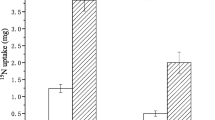
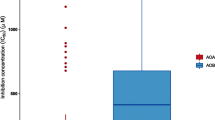
Abstract
Regulating nitrification could be a key strategy in improving nitrogen (N) recovery and agronomic N-use efficiency in situations where the loss of N following nitrification is significant. A highly sensitive bioassay using recombinant luminescent Nitrosomonas europaea, has been developed that can detect and quantify the amount of nitrification inhibitors produced by plants (hereafter referred to as BNI activity). A number of species including tropical and temperate pastures, cereals and legumes were tested for BNI in their root exudate. There was a wide range in BNI capacity among the 18 species tested; specific BNI (AT units activity g−1 root dry wt) ranged from 0 (i.e. no detectable activity) to 18.3 AT units. Among the tested cereal and legume crops, sorghum [Sorghum bicolor (L.)], pearl millet [Pennisetum glaucum (L.) R. Br.], and groundnut [Arachis hypogaea (L.)] showed detectable BNI in root exudate. Among pasture grasses, Brachiaria humidicola (Rendle) Schweick, B. decumbens Stapf showed the highest BNI capacity. Several high- and low-BNI genotypes were identified within the B. humidicola species. Soil collected from field plots of 10 year-old high-BNI genotypes of B. humidicola, showed a near total suppression (>90%) of nitrification; most of the soil inorganic N remained in the NH +4 form after 30 days of incubation. In contrast, soils collected from low-BNI genotypes did not show any inhibitory effect; most of the soil inorganic N was converted to NO –3 after 30 days of incubation. In both the high- and low-BNI genotypes, BNI was detected in root exudate only when plants were grown with NH +4 , but not when grown with NO –3 as the sole source of N. BNI compounds when added to the soil inhibited nitrification and the relationship was linear (r 2 = 0.92**; n = 12). The BNI from high- and low-BNI types when added to N. europaea in pure culture, blocked both the ammonia monooxygenase (AMO) and the hydroxylamine oxidoreductase (HAO) pathways. Our results indicated that BNI capacity varies widely among and within species; and that some degree of BNI capacity is likely a widespread phenomenon in tropical pasture grasses. We suggest that the BNI capacity could either be managed and/or introduced into pastures/crops with an expression of this phenomenon, via genetic improvement approaches that combine high productivity along with some capacity to regulate soil nitrification process.






Similar content being viewed by others
References
Anon (1974) Technicon Autoanalyzer II, Technicon Industrial Systems. Tarrytown, NY, 10591
Basu P, Katterle B, Anderson KK, Dalton H (2003) The membrane-associated form of methane mono-oxygenase from Methylococcus capsulatus (Bath) is a copper/iron protein. Biochem J 369:417–427
Bock E, Koops HP, Harms H, Ahlers B (1991) The biochemistry of nitrifying organisms. In: Shively JM, Barton LL (eds) Variations in autotrophic life. Academic Press, San diego, CA, pp 171–200
Giles J (2005) Nitrogen study fertilizes fears of pollution. Nature 433:791
Glass ADM (2003) Nitrogen use efficiency of crop plants: physiological constraints upon nitrogen absorption. Crit Rev Plant Sci 22:453–470
Hauck RD (1980) Mode of nitrification inhibitors. In: Nitrification inhibitors—potentials and limitations. ASA Special Publication No. 38, American Society of Agronomy, Madison, Wisconsin, pp19–32
Ishikawa T, Subbarao GV, Ito O, Okada (2003) Suppression of nitrification and nitrous oxide emission by the tropical grass Brachiaria humidicola. Plant Soil 255:413–419
Iizumi T, Nakamura K (1997) Cloning, nucleotide sequence, and regulatory analysis of the Nitrosomonas europaea dnaK gene. Appl Environ Microbiol 63:1777–1784
Iizumi T, Mizumoto M, Nakamura K (1998) A bioluminescence assay using Nitrosomonas europaea for rapid and sensitive detection of nitrification inhibitors. Appl Environ Microbiol 64:3656–3662
Jarvis SC (1996) Future trends in nitrogen research. Plant Soil 181:47–56
Lata JC, Guillaume K, Degrange V, Abbadie L, Lensi R (2000) Relationships between root density of the African grass Hyparrhenia diplandra and nitrification at the decimetric scale: an inhibition–stimulation balance hypothesis. Proc Royal Soc London Series B, Biological Sci 267:1–6
