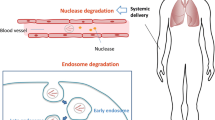
Abstract
Purpose
This study demonstrates the nasal administration (NA) of nanoemulsions complexed with the plasmid encoding for IDUA protein (pIDUA) as an attempt to reach the brain aiming at MPS I gene therapy.
Methods
Formulations composed of DOPE, DOTAP, MCT (NE), and DSPE-PEG (NE-PEG) were prepared by high-pressure homogenization, and assessed in vitro on human fibroblasts from MPS I patients and in vivo on MPS I mice for IDUA production and gene expression.
Results
The physicochemical results showed that the presence of DSPE-PEG in the formulations led to smaller and more stable droplets even when submitted to dilution in simulated nasal medium (SNM). In vitro assays showed that pIDUA/NE-PEG complexes were internalized by cells, and led to a 5% significant increase in IDUA activity, besides promoting a two-fold increase in IDUA expression. The NA of pIDUA/NE-PEG complexes to MPS I mice demonstrated the ability to reach the brain, promoting increased IDUA activity and expression in this tissue, as well as in kidney and spleen tissues after treatment. An increase in serum IL-6 was observed after treatment, although with no signs of tissue inflammatory infiltrate according to histopathology and CD68 assessments.
Conclusions
These findings demonstrated that pIDUA/NE-PEG complexes could efficiently increase IDUA activity in vitro and in vivo after NA, and represent a potential treatment for the neurological impairment present in MPS I patients.





Similar content being viewed by others
Abbreviations
- BNE:
-
Blank cationic nanoemulsions
- BNE-PEG:
-
Blank cationic nanoemulsions containing DSPE-PEG
- DOPE:
-
1,2-dioleoyl-sn-glycero-3-phosphoethanolamine
- DOTAP:
-
1,2-dioleoyl-sn-glycero-3-trimethylammonium propane
- DSPE-PEG:
-
1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino (polyethylene glycol)-2000]
- IDUA:
-
α-L-iduronidase
- IL-6:
-
Interleukin-6
- MCT:
-
Medium Chain Triglycerides
- NA:
-
Nasal administration
- pIDUA:
-
Plasmid encoding for IDUA protein
- pIDUA/NE:
-
Nanoemulsion complexed with pIDUA
- pIDUA/NE-PEG:
-
Nanoemulsion containing DSPE-PEG complexed with pIDUA
- SNM:
-
Simulated nasal medium
References
Abbott NJ. Blood-brain barrier structure and function and the challenges for CNS drug delivery. J Inherit Metab Dis. 2013;36(3):437–49.
Baldo G, Mayer FQ, Martinelli B, Meyer FS, Burin M, Meurer L, et al. Intraperitoneal implant of recombinant encapsulated cells overexpressing alpha-L-iduronidase partially corrects visceral pathology in mucopolysaccharidosis type I mice. Cytotherapy. 2012;14(7):860–7.
Benedict C, Frey WH 2nd, Schioth HB, Schultes B, Born J, Hallschmid M. Intranasal insulin as a therapeutic option in the treatment of cognitive impairments. Exp Gerontol. 2011;46(2–3):112–5.
Bidone J, Schuh RS, Farinon M, Poletto É, Pasqualim G, de Oliveira PG, et al. Intra-articular nonviral gene therapy in mucopolysaccharidosis I mice. Int J Pharm. 2018;548(1):151–8.
Bruxel F, Vilela JMC, Andrade MS, Malachias A, Perez CA, Magalhaes-Paniago R, et al. Investigation of the structural organization of cationic nanoemulsion/antisense oligonucleotide complexes. Col Surf B. 2013;112:530–6.
Camassola M, Braga LM, Delgado-Cañedo A, Dalberto TP, Matte U, Burin M, et al. Nonviral in vivo gene transfer in the mucopolysaccharidosis I murine model. J Inherit Metab Dis. 2005;28(6):1035–43.
