Abstract
Purpose
To report outcomes of salvage re-irradiation (re-RT) in recurrent/progressive medulloblastoma (MB).
Methods
Medical records of patients treated with curative-intent re-RT as multi-modality management for recurrent/progressive MB between 2008 and 2018 were analyzed retrospectively.
Results
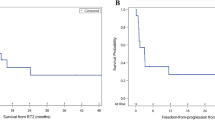
A total of 28 patients (median age 18 years at index diagnosis) were included. Molecular subgrouping was done using real-time reverse transcriptase polymerase chain reaction (RT-PCR) based on the differential expression of select set of 12 protein coding genes and 9 microRNAs. Fifteen of 17 (88%) patients with sonic hedgehog (SHH)-MB developed isolated local recurrence within the index tumor-bed, while 5 of 7 (72%) patients with Group 4 MB developed localized relapse outside the posterior fossa. Diffuse neuraxial dissemination was seen in 2 patients with SHH-MB, and one each of Group 4 and wingless (WNT)-MB. Molecular subgrouping was not known in 3 patients. The dose and volume of re-RT was based on site and patterns of relapse, comprising unifocal in 18 (64%), multi-focal in 3 (11%), and repeat craniospinal irradiation (re-CSI) in 7 (25%) patients. Median interval from primary irradiation to re-RT was 49.5 months (range 24–98 months) with median cumulative biologically effective dose of 117 Gy (range 78–132 Gy). All patients received platinum-based salvage chemotherapy either before or after re-RT. One patient developed symptomatic radiation necrosis following re-CSI. At a median follow-up of 24 months (range 6–84 months), 2-year post-re-RT progression-free survival (PFS) and overall survival (OS) was 46% and 51% respectively. Younger age (< 18 years) at index diagnosis, primary risk stratification (standard-risk) and molecular subgrouping (Group 4) were associated with significantly better post-re-RT outcomes.
Conclusion
Salvage re-RT provides good local control and encouraging survival outcomes with acceptable toxicity in selected patients with recurrent/progressive MB.


Similar content being viewed by others
References
Leece R, Xu J, Ostrom QT, Chen Y, Kruchko C, Barnholtz-Sloan JS (2017) Global incidence of malignant brain and other central nervous system tumors by histology, 2003–2007. Neuro-Oncology 19(11):1553–1564
Khanna V, Achey R, Oostrom Q, Block-Beach H, Kruchko C, Barnholtz-Sloan JS et al (2017) Incidence and survival trends for medulloblastoma in the United States from 2001 to 2013. J Neurooncol 135(Suppl 10):1–9
Kline CN, Packer RJ, Hwang EI, Raleigh DR, Braunstein S, Raffel C et al (2017) Case-based review: pediatric medulloblastoma. Neuro-Oncol Pract 4(3):138–150
Northcott PA, Korshunov A, Witt H, Hielscher T, Eberhart CG, Mack S et al (2011) Medulloblastoma comprises four distinct molecular variants. J Clin Oncol 29(11):1408–1414
Kunder R, Jalali R, Sridhar E, Moiyadi A, Goel N, Goel A et al (2013) Real-time PCR assay based on the differential expression of microRNAs and protein-coding genes for molecular classification of formalin-fixed paraffin embedded medulloblastomas. Neuro-Oncology 15(12):1644–1651
Taylor MD, Northcott PA, Korshunov A, Remke M, Cho YJ, Clifford SC et al (2012) Molecular subgroups of medulloblastoma: the current consensus. Acta Neuropathol 123(4):465–472
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK et al (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131(6):803–820
Packer RJ, Gajjar A, Vezina G, Rorke-Adams L, Burger PC, Robertson PL et al (2006) Phase III study of craniospinal radiation therapy followed by adjuvant chemotherapy for newly diagnosed average-risk medulloblastoma. J Clin Oncol 24(25):4202–4208
Michalski JM, Janss A, Vezina G, Gajjar A, Pollack I, Merchant TE et al (2016) Results of COG ACNS0331: a phase III trial of involved-field radiation therapy (IFRT) and low-dose craniospinal irradiation (LDCSI) with chemotherapy in average-risk medulloblastoma: a report from the Children’s Oncology Group. Int J Radiat Oncol Biol Phys 96(Issue_2, Supplement S1-S268):e1–e717
Gajjar A, Chintagumpala M, Ashley D, Kellie S, Kun LE, Merchant TE et al (2006) Risk-adapted craniospinal radiotherapy followed by high-dose chemotherapy and stem-cell rescue in children with newly diagnosed medulloblastoma (St Jude Medulloblastoma-96): long-term results from a prospective, multicentre trial. Lancet Oncol 7(10):813–820
Tarbell NJ, Friedman H, Polkinghorn WR, Yock T, Zhou T, Chen Z et al (2013) High-risk medulloblastoma: a pediatric oncology group randomized trial of chemotherapy before or after radiation therapy (POG 9031). J Clin Oncol 31(23):2936–2941
