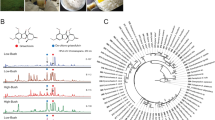
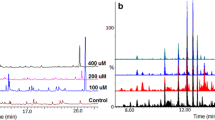
Abstract
Plants produce a wide variety of secondary metabolites (SMs) of pharmacological interest that, due to their high structural complexity, are difficult to obtain from chemical synthesis. Knowledge of the enzymatic process involved in the biosynthesis of these SMs may contribute to the development of new strategies for obtaining them from plant tissues. Maytenus ilicifolia contains three main classes of bioactive compounds: sesquiterpene pyridine alkaloids, flavonoids and, primarily, quinonemethide triterpenes, which are well known for their high and broad antitumor activity. Thus, to determine the enzymatic composition in the secondary metabolism of M. ilicifolia, we carried out proteomic profiling of M. ilicifolia cell cultures with and without increased concentrations of maytenin and 22β-hydroxy-maytenin by elicitation with methyl jasmonate for 48 h. For this purpose, the analysis of soluble proteins was performed via LCMS/MS experiments using a shotgun strategy and compared to specific databases, resulting in 1319 identifications in the two types of cells. Many enzymes involved in secondary metabolism were detected, including cytochrome P450-dependent monooxygenases, which potentially catalyze the final steps in the quinonemethide triterpene biosynthesis. In conclusion, we report for the first time a proteomic study of a cell culture system that is an effective elicitor of quinonemethide triterpene production.




Similar content being viewed by others
References
Abe I (2007) Enzymatic synthesis of cyclic triterpenes. Nat Prod Rep 24:1311–1331. doi:10.1039/b616857b
Ahmed MS, Fong HHS, Soejarto DD et al (1981) High-performance liquid chromatographic separation and quantitation of maytansinoids in Maytenus ilicifolia. J Chromatogr A 213:340–344. doi:10.1016/S0021-9673(00)81919-6
Barros J, Serk H, Granlund I, Pesquet E (2015) The cell biology of lignification in higher plants. Ann Bot 115:1053–1074. doi:10.1093/aob/mcv046
Blakeley SD, Dekroon C, Cole KP et al (1994) Isolation of a full-length cDNA encoding cytosolic enolase from Ricinus communis. Plant Physiol 105:455–456. doi:10.1104/pp.105.1.455
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Buffa Filho W, Bolzani V da S, Furlan M et al (2004) In vitro propagation of Maytenus ilicifolia (Celastraceae) as potential source for antitumoral and antioxidant quinomethide triterpenes production. A rapid quantitative method for their analysis by reverse-phase high-performance liquid chromatograp. Arkivoc 2004:137–146. doi:10.3998/ark.5550190.0005.617
Cahoon EB, Lindqvist Y, Schneider G, Shanklin J (1997) Redesign of soluble fatty acid desaturases from plants for altered substrate specificity and double bond position. Proc Natl Acad Sci 94:4872–4877. doi:10.1073/pnas.94.10.4872
Camelio AM, Johnson TC, Siegel D (2015) Total synthesis of celastrol, development of a platform to access celastroid natural products. J Am Chem Soc 137:11864–11867. doi:10.1021/jacs.5b06261
Champagne A, Rischer H, Oksman-Caldentey K-M, Boutry M (2012) In-depth proteome mining of cultured Catharanthus roseus cells identifies candidate proteins involved in the synthesis and transport of secondary metabolites. Proteomics 12:3536–3547. doi:10.1002/pmic.201200218
