Abstract
Purpose
Health outcomes may depend on which treatment is received, whether choice of treatment is given, and whether a received treatment is the preferred therapy. We examined the effects of these key factors on the EuroQol-5D (EQ-5D-3L) in patients with PTSD.
Methods
Two hundred patients aged 18–65 years with PTSD diagnosis enrolled in a doubly randomized preference trial (DRPT) examining treatment, choice of treatment, and treatment-preference effects of prolonged exposure therapy (PE) and pharmacotherapy with sertraline (SER) (clinicaltrials.gov Identifier: NCT00127673). We performed difference-in-difference analysis to estimate the treatment effects of prolonged exposure therapy (PE) as compared to pharmacotherapy with sertraline (SER), receipt of choice versus no-choice of treatment, and receipt of preferred versus non-preferred treatment on health-related quality-of-life (HRQOL) outcome using the EQ-5D-3L completed at baseline and 10-week post-treatment.
Results
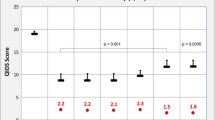
The treatment effects of PE on the EQ-5D scores in overall patients and subgroup of patients who preferred PE were 0.150 (p = 0.025) and 0.223 (p < 0.001), respectively. The effects of treatment choice were 0.088 (p = 0.050) and 0.156 (p = 0.043) in overall patients and subgroup of patients received SER, respectively. The effects of treatment preference were 0.101 (p = 0.038) and 0.249 (p = 0.004) in overall patients and subgroup of patients SER, respectively.
Conclusions
Overall, PE is associated with better improved HRQOL, especially in patients who prefer it. Independently, allowing patients to choose their preferred treatment resulted in better HRQOL than either assigning them a treatment or giving them a treatment that is not preferred.

Similar content being viewed by others
References
Kilpatrick, D. G., Resnick, H. S., Milanak, M. E., Miller, M. W., Keyes, K. M., & Friedman, M. J. (2013). National estimates of exposure to traumatic events and PTSD prevalence using DSM-IV and DSM-5 criteria. Journal of Traumatic Stress, 5, 537–547.
Eibner, C., Ringel, J. S., Kilmer, B., Pacula, R. L., & Diaz, C. (2008). The cost of post-deployment mental health and cognitive conditions. In T. Tanielian & L. H. Jaycox (Eds.), Invisible wounds of war: Psychological and cognitive injuries, their consequences, and services to assist recovery. Santa Monica: RAND.
Brady, K., Pearlstein, T., Asnis, G. M., Baker, D., Rothbaum, B., Sikes, C. R., & Farfel, G. M. (2000). Efficacy and safety of sertraline treatment of posttraumatic stress disorder: A randomized controlled trial. JAMA, 283(14), 1837–1844.
Resick, P. A., Nishith, P., Weaver, T. L., Astin, M. C., & Feuer, C. A. (2002). A comparison of cognitive-processing therapy with prolonged exposure and a waiting condition for the treatment of chronic posttraumatic stress disorder in female rape victims. Journal of Consulting and Clinical Psychology, 70(4), 867–879.
Jaeger, J. A., Echiverri, A., Zoellner, L. A., Post, L., & Feeny, N. C. (2009). Factors associated with choice of exposure therapy for PTSD. International Journal of Behavioral and Consultation Therapy, 5(3–4), 294–310.
Feeny, N. C., Zoellner, L. A., Mavissakalian, M. R., & Roy-Byrne, P. P. (2009). What would you choose? Sertraline or prolonged exposure in community and PTSD treatment seeking women. Depress Anxiety, 26(8), 724–731.
Rucker, G. (1989). A two-stage trial design for testing treatment, self-selection and treatment preference effects. Statistics in Medicine, 4, 477–485.
Torgerson, D. J., & Sibbald, B. (1998). Understanding controlled trials. What is a patient preference trial? BMJ, 316, 360.
Long, Q., Little, R. J., & Lin, X. H. (2008). Causal inference in hybrid intervention trials involving treatment choice. Journal of the American Statistical Association, 103, 474–484.
Shadish, W. R., Clark, M. H., & Steiner, P. M. (2008). Can nonrandomized experiments yield accurate answers? A randomized experiment comparing random and nonrandom assignments. Journal of the American Statistical Association, 103, 1334–1344.
Marcus, S. M., Stuart, E. A., Wang, P., Shadish, W. R., & Steiner, P. M. (2012). Estimating the causal effect of randomization versus treatment preference in a doubly randomized preference trial. Psychol Methods, 17(2), 244–254.