Lata JC, Degrange V, Raynaud X, Maron PA, Lensi R, Abbadie L (2004) Grass population control nitrification in Savanna soils. Funct Ecol 13:762–763
Lensi R, Mazurier S, Gourbiere F, Josserand A (1986) Rapid determination of the nitrification potential of an acid forest soil and assessment of its variability. Soil Biol Biochem 18:239–240
Li Z, Peng S, Rae DJ, Zhou G (2001) Litter decomposition and nitrogen mineralization of soils in subtropical plantation forests of southern China, with special attention to comparisons between legumes and non-legumes. Plant Soil 229:105–116
Litchfield MD (1967) The automated analysis of nitrite and nitrate in blood. Analyst 92:132–136
McCarty GW (1999) Modes of action of nitrification inhibitors. Biol Fertil Soils 29:1–9
Mosier AR, Duxbury JM, Freney JR, Heinemeyer O, Minami K (1996) Nitrous oxide emissions from agricultural fields: assessment, measurement, and mitigation. Plant Soil 181:95–108
Prasad R, Power JF (1995) Nitrification inhibitors for agriculture, health, and the environment. Adv Agron 54:233–281
Rao IM, Ayarza MA, Garcia R (1995) Adaptive attributes of tropical forage species to acid soils I. Differences in plant growth, nutrient acquisition and nutrient utilization among C4 grasses and C3 legumes. J Plant Nutr 18:2135–3155
Rao IM, Kerridge PC, Macedo M (1996) Adaptation to low fertility acid soils and nutritional requirements of Brachiaria. In: Miles JW, Maass BL, do valle CB (eds) The biology, agronomy and improvement of Brachiaria. CIAT, Cali, Colombia, pp 53–71
Raun WR, Johnson GV (1999) Improving nitrogen use efficiency for cereal production. Agron J 91:357–363
Rice CW, Pancholy SK (1972) Inhibition of nitrification by climax ecosystem. Am J Bot. 59:1033–1040
Robinson JB (1963) Nitrification in a New Zealand grassland soil. Plant and Soil 14:173–183
Sahrawat KL, Keeney DR (1985) Perspectives for research on development of nitrification inhibitors. Commun Soil Sci Plant Anal 16:517–524
Serca D, Delmas R, Le Roux X (1998) Comparison of nitrogen monoxide emissions from several African tropical ecosystems and influence of season and fire. Global Biogeochem Cycles 12:637–651
Subbarao GV, Ito O, Sahrawat KL, Berry WL, Nakahara K, Ishikawa T, Watanabe T, Suenaga K, Rondon M, Rao IM (2006a) Scope and strategies for regulation of nitrification in agricultural systems—Challenges and opportunities. Crit Rev Plant Sci 25:1–33
Subbarao GV, Ishikawa T, Ito O, Nakahara K, Wang HY, Berry WL (2006b) A bioluminescence assay to detect nitrification inhibitors released from plant roots—A case study with Brachiaria humidicola. Plant Soil. Online first http://www.springerlink.com/content/r435654g2r514044.
Subbarao GV, Wang HY, Ito O, Nakahara K, Berry WL (2006c) NH +4 triggers the synthesis and release of BNI (biological nitrification inhibition) compounds in B. humidicola roots. Plant Soil (in press)
Sylvester-Bradley R, Mosquera D, Mendez JE (1988) Inhibition of nitrate accumulation in tropical grassland soils: effect of nitrogen fertilization and soil disturbance. J Soil Sci 39:407–416
Theron JJ (1963) The mineralization of nitrogen in soils under grass. S Afr J Agr Sci 6:155–164
Varley JA (1966) Automated method for the determination of nitrogen, phosphorus and potassium in plant material. Analyst 91:119–126
Ward BB, Courtney KJ, Langenheim JH (1997) Inhibition of Nitrosomonas europaea by monoterpenes from coastal redwood (Sequoia sempervirens) in whole-cell studies. J Chem Ecol 23:2583–2598
Acknowledgements
We are grateful to Dr. Taro Iizumi (Kurita Water Industries Limited, Wakamtya, Japan) who has kindly provided us with the luminescent recombinant N. europaea strain for this research. Also, we are grateful to Dr. K.L. Sahrawat (ICRISAT, India), who has gone through our manuscript critically and offered many suggestions to improve the clarity of our presentation and interpretation of the data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Subbarao, G.V., Rondon, M., Ito, O. et al. Biological nitrification inhibition (BNI)—is it a widespread phenomenon?. Plant Soil 294, 5–18 (2007). https://doi.org/10.1007/s11104-006-9159-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-006-9159-3