Chan C-L, Majzoub RN, Shirazi RS, Ewert KK, Chen Y-J, Liang KS, et al. Endosomal escape and transfection efficiency of PEGylated cationic liposome–DNA complexes prepared with an acid-labile PEG-lipid. Biomaterials. 2012;33(19):4928–35.
Fraga M, Bruxel F, Lagranha VL, Teixeira HF, Matte U. Influence of phospholipid composition on cationic emulsions/DNA complexes: physicochemical properties, cytotoxicity, and transfection on Hep G2 cells. Int J Nanomedicine. 2011;6:2213–20.
Schuh RS, Poletto É, Pasqualim G, Tavares AMV, Meyer FS, Gonzalez EA, Giugliani R, et al. In vivo genome editing of mucopolysaccharidosis I mice using the CRISPR/Cas9 system. J Control Release. 2018;288:23-33.
Fraga M, de Carvalho TG, Bidone J, Schuh RS, Matte U, Teixeira HF. Factors influencing transfection efficiency of pIDUA/nanoemulsion complexes in a mucopolysaccharidosis type I murine model. Int J Nanomedicine. 2017;12:2061–7.
Ghori MU, Mahdi MH, Smith AM, Conway BR. Nasal Drug Delivery Systems: An Overview. Am J Pharmacol Sci. 2015;3(5):110–9.
Giugliani R. Mucopolysacccharidoses: from understanding to treatment, a century of discoveries. Genet Mol Biol. 2012;35(4):924–31.
Giugliani R, Federhen A, Rojas MVM, Vieira T, Artigalás O, Pinto LL, et al. Mucopolysaccharidosis I, II, and VI: brief review and guidelines for treatment. Genet Mol Biol. 2010;33(4):589–604.
Hanson LR, Fine JM, Svitak AL, Faltesek KA. Intranasal administration of CNS therapeutics to awake mice. J Vis Exp. 2013;74:4440.
Khatri K, Goyal AK, Gupta PN, Mishra N, Mehta A, Vyas SP. Surface modified liposomes for nasal delivery of DNA vaccine. Vaccine. 2008;26(18):2225–33.
Kwon SM, Nam HY, Nam T, Park K, Lee S, Kim K, et al. In vivo time-dependent gene expression of cationic lipid-based emulsion as a stable and biocompatible non-viral gene carrier. J Control Release. 2008;128(1):89–97.
Liu C-H, Yu S-Y. Cationic nanoemulsions as non-viral vectors for plasmid DNA delivery. Colloids Surf B Biointerfaces. 2010;79(2):509–15.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods. 2001;25(4):402–8.
Lochhead JJ, Thorne RG. Intranasal delivery of biologics to the central nervous system. Adv Drug Deliv Rev. 2012;64(7):614–28.
Lovelyn C, Attama A. Current state of Nanoemulsions in drug delivery. J Biomater Nanobiotechnol. 2011;2(5A):626–39.
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265–75.
Makidon PE, Belyakov IM, Blanco LP, Janczak KW, Landers J, Bielinska AU, et al. Nanoemulsion mucosal adjuvant uniquely activates cytokine production by nasal ciliated epithelium and induces dendritic cell trafficking. Eur J Immunol. 2012 Aug;42(8):2073–86.
Masiuk T, Kadakia P, Wang Z. Development of a physiologically relevant dripping analytical method using simulated nasal mucus for nasal spray formulation analysis. J Pharm Anal. 2016;6(5):283–91.
Mayer FQ, Adorne MD, Bender EA, de Carvalho TG, Dilda AC, Pohlmann AR, et al. Laronidase-functionalized multiple-Wal l lipid-Core Nanocapsules: promising formulation for a more effective treatment of Mucopolysaccharidosis type I. Pharm Res. 2015;32:941–54.
Miller MA, Stabenow JM, Parvathareddy J, Wodowski AJ, Fabrizio TP, Bina XR, et al. Visualization of murine intranasal dosing efficiency using luminescent Francisella tularensis: effect of instillation volume and form of anesthesia. PLoS One. 2012;7(2):e31359.
Nag OK, Awasthi V. Surface engineering of liposomes for stealth behavior. Pharmaceutics. 2013;5(4):542–69.