Choi JY, Ramasamy T, Kim SY et al (2016) PEGylated lipid bilayer-supported mesoporous silica nanoparticle composite for synergistic co-delivery of axitinib and celastrol in multi-targeted cancer therapy. Acta Biomater 39:94–105. doi:10.1016/j.actbio.2016.05.012
Coppede J da S, Pina ES, Paz TA et al (2014) Cell cultures of Maytenus ilicifolia Mart. are richer sources of quinone-methide triterpenoids than plant roots in natura. Plant Cell Tissue Organ Cult 118:33–43. doi:10.1007/s11240-014-0459-7
Corsino J, De Carvalho PRF, Kato MJ et al (2000) Biosynthesis of friedelane and quinonemethide triterpenoids is compartmentalized in Maytenus aquifolium and Salacia campestris. Phytochemistry 55:741–748. doi:10.1016/S0031-9422(00)00285-5
de Souza LM, Cipriani TR, Iacomini M et al (2008) HPLC/ESI-MS and NMR analysis of flavonoids and tannins in bioactive extract from leaves of Maytenus ilicifolia. J Pharm Biomed Anal 47:59–67. doi:10.1016/j.jpba.2007.12.008
Demidchik V (2015) Mechanisms of oxidative stress in plants: from classical chemistry to cell biology. Environ Exp Bot 109:212–228. doi:10.1016/j.envexpbot.2014.06.021
Deng W-W, Zhang M, Wu J-Q et al (2013) Molecular cloning, functional analysis of three cinnamyl alcohol dehydrogenase (CAD) genes in the leaves of tea plant, Camellia sinensis. J Plant Physiol 170:272–282. doi:10.1016/j.jplph.2012.10.010
Dewick PM (1995) The biosynthesis of shikimate metabolites. Nat Prod Rep 12:579–607
Dewick PM (2009) The shikimate pathway: aromatic amino acids and phenylpropanoids. In: Medicinal natural products: a biosynthetic approach, 3rd edn. Wiley, pp 137–186
Diebold R, Schuster J, Däschner K, Binder S (2002) The branched-chain amino acid transaminase gene family in Arabidopsis encodes plastid and mitochondrial proteins. Plant Physiol 129:540–550. doi:10.1104/pp.001602
dos Santos VADFFM, Dos Santos DP, Castro-Gamboa I et al (2010) Evaluation of antioxidant capacity and synergistic associations of quinonemethide triterpenes and phenolic substances from Maytenus ilicifolia (celastraceae). Molecules 15:6956–6973. doi:10.3390/molecules15106956
dos Santos VFFM, Regasini LO, Nogueira CR et al (2012) Antiprotozoal sesquiterpene pyridine alkaloids from Maytenus ilicifolia. J Nat Prod 75:991–995. doi:10.1021/np300077r
dos Santos VAFFM, Leite KM, Da Costa Siqueira M et al (2013) Antiprotozoal activity of quinonemethide triterpenes from Maytenus ilicifolia (Celastraceae). Molecules 18:1053–1062. doi:10.3390/molecules18011053
Forsthoefel NR, Cushman MA, Cushman JC (1995) Posttranscriptional and post translational control of enolase expression in the facultative crassulacean acid metabolism plant Mesembryanthemum Crystallinum L. Plant Physiol 108:1185–1195. doi:10.1104/pp.108.3.1185
Fox TC, Mujer CV, Andrews DL et al (1995) Identification and gene expression of anaerobically induced enolase in Echinochloa phyllopogon and Echinochloa crus-pavonis. Plant Physiol 109:433–443. doi:10.1104/pp.109.2.433
Georgiev V, Marchev A, Berkov S, Pavlov A (2013) Plant in vitro systems as sources of tropane alkaloids. In: Ramawat GK, Mérillon J-M (eds) Natural products: phytochemistry, botany and metabolism of alkaloids, phenolics and terpenes. Springer, Berlin, pp 173–211
Götz S, García-Gómez JM, Terol J et al (2008) High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res 36:3420–3435. doi:10.1093/nar/gkn176