Mitchell-Jones, N., Farren, J. A., Tobias, A., Bourne, T., & Bottomley, C. (2017). Ambulatory versus inpatient management of severe nausea and vomiting of pregnancy: A randomised control trial with patient preference arm. British Medical Journal Open, 7(12), e017566.
Hubacher, D., Spector, H., Monteith, C., Chen, P. L., & Hart, C. (2017). Long-acting reversible contraceptive acceptability and unintended pregnancy among women presenting for short-acting methods: A randomized patient preference trial. American Journal of Obstetrics and Gynecology, 216(2), 101–109.
Elmagied, A. M., Vaughan, L. E., Weaver, A. L., et al. (2016). Fibroid interventions: Reducing symptoms today and tomorrow: Extending generalizability by using a comprehensive cohort design with a randomized controlled trial. American Journal of Obstetrics and Gynecology, 215, 338.e1–338.e18.
Howie, F. L., Henshaw, R. C., Naji, S. A., et al. (1997). Medical abortion or vacuum aspiration? Two year follow up of a patient preference trial. British Journal of Obstetrics and Gynaecology, 104, 829–833.
Henshaw, R. C., Naji, S. A., Russell, I. T., et al. (1994). A comparison of medical abortion (using mifepristone and gemeprost) with surgical vacuum aspiration: efficacy and early medical sequelae. Human Reproduction, 9, 2167–2172.
Janevic, M. R., Janz, N. K., Dodge, J. A., Lin, X., Pan, W., Sinco, B. R., & Clark, N. M. (2003). The role of choice in health education intervention trials: a review and case study. Social Science and Medicine, 56(7), 1581–1594.
Yancy, W. S. Jr., Mayer, S. B., Coffman, C. J., Smith, V. A., Kolotkin, R. L., Geiselman, P. J., McVay, M. A., Oddone, E. Z., & Voils, C. I. (2015). Effect of allowing choice of diet on weight loss: A randomized trial. Annals of Internal Medicine, 162(12), 805–814.
He, Y., Gewirtz, A., Lee, S., Morrell, N., & August, G. (2016). A randomized preference trial to inform personalization of a parent training program implemented in community mental health clinics. Translational Behavioral Medicine, 6(1), 73–80.
Swift, J. K., & Callahan, J. L. (2009). The impact of client treatment preferences on outcome: A meta-analysis. Journal of Clinical Psychology, 65(4), 368–381.
Swift, J. K., Callahan, J. L., Vollmer, B. (2011). Preferences. Journal of Clinical Psychology, 67, 155–165.
Freed, M. C., Yeager, D. E., Liu, X., Gore, K. L., Engel, C. C., & Magruder, K. M. (2009). Preference-weighted health status of PTSD among veterans: An outcome for cost-effectiveness analysis using clinical data. Psychiatric Services, 60, 1230–1238.
Craig, B. M., Pickard, S. A., & Lubetkin, E. I. (2014). Health problems are more common, but less severe when measured using newer EQ-5D versions. Journal of Clinical Epidemiology, 67, 93–99.
Neumann, P. J., Goldie, S. J., & Weinstein, M. C. (2000). Preference-based measures in economic evaluation in health care. Annual Review of Public Health, 21, 587–611.
Shaw, J. W., Johnson, J. A., & Coons, S. J. (2005). US valuation of the EQ-5D health states: Development and testing of the D1 valuation model. Medical Care, 43, 203–220.
Feeny, N. C., & Zoellner, L. A. Talk presented at: 2010 American Psychiatric Association Annual Meeting (Symposium 12: Anxiety Treatment—New Research Findings for the Clinician); May 22, 2010; New Orleans, LA.
Youngstrom, E. A., Feeny, N. C., Zoellner, L. A., Mavissakalian, M., & Roy-Byrne, P. Doubly Randomized Preference Trial in PTSD—Prolonged Exposure versus Sertraline (Symposium 5032). Talk presented at: American Psychological Association Annual Convention; August 3, 2013; Honolulu, HI.
Le, Q. A., Doctor, J. N., Zoellner, L. A., & Feeny, N. C. (2014). Cost-effectiveness of prolonged exposure therapy versus pharmacotherapy and treatment choice in posttraumatic stress disorder (the Optimizing PTSD Treatment Trial): A doubly randomized preference trial. The Journal of Clinical Psychiatry, 75(3), 222–230.