Sabel M, Fleischhack G, Tippelt S, Gustafsson G, Doz F, Kortmann R et al (2016) Relapse patterns and outcome after relapse in standard risk medulloblastoma: a report from the HIT-SIOP-PNET4 study. J Neurooncol 129(3):515–524
Johnston DL, Keene D, Strother D, Taneva M, Lafay-Cousin L, Fryer C et al (2018) Survival following tumor recurrence in children with medulloblastoma. J Pediatr Hematol Oncol 40(3):e159–e163
Ramaswamy V, Remke M, Bouffet E, Faria CC, Perreault S, Cho YJ et al (2013) Recurrence patterns across medulloblastoma subgroups: an integrated clinical and molecular analysis. Lancet Oncol 14(12):1200–1207
Dasgupta A, Gupta T, Sridhar E, Shirsat N, Krishnatry R, Goda JS et al (2019) Pediatric patients with SHH medulloblastoma fail differently as compared with adults: possible implications for treatment modifications. J Pediatr Hematol Oncol (in press)
Gururangan S, Krauser J, Watral MA, Driscoll T, Larrier N, Reardon DA et al (2008) Efficacy of high-dose chemotherapy or standard salvage therapy in patients with recurrent medulloblastoma. Neuro-Oncology 10(5):745–751
Bode U, Zimmermann M, Moser O, Rutkowski S, Warmuth-Metz M, Pietsch T et al (2014) Treatment of recurrent primitive neuroectodermal tumors (PNET) in children and adolescents with high-dose chemotherapy (HDC) and stem cell support: results of the HITREZ 97 multicentre trial. J Neurooncol 120(3):635–642
Tsang DS, Laperriere NJ (2019) Re-irradiation for paediatric tumours. Clin Oncol 31(3):191–198
Ruifrok AC, van der Kogel AJ (1993) A ‘re-appraisal’ of the LQ model for the understanding of dose-fractionation in radiotherapy. Int J Radiat Oncol Biol Phys 25(5):926–929
Mayer R, Sminia P (2008) Re-irradiation tolerance of the human brain. Int J Radiat Oncol Biol Phys 70(5):1350–1360
Bakst RL, Dunkel IJ, Gilheeney S, Khakoo Y, Becher O, Souweidane MM et al (2011) Reirradiation for recurrent medulloblastoma. Cancer 117(21):4977–4982
Wetmore C, Herington D, Lin T, Onar-Thomas A, Gajjar A, Merchant TE (2014) Reirradiation of recurrent medulloblastoma: does clinical benefit outweigh risk for toxicity? Cancer 120(23):3731–3737
Rao AD, Rashid AS, Chen Q, Villar RC, Kobyzeva D, Nilsson K et al (2017) Reirradiation for recurrent pediatric central nervous system malignancies: a multi-institutional review. Int J Radiat Oncol Biol Phys 99(3):634–641
Milker-Zabel S, Zabel A, Thilmann C, Zuna I, Hoess A, Wannenmacher M et al (2002) Results of three-dimensional stereotactically-guided radiotherapy in recurrent medulloblastoma. J Neurooncol 60(3):227233
Saran F, Baumert BG, Creak AL, Warrington AP, Ashley S, Traish D et al (2008) Hypofractionated stereotactic radiotherapy in the management of recurrent or residual medulloblastoma/PNET. Pediatr Blood Cancer 50(3):554–560
Napieralska A (2019) Radiosurgery or hypofractionated stereotactic radiotherapy after craniospinal irradiation in children and adults with medulloblastoma and ependymoma. Childs Nerv Syst 35(2):267–275
Dunkel IJ, Gardner SL, Garvin JH, Goldman S, Shi W, Finlay JL (2010) High-dose carboplatin, thiotepa, and etoposide with autologous stem cell rescue for patients with previously irradiated recurrent medulloblastoma. Neuro-Oncology 12(3):297–303
Bouffet E, Doz F, Demaille M, Tron P, Roche H, Plantaz D et al (1998) Improving survival in recurrent medulloblastoma: earlier detection, better treatment or still an impasse? Br J Cancer 77(8):13211326
Bowers DC, Gargan L, Weprin BE, Mulne AF, Elterman RD, Munoz L et al (2007) Impact of site of tumor recurrence upon survival for children with recurrent or progressive medulloblastoma. J Neurosurg Pediatr 107(1):5–10
Hashimoto Y, Penas-Prado M, Zhou S, Wei J, Khatua S, Hodges TR et al (2018) Rethinking medulloblastoma from a targeted theraputics perspective. J Neurooncol 139(3):713–720
Funding
No source of funding was involved in this retrospective study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11060_2019_3225_MOESM1_ESM.png
Supplementary material 1 (PNG 96 kb) Figure S2: Kaplan-Meier curves of post-re-irradiation progression-free survival (a) and overall survival (b) for the study cohort stratified by extent of re-irradiation (unifocal vs multi-focal vs craniospinal re-irradiation)
Rights and permissions
About this article
Cite this article
Gupta, T., Maitre, M., Sastri, G.J. et al. Outcomes of salvage re-irradiation in recurrent medulloblastoma correlate with age at initial diagnosis, primary risk-stratification, and molecular subgrouping. J Neurooncol 144, 283–291 (2019). https://doi.org/10.1007/s11060-019-03225-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-019-03225-9