł, Kunysz P, Kawa P, Czyż S, Czechowski P, Polakowski M, Zduniak P, Jerzak L, Janiszewski T, Goławski A, Duduś L, Nowakowski JJ, Wuczyński A, Wysocki D (2015) Winter bird assemblages in rural and urban environments: a national survey. PLoS One 10:1–25. https://doi.org/10.1371/journal.pone.0130299
Tzortzakaki O, Kati V, Kassara C, Tietze DT, Giokas S (2018) Seasonal patterns of urban bird diversity in a Mediterranean coastal city: the positive role of open green spaces. Urban Ecosyst 21:27–39. https://doi.org/10.1007/s11252-017-0695-9
United Nations (2018) Revision of world urbanisation prospects. United Nations Department of Economic and Social Affair. https://www.unorg/development/desa/publications/2018-revision-of-world-urbanization-prospects.html. Accessed 23 February 2019
Van Doren BM, Horton KG, Dokter AM, Klinck H, Elbin SB, Farnsworth A (2017) High-intensity urban light installation dramatically alters nocturnal bird migration. P Natl Acad Sci USA 114:11175–11180. https://doi.org/10.1073/pnas.1708574114
Vilisics F, Hornung E (2009) Urban areas as hot-spots for introduced and shelters for native isopod species. Urban Ecosyst 12:333–345. https://doi.org/10.1007/s11252-009-0097-8
Vuorisalo T, Andersson H, Hugg T, Lahtinen R, Laaksonen H, Lehikoinen E (2003) Urban development from an avian perspective: causes of hooded crow (Corvus corone cornix) urbanisation in two Finnish cities. Landscape Urban Plan 62:69–87. https://doi.org/10.1016/S0169-2046(02)00124-X
Waite RK (1984a) Winter habitat selection and foraging behaviour in sympatric corvids. Ornis Scand 15:55–62. https://doi.org/10.2307/3676003
Waite RK (1984b) Sympatric corvids – effects of social behaviour aggression and avoidance on feeding. Behav Ecol Sociobiol 15:55–59. https://doi.org/10.1007/BF00310215
Waite RK (1985) Food caching and recovery by farmland corvids. Bird Study 32(1):45–49. https://doi.org/10.1080/00063658509476854
Wickham H (2009) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Wilson JD, Taylor R, Muirhead LB (1996) Field use by farmland birds in winter: an analysis of field type preferences using resampling methods. Bird Study 43:320–332. https://doi.org/10.1080/00063659609461025
Zduniak P (2005) Wrona siwa Corvus cornix w Polsce – stan wiedzy i perspektywy badań In: Jerzak L, Kavanagh BP, Tryjanowski P (eds) Ptaki Krukowate Polski [Corvids of Poland]. Bogucki Wydawnictwo Naukowe, Poznań, pp 113–125
Zduniak P, Kuczynski L (2003) Breeding biology of the hooded crow Corvus corone cornix in Warta river valley (W Poland). Acta Ornithol 38:143–150. https://doi.org/10.3161/068.038.020
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material

Fig. A1
An example of field visit (2017-01-14, research site no. 24 “Górczynek”). Black dotted line is the route of the observer recorded with a smartphone with the GPS localisation application: Geo Tracker, version.3.3.0.1338 (Bogdanovich 2016). Map based on Open Street Map background raster layer (JPG 153 kb)
ESM 2
(DOCX 174 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szala, K., Dylewski, Ł. & Tobolka, M. Winter habitat selection of Corvids in an urban ecosystem. Urban Ecosyst 23, 483–493 (2020). https://doi.org/10.1007/s11252-020-00942-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-020-00942-2