Gullo FP, Sardi JCO, Santos VAFFM et al (2012) Antifungal activity of maytenin and pristimerin. Evidence-Based Complement Altern Med 2012:1–6. doi:10.1155/2012/340787
Guo J, Carrington Y, Alber A, Ehlting J (2014) Molecular characterization of quinate and shikimate metabolism in Populus trichocarpa. J Biol Chem 289:23846–23858. doi:10.1074/jbc.M114.558536
Gutiérrez F, Estévez-Braun A, Ravelo ÁG et al (2007) Terpenoids from the medicinal plant Maytenus ilicifolia. J Nat Prod 70:1049–1052. doi:10.1021/np070019g
Harakava R (2005) Genes encoding enzymes of the lignin biosynthesis pathway in Eucalyptus Genet. Mol Biol 28:601–607. doi:10.1590/S1415-47572005000400015
Herrmann KM, Weaver LM (1999) The shikimate pathway. Annu Rev Plant Physiol Plant Mol Biol 50:473–503. doi:10.1146/annurev.arplant.50.1.473
Huang C, Ding S, Zhang H et al (2011) CIPK7 is involved in cold response by interacting with CBL1 in Arabidopsis thaliana. Plant Sci 181:57–64. doi:10.1016/j.plantsci.2011.03.011
Jardim ACG, Igloi Z, Shimizu JF et al (2015) Natural compounds isolated from Brazilian plants are potent inhibitors of hepatitis C virus replication in vitro. Antiviral Res 115:39–47. doi:10.1016/j.antiviral.2014.12.018
Kosová K, Vítámvás P, Prášil IT (2014) Proteomics of stress responses in wheat and barley—search for potential protein markers of stress tolerance. Front Plant Sci 5:14. doi:10.3389/fpls.2014.00711
Koutaniemi S, Toikka MM, Kärkönen A et al (2005) Characterization of basic p-coumaryl and coniferyl alcohol oxidizing peroxidases from a lignin-forming Picea abies suspension culture. Plant Mol Biol 58:141–157. doi:10.1007/s11103-005-5345-6
Kutsuki H, Shimada M, Higuchi T (1982) Regulatory role of cinnamyl alcohol dehydrogenase in the formation of guaiacyl and syringyl lignins. Phytochemistry 21:19–23. doi:10.1016/0031-9422(82)80006-X
Lal SK, Lee C, Sachs MM (1998) Differential regulation of enolase during anaerobiosis in maize. Plant Physiol 118:1285–1293. doi:10.1104/pp.118.4.1285
Lippmann R, Kaspar S, Rutten T et al (2009) Protein and metabolite analysis reveals permanent induction of stress defense and cell regeneration processes in a tobacco cell suspension culture. Int J Mol Sci 10:3012–3032. doi:10.3390/ijms10073012
Liu M-H, Yang B-R, Cheung W-F et al (2015) Transcriptome analysis of leaves, roots and flowers of Panax notoginseng identifies genes involved in ginsenoside and alkaloid biosynthesis. BMC Genomics 16:265. doi:10.1186/s12864-015-1477-5
Martínez-Esteso MJ, Martínez-Márquez A, Sellés-Marchart S et al (2015) The role of proteomics in progressing insights into plant secondary metabolism. Front Plant Sci 6:504. doi:10.3389/fpls.2015.00504
Messner B, Boll M (1993) Elicitor-mediated induction of enzymes of lignin biosynthesis and formation of lignin-like material in a cell suspension culture of spruce (Picea abies). Plant Cell Tissue Organ Cult 34:261–269. doi:10.1007/BF00029715
Mossi AJ, Mazutti M, Paroul N et al (2009) Chemical variation of tannins and triterpenes in Brazilian populations of Maytenus ilicifolia Mart. Ex Reiss. Braz J Biol 69:339–345. doi:10.1590/S1519-69842009000200015
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1021/jf9040386
Negrel J, Javelle F (1995) Induction of phenylpropanoid and tyramine metabolism in pectinase- or pronase-elicited cell suspension cultures of tobacco (Nicotiana tabacum). Physiol Plant 95:569–574. doi:10.1111/j.1399-3054.1995.tb05524.x