Osborn MJ, McElmurry RT, Peacock B, Tolar J, Blazar BR. Targeting of the CNS in MPS-IH using a nonviral transferrin-alpha-L-iduronidase fusion gene product. Mol Ther. 2008;16(8):1459–66.
Ramamoorth M, Narvekar A. Non viral vectors in gene therapy- an overview. J Clin Diagn Res. 2015;9(1):GE01–6.
Schuh R, Baldo G, Teixeira H. Nanotechnology applied to treatment of mucopolysaccharidoses. Expert Opin Drug Del. 2016;13(12):1709–18.
Schuh RS, Poletto É, Fachel FNS, Matte U, Baldo G, Teixeira HF. Physicochemical properties of cationic nanoemulsions and liposomes obtained by microfluidization complexed with a single plasmid or along with an oligonucleotide: implications for CRISPR/Cas technology. J Colloid Interface Sci. 2018;530:243–55.
Schuh RS, de Carvalho TG, Giugliani R, Matte U, Baldo G, Teixeira HF. Gene editing of MPS I human fibroblasts by co-delivery of a CRISPR/Cas9 plasmid and a donor oligonucleotide using nanoemulsions as nonviral carriers. Eur J Pharm Biopharm. 2018;122:158–66.
Teixeira HF, Bruxel F, Fraga M, Schuh RS, Zorzi GK, Matte U, et al. Cationic nanoemulsions as nucleic acids delivery systems. Int J Pharm. 2017;534(1–2):356–67.
Xue HY, Liu S, Wong HL. Nanotoxicity: a key obstacle to clinical translation of siRNA-based nanomedicine. Nanomedicine (Lond). 2014;9(2):295–312.
Yadav S, Gandham SK, Panicucci R, Amiji MM. Intranasal brain delivery of cationic nanoemulsion-encapsulated TNFalpha siRNA in prevention of experimental neuroinflammation. Nanomedicine. 2016;12(4):987–1002.
ACKNOWLEDGMENTS AND DISCLOSURES
Authors would like to thank the Brazilian National Council for Scientific and Technological Development (CNPq), Foundation for Research Support of the State of Rio Grande do Sul (FAPERGS, #17/2551–0001273-9), and HCPA Foundation for Incentive of Research (FIPE/HCPA, #150215) for the financial support. J. Bidone wishes to thank the Foundation for Research Support of the State of Rio Grande do Sul (DOCFIX/FAPERGS) and R. S. Schuh would like to thank CNPq (#151021/2018–0) for their postdoctoral grant. H. F. Teixeira, G. Baldo, and U. Matte are recipients of CNPq research fellowship. The authors have made substantial contributions to all of the following: R. S. Schuh and J. Bidone contributed to the conception and design of the study. G. Pasqualim, T. G. de Carvalho, É. Poletto, C. V. Pinheiro, D. S. Diel, M. Farinon, and R. M. Xavier contributed to the acquisition of data, analysis, and interpretation of data. G. Baldo, U. Matte, and H. F. Teixeira contributed to drafting the article and revising it critically for important intellectual content. All authors approved the final version to be submitted. All institutional and national guidelines for the care and use of patients’ material were followed and all patients and/or their caregivers gave written informed consent for this study. All institutional and national guidelines for the care and use of laboratory animals were followed (Hospital de Clínicas de Porto Alegre Ethics Committee #15–0215). All authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Synopsis: This study demonstrates the nasal administration of nanoemulsions complexed with the plasmid encoding for IDUA protein (pIDUA) as an attempt to reach the brain aiming at mucopolysaccharidosis type I gene therapy.
Electronic supplementary material
ESM 1
(DOCX 533 kb)
Rights and permissions
About this article
Cite this article
Schuh, R.S., Bidone, J., Poletto, E. et al. Nasal Administration of Cationic Nanoemulsions as Nucleic Acids Delivery Systems Aiming at Mucopolysaccharidosis Type I Gene Therapy. Pharm Res 35, 221 (2018). https://doi.org/10.1007/s11095-018-2503-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-018-2503-5