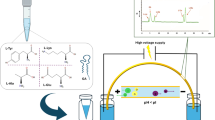
An HPLC method for quantitative determination of the new biologically active GABA derivative Solifen was developed. The method sensitivity, compound retention time and UV absorption maximum were determined. The optimum extractant for the compound from biological samples was determined.



Similar content being viewed by others
References
N. N. Karkishchenko, V. V. Khoron'ko, and S. A. Sergeeva, Pharmacokinetics [in Russian], Feniks, Rostov-on-Don (2001).
M. V. Karlina, O. N. Pozharitskaya, V. M. Kosman, et al., Eksp. Klin. Farmakol., 73(8), 46 – 48 (2010).
V. P. Zherdev, S. S. Boiko, N. V. Mesonzhnik, et al., Eksp. Klin. Farmakol., 72(3), 16 – 21 (2009).
I. N. Tyurenkov, V. N. Perfilova, and T. A. Popova, Byull. Eksp. Biol. Med., 155(3), 340 – 343 (2013).
L. B. Ivanova, V. I. Karamysheva, V. N. Perfilova, et al., Probl. Reprod., 18(1), 28 – 30 (2012).
I. N. Tyurenkov, V. N. Perfilova, L. A. Smirnova, et al., Khim.-farm. Zh., 47(3), 55 – 56 (2013); Pharm. Chem. J., 47(3), 179 – 180 (20130.
I. N. Tyurenkov, V. N. Perfilova, L. A. Smirnova, et al., Khim.-farm. Zh., 44(12), 68 – 70 (2010); Pharm. Chem. J., 44(12), 702 – 704 (2013).
Guideline on Bioanalytical Method Validation (EMEA 2012); http://www.ema.europa.eu.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 53, No. 11, pp. 62 – 64, November, 2019.
Rights and permissions
About this article
Cite this article
Smirnova, L.A., Ryabukha, A.F., Kuznetsov, K.A. et al. Method Development for Quantitative Determination of the Gaba Derivative Solifen. Pharm Chem J 53, 1090–1092 (2020). https://doi.org/10.1007/s11094-020-02128-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-020-02128-8