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79:629–661. doi:10.1021/acs.jnatprod.5b01055
Oksman-Caldentey K-M, Inzé D (2004) Plant cell factories in the post-genomic era: new ways to produce designer secondary metabolites. Trends Plant Sci 9:433–440. doi:10.1016/j.tplants.2004.07.006
Oramas-Royo SM, Chávez H, Martín-Rodríguez P et al (2010) Cytotoxic triterpenoids from Maytenus retusa. J Nat Prod 73:2029–2034. doi:10.1021/np100517u
Pauwels L, Inzé D, Goossens A (2009) Jasmonate-inducible gene: what does it mean? Trends Plant Sci 14:87–91. doi:10.1016/j.tplants.2008.11.005
Paz TA, dos Santos VAFFM, Inácio MC et al (2013) Production of the quinone-methide triterpene maytenin by in vitro adventitious roots of Peritassa campestris (Cambess.) A.C. Sm. (Celastraceae) and rapid detection and identification by APCI-IT-MS/MS. Biomed Res Int 2013:485837. doi:10.1155/2013/485837
Pereira AMS, Moro JR, Cerdeira RMM, França SC (1994) Micropropagation of maytenus aquifolium martius. J Herbs Spices Med Plants 2:11–18. doi:10.1300/J044v02n03_03
Phillips DR, Rasbery JM, Bartel B, Matsuda SPT (2006) Biosynthetic diversity in plant triterpene cyclization. Curr Opin Plant Biol 9:305–314. doi:10.1016/j.pbi.2006.03.004
Pina ES, Silva DB, Teixeira SP et al (2016) Mevalonate-derived quinonemethide triterpenoid from in vitro roots of Peritassa laevigata and their localization in root tissue by MALDI imaging. Sci Rep 6:22627. doi:10.1038/srep22627
Plaxton WC (1996) The regulation and organization of plant glycolysis. Annu Rev Plant Physiol Plant Mol Biol 47:185–214. doi:10.1146/annurev.arplant.47.1.185
Radakovits R, Jinkerson RE, Darzins A, Posewitz MC (2010) Genetic engineering of algae for enhanced biofuel production. Eukaryot Cell 9:486–501. doi:10.1128/EC.00364-09
Ralph J, Lundquist K, Brunow G et al (2004) Lignins: natural polymers from oxidative coupling of 4-hydroxyphenyl-propanoids. Phytochem Rev 3:29–60. doi:10.1023/B:PHYT.0000047809.65444.a4
Ren L, Sun J, Chen S et al (2014) A transcriptomic analysis of Chrysanthemum nankingense provides insights into the basis of low temperature tolerance. BMC Genomics 15:844. doi:10.1186/1471-2164-15-844
Rizhsky L, Liang H, Shuman J et al (2004) When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol 134:1683–1696. doi:10.1104/pp.103.033431.1
Sasaki S, Baba K, Nishida T et al (2006) The cationic cell-wall-peroxidase having oxidation ability for polymeric substrate participates in the late stage of lignification of Populus alba L. Plant Mol Biol 62:797–807. doi:10.1007/s11103-006-9057-3
Shekhar S, Mishra D, Gayali S et al (2016) Comparison of proteomic and metabolomic profiles of two contrasting ecotypes of sweetpotato (Ipomoea batata L.). J Proteomics 143:306–317. doi:10.1016/j.jprot.2016.03.028
Spitzer-Rimon B, Marhevka E, Barkai O et al (2010) EOBII, a gene encoding a flower-specific regulator of phenylpropanoid volatiles’ biosynthesis in petunia. Plant Cell 22:1961–1976. doi:10.1105/tpc.109.067280
Su CF, Wang YC, Hsieh TH et al (2010) A novel MYBS3-dependent pathway confers cold tolerance in rice. Plant Physiol. doi:10.1104/pp.110.153015
Van Cutsem E, Simonart G, Degand H et al (2011) Gel-based and gel-free proteomic analysis of Nicotiana tabacum trichomes identifies proteins involved in secondary metabolism and in the (a)biotic stress response. Proteomics 11:440–454. doi:10.1002/pmic.201000356
Vogt T (2010) Phenylpropanoid biosynthesis. Mol Plant 3:2–20. doi:10.1093/mp/ssp106
