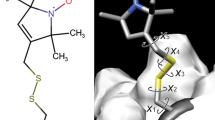
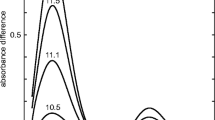
The hybrid multilevel approach based on molecular dynamics, quantum mechanics, and statistical theory has been applied to profiles of electronic absorption bands of a number of thiazine dyes (phenothiazine, methylene blue, and methylene green), whose photochemical and biological properties depend on the external medium. The effect of single fragments of the methylene green (MG) molecule on its spectral-luminescent properties is studied. The most preferred geometry for the MG molecule is identified, and its relations with the spectral properties and the solvent nature are established. The energies of the electron states are calculated, and their nature is revealed. The electrostatic potential is analyzed, and the places of the most probable interaction of the molecule with the medium are found. Conclusions on the mechanism of forming the MG absorption spectrum are made based on the observations and calculations performed within the framework of the present work. It is established that the formation of the N–H bonds leads to the formation of an inactive lower excited singlet state and delocalization of the electron charge upon excitation. The position of the maximum of the long-wavelength absorption band of the MG correlates with the donor number of solvents in the series acetonitrile < water < ethanol < isopropanol < dimethyl sulfoxide. The position of the MG electron transition in the region of 630 nm is determined by the interaction of nitrogen and sulfur atoms of the central ring rather than by the increased number of substituents in the structure of the molecule.
Similar content being viewed by others
References
O. E. Glukhova, G. V. Savostyanov, and M. M. Slepchenkov, Procedia Mater. Sci., 6, 256–264 (2014).
V. Jahed, A. Zarrabi, A. Bordbar, et al., Food Chem., 165, 241 (2014).
S. Sakthivel, T. Alagesan, A. A. Al-Saadi, et al., Spectrochim. Acta A, 127, 157–167 (2014).
K. Aggarwal and J. M. Khurana, J. Mol. Struct., 21, 1079 (2015).
URL: http://wikipedia.org (access date January 21, 2018).
O. B. Rudakov, E. A. Khorokhordina, L. V. Rudakova, et al., Proc. Voronezh State Univ. Ser.: Chem. Biol. Pharm., 3, 41–46 (2015).
Y. Shen, S. Liu, L. Wang, et al., Luminescence, 29, 884–892 (2014); DOI: https://doi.org/10.1002/bio.2637.
M. Ghaedi, H. Karimi, and F. Yousefi, Hum. Exp. Toxicol., 33, No. 9, 956–967 (2014).
H. Ryosuke and T. Masayoshi, Proc. Am. Math. Soc., 140, 3905–3920 (2012).
H. Xu, N. Yao, H. Xu, et al., Int. J. Mol. Sci., 14, 14185–14203 (2013).
K. A. Majorek, P. J. Porebski, A. Dayal, et al., Mol. Immunol., 52, 174–182 (2012).
F. Samari, B. Hemmateenejad, M. Shamsipur, et al., Inorg. Chem., 51, 3454–3464 (2012).
G. V. Mayer, V. G. Plotnikov, and V. Ya. Artyukhov, Russ. Phys. J., 59, No. 4, 513–524 (2016).
D. V. Kryl’skii and A. I. Slivkin, Heterocyclic Medicinal Substances: Pharmaceutical Chemistry Handbook [in Russian], Publishing House of Voronezh State University, Voronezh (2007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Izvestiya Vysshikh Uchebnykh Zavedenii, Fizika, No. 10, pp. 9–14, October, 2018.
Rights and permissions
About this article
Cite this article
Tchaikovskaya, O.N., Krayukhina, V.S., Pomogaev, V.A. et al. Experimental and Theoretical Investigation of Optical Spectra of Methylene Green in Solutions. Russ Phys J 61, 1752–1758 (2019). https://doi.org/10.1007/s11182-019-01597-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11182-019-01597-z