Kind, P. (2003). Guidelines for value sets in economic and on-economic studies using EQ-5D. In R. Brooks, R. Rabin & F. D. Charro. The measurement and valuation of health status using EQ-5D: A European perspective (pp. 29–42). Amsterdam: Springer.
Abadie, A. (2005). Semiparametric difference-in-differences estimators. The Review of Economic Studies, 72, 1–19.
Athey, S., & Imbens, G. W. (2006). Identification and inference in nonlinear difference-in-differences models. Econometrica, 74, 431–497.
Beard, C., Weisberg, R. B., & Keller, M. B. (2010). Health-related Quality of Life across the anxiety disorders: Findings from a sample of primary care patients. Journal of Anxiety Disorders, 24(6), 559–564.
Stoll, C., Schelling, G., Goetz, A. E., Kilger, E., Bayer, A., et al. (2000). Health-related quality of life and post-traumatic stress disorder in patients after cardiac surgery and intensive care treatment. The Journal of Thoracic and Cardiovascular Surgery, 120(3), 505–512.
Schnurr, P. P., Hayes, A. F., Lunney, C. A., McFall, M., & Uddo, M. (2006). Longitudinal analysis of the relationship between symptoms and quality of life in veterans treated for posttraumatic stress disorder. Journal of Consulting and Clinical Psychology, 74(4), 707–713.
Haagsma, J. A., Polinder, S., Olff, M., Toet, H., Bonsel, G. J., & van Beeck, E. F. (2012). Posttraumatic stress symptoms and health-related quality of life: A two year follow up study of injury treated at the emergency department. BMC Psychiatry, 12, 1.
Kapfhammer, H. P., Rothenhäusler, H. B., Krauseneck, T., Stoll, C., & Schelling, G. (2004). Posttraumatic stress disorder and health-related quality of life in long-term survivors of acute respiratory distress syndrome. American Journal of Psychiatry, 161(1), 45–52.
Pittman, J. O., Goldsmith, A. A., Lemmer, J. A., Kilmer, M. T., & Baker, D. G. (2012). Post-traumatic stress disorder, depression, and health-related quality of life in OEF/OIF veterans. Quality of Life Research, 21(1), 99–103.
Mancino, M. J., Pyne, J. M., Tripathi, S., Constans, J., Roca, V., & Freeman, T. (2006). Quality-adjusted health status in veterans with posttraumatic stress disorder. The Journal of Nervous and Mental Disease, 194(11), 877–879.
Giacco, D., Matanov, A., & Priebe, S. (2013). Symptoms and subjective quality of life in post-traumatic stress disorder: A longitudinal study. PLoS ONE 8(4), e60991.
Johansen, V. A., Wahl, A. K., Eilertsen, D. E., Weisaeth, L., & Hanestad, B. R. (2007). The predictive value of post-traumatic stress disorder symptoms for quality of life: A longitudinal study of physically injured victims of non-domestic violence. Health Qual Life Outcomes, 5, 26.
Le, Q. A., Doctor, J. N., Zoellner, L. A., & Feeny, N. C. (2013). Minimal clinically important differences for the EQ-5D and QWB-SA in post-traumatic stress disorder (PTSD): Results from a Doubly Randomized Preference Trial (DRPT). Health Qual Life Outcomes, 11, 59.
Zoellner, L. A., Feeny, N. C., Cochran, B., & Pruitt, L. (2003). Treatment choice for PTSD. Behaviour Research and Therapy, 41, 879–886.
Williams, M., Cahill, S., & Foa, E. Psychotherapy for post-traumatic stress disorder. D. Stein & E. Hollander, B. Rothbaum (Eds.), Textbook of anxiety disorders (2nd edn.) Washington:American Psychiatric Publishing, 2010.
Rubin, D. B. (1978). Bayesian inference for causal effects: The role of randomization. Annals of Statistics, 6, 34–58.
Funding
This research was supported by Grants R01MH066347 and R01MH066348 from the National Institute of Mental Health (“Effectiveness of PTSD Treatment: Prolonged Exposure Therapy vs. Zoloft”).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have conflicts of interest.
Ethical approval
The study was approved by the Institutional Review Boards (IRB) of the Case Western Reserve University/University Hospital of Cleveland and University of Washington. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Le, Q.A., Doctor, J.N., Zoellner, L.A. et al. Effects of treatment, choice, and preference on health-related quality-of-life outcomes in patients with posttraumatic stress disorder (PTSD). Qual Life Res 27, 1555–1562 (2018). https://doi.org/10.1007/s11136-018-1833-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-018-1833-4