Voll LM, Hajirezaei MR, Czogalla-Peter C et al (2009) Antisense inhibition of enolase strongly limits the metabolism of aromatic amino acids, but has only minor effects on respiration in leaves of transgenic tobacco plants. New Phytol 184:607–618. doi:10.1111/j.1469-8137.2009.02998.x
Weaver LM, Herrmann KM (1997) Dynamics of the shikimate pathway in plants. Trends Plant Sci 2:346–351. doi:10.1016/S1360-1385(97)84622-5
Wendt KU (2005) Enzyme mechanisms for triterpene cyclization: new pieces of the puzzle. Angew Chemie Int Ed 44:3966–3971. doi:10.1002/anie.200500804
Weng J-K, Chapple C (2010) The origin and evolution of lignin biosynthesis. New Phytol 187:273–285. doi:10.1111/j.1469-8137.2010.03327.x
Wildermuth MC, Dewdney J, Wu G, Ausubel FM (2001) Isochorismate synthase is required to synthesize salicylic acid for plant defence. Nature 414:562–565. doi:10.1038/417571a
Yamazaki M, Mochida K, Asano T et al (2013) Coupling deep transcriptome analysis with untargeted metabolic profiling in Ophiorrhiza pumila to further the understanding of the biosynthesis of the anti-cancer alkaloid camptothecin and anthraquinones. Plant Cell Physiol 54:686–696. doi:10.1093/pcp/pct040
Yang Q-S, Gao J, He W-D et al (2015) Comparative transcriptomics analysis reveals difference of key gene expression between banana and plantain in response to cold stress. BMC Genomics 16:446. doi:10.1186/s12864-015-1551-z
Yue W, Ming Q-L, Lin B et al (2016) Medicinal plant cell suspension cultures: pharmaceutical applications and high-yielding strategies for the desired secondary metabolites. Crit Rev Biotechnol 36:215–232. doi:10.3109/07388551.2014.923986
Zhao J, Davis LC, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23:283–333. doi:10.1016/j.biotechadv.2005.01.003
Acknowledgements
We thank the São Paulo Research Foundation (FAPESP) for the CIBFar-2013/07600-3 grant. T. A. Paz thanks CAPES for the provision of a scholarship. V. A. F. F. M. dos Santos and M. C. Inácio thank FAPESP for their fellowships (2011/01544-9 and 2014/19362-2, respectively). V. A. F. F. M. dos Santos is also grateful to the National Council for Scientific and Technological Development (CNPq) for her fellowship. M. Furlan and M. S. Palma would also like to thank CNPq for their research fellowships.
Author contributions
TAP was responsible for the conception and design of all experiments, data analysis, drafting and editing of the manuscript. MCI contributed with plant cell experiments and revision of the manuscript. VAFFMS contributed with proteomic experiments. NBD and MSP contributed with proteomic experiments and revised the manuscript. AMSP supervised part of experiments and contributed to data interpretation and final revision of the manuscript. MF contributed to the experimental design, to data interpretation, drafting and final revision of the manuscript, and supervised all experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Ali R. Alan.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paz, T.A., dos Santos, V.A.F.F.M., Inácio, M.C. et al. Proteome profiling reveals insights into secondary metabolism in Maytenus ilicifolia (Celastraceae) cell cultures producing quinonemethide triterpenes. Plant Cell Tiss Organ Cult 130, 405–416 (2017). https://doi.org/10.1007/s11240-017-1236-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-017-1236-1