Abstract
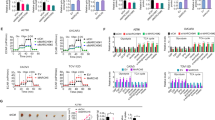
Mitochondrial dysfunction is known to contribute to cancer initiation, progression, and chemo-and radio-resistance. However, the precise role of mitochondria in cancer is controversial. Hence, here we tried to further clarify the role of mitochondria in cancer by transferring healthy mitochondria to cancer cells, and also to cells with depleted mitochondrial DNA (ρ0). Healthy mitochondria were isolated from WI-38 cells and were transferred to HeLa, SAS, HeLa ρ0, and SAS ρ0 cells. Then, cell proliferation was verified. In addition, the cells were treated by different concentrations of cisplatin and assessed for apoptosis induction and quantifying the mRNA expression of apoptosis-related genes. Results revealed that incubation of the HeLa, SAS and HeLa ρ0 cells with 5 µg/ml of the isolated mitochondria for 24 h significantly (p < 0.001) increased cell proliferation compared to non-treated controls. Interestingly, the mitochondria transfer rescued the ρ0 cells and made them capable of growing under conventional culture medium. However, the number of apoptotic cells was significantly higher in the HeLa ρ0 cells that received the mitochondria (HeLa-Fibro-Mit) compared to the HeLa ρ0. Furthermore, the expression level of BCL-2 anti-apoptotic gene was down-regulated in both HeLa-Fibro-Mit and SAS-Fibro-Mit cell lines while the expression levels of the BAX, caspase8, caspase9, and AIF pro-apoptotic genes were upregulated. Our findings indicated that although the response of cancer cells to the mitochondria transfer is cancer-type dependent, but the introduction of normal exogenous mitochondria to some cancer cells might be considered as a potential novel therapeutic strategy.





Similar content being viewed by others
References
Abbott A (2014) Doubts over heart stem-cell therapy: study queries early-phase trials of heart-disease treatment. Nature 509(7498):15–17
Zimmer K, Kocher F, Spizzo G, Salem M, Gastl G, Seeber A (2019) Treatment according to molecular profiling in relapsed/refractory cancer patients: a review focusing on latest profiling studies. Comput Struct Biotechnol J. https://doi.org/10.1016/j.csbj.2019.03.012
Galluzzi L, Linkermann A, Kepp O, Kroemer G (2020) Pathophysiology of Cancer Cell Death. Abeloff's Clinical Oncology. Elsevier, Amsterdam, pp 74–83
Demarest TG, Babbar M, Okur MN, Dan X, Croteau DL, Fakouri NB, Mattson MP, Bohr VA (2019) NAD+ metabolism in aging and cancer. Ann Rev Cancer Biol 3:105–130
Johansson B, Mertens F, Schyman T, Björk J, Mandahl N, Mitelman F (2019) Most gene fusions in cancer are stochastic events. Genes Chromosom Cancer 58(9):607–611
Duan Y-T, Sangani CB, Liu W, Soni KV, Yao Y (2019) New promises to cure cancer and other genetic diseases/disorders: Epi-drugs through epigenetics. Curr Top Med Chem 19(12):972–994
Warburg O, Dickens F (1949) The metabolism of tumor, Constable, London 1930. In: Dickens F (ed) Trans
Gatenby RA, Gillies RJ (2004) Why do cancers have high aerobic glycolysis? Nat Rev Cancer 4(11):891–899
Tataranni T, Piccoli C (2019) Dichloroacetate (DCA) and cancer: an overview towards clinical applications. Oxidative Med Cell Longev. https://doi.org/10.1155/2019/8201079
Fantin VR, St-Pierre J, Leder P (2006) Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell 9(6):425–434
Sarosiek KA, Chonghaile TN, Letai A (2013) Mitochondria: gatekeepers of response to chemotherapy. Trends Cell Biol 23(12):612–619
Guerra F, Arbini AA, Moro L (2017) Mitochondria and cancer chemoresistance. Biochim Biophys Acta (BBA) Bioenerg 1858(8):686–699
Grasso D (2020) Common metabolic alterations in cancer chemoresistance and radioresistance. UCL-Université Catholique de Louvain, Ottignies-Louvain-la-Neuve
Petros JA, Baumann AK, Ruiz-Pesini E, Amin MB, Sun CQ, Hall J, Lim S, Issa MM, Flanders WD, Hosseini SH (2005) mtDNA mutations increase tumorigenicity in prostate cancer. Proc Natl Acad Sci 102(3):719–724
Kheirandish-Rostami M, Roudkenar MH, Jahanian-Najafabadi A, Tomita K, Kuwahara Y, Sato T, Roushandeh AM (2020) Mitochondrial characteristics contribute to proliferation and migration potency of MDA-MB-231 cancer cells and their response to cisplatin treatment. Life Sci 244:117339
Caicedo A, Fritz V, Brondello J-M, Ayala M, Dennemont I, Abdellaoui N, De Fraipont F, Moisan A, Prouteau CA, Boukhaddaoui H (2015) MitoCeption as a new tool to assess the effects of mesenchymal stem/stromal cell mitochondria on cancer cell metabolism and function. Sci Rep 5:9073
Mombo BN, Gerbal-Chaloin S, Bokus A, Daujat-Chavanieu M, Jorgensen C, Hugnot J-P, Vignais M-L (2017) MitoCeption: transferring isolated human MSC mitochondria to glioblastoma stem cells. JoVE (J Visual Exp) 120:e55245
Kaipparettu BA, Ma Y, Park JH, Lee T-L, Zhang Y, Yotnda P, Creighton CJ, Chan W-Y, Wong L-JC (2019) Correction: crosstalk from non-cancerous mitochondria can inhibit tumor properties of metastatic cells by suppressing oncogenic pathways. PLoS ONE 14(8):e0221671
Luo M, Wicha MS (2019) Targeting cancer stem cell redox metabolism to enhance therapy responses. Seminars in radiation oncology, vol 1. Elsevier, Amsterdam, pp 42–54
Vincent AE, Turnbull DM, Eisner V, Hajnóczky G, Picard M (2017) Mitochondrial nanotunnels. Trends Cell Biol 27(11):787–799
Nakamura Y, Park J-H, Hayakawa K (2019) Therapeutic use of extracellular mitochondria in CNS injury and disease. Exp Neurol 324:113114
Tomita K, Takashi Y, Ouchi Y, Kuwahara Y, Igarashi K, Nagasawa T, Nabika H, Kurimasa A, Fukumoto M, Nishitani Y (2019) Lipid peroxidation increases hydrogen peroxide permeability leading to cell death in cancer cell lines that lack mtDNA. Cancer Sci 110(9):2856
Kuwahara Y, Roudkenar MH, Urushihara Y, Saito Y, Tomita K, Roushandeh AM, Sato T, Kurimasa A, Fukumoto M (2017) Clinically relevant radioresistant cell line: a simple model to understand cancer radioresistance. Med Mol Morphol 50(4):195–204
Cosgrove D, Park BH, Vogelstein B (2016) Tumor suppressor genes. Holland-Frei Cancer Med. https://doi.org/10.1002/9781119000822.hfcm005
Kuwahara Y, Roudkenar MH, Suzuki M, Urushihara Y, Fukumoto M, Saito Y, Fukumoto M (2016) The involvement of mitochondrial membrane potential in cross-resistance between radiation and docetaxel. Int J Radiat Oncol* Biol* Phys 96(3):556–565
Lin H-Y, Liou C-W, Chen S-D, Hsu T-Y, Chuang J-H, Wang P-W, Huang S-T, Tiao M-M, Chen J-B, Lin T-K (2015) Mitochondrial transfer from Wharton's jelly-derived mesenchymal stem cells to mitochondria-defective cells recaptures impaired mitochondrial function. Mitochondrion 22:31–44
Spees JL, Olson SD, Whitney MJ, Prockop DJ (2006) Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci 103(5):1283–1288
Elliott R, Jiang X, Head J (2012) Mitochondria organelle transplantation: introduction of normal epithelial mitochondria into human cancer cells inhibits proliferation and increases drug sensitivity. Breast Cancer Res Treat 136(2):347–354
Chang J-C, Chang H-S, Wu Y-C, Cheng W-L, Lin T-T, Chang H-J, Kuo S-J, Chen S-T, Liu C-S (2019) Mitochondrial transplantation regulates antitumour activity, chemoresistance and mitochondrial dynamics in breast cancer. J Exp Clin Cancer Res 38(1):30
Dasari S, Tchounwou PB (2014) Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol 740:364–378
Acknowledgements
Part of this study was supported by Guilan University of Medical Sciences (Grant No: IR.GUMS.REC.1397.35) and JSPS KAKENHI (Grant-in Aid for Scientific Research C: No. 19K10318 to K.T).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study only involves in vitro experiments and there was no involvement of Human Participants and/or Animals in it.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Roushandeh, A.M., Tomita, K., Kuwahara, Y. et al. Transfer of healthy fibroblast-derived mitochondria to HeLa ρ0 and SAS ρ0 cells recovers the proliferation capabilities of these cancer cells under conventional culture medium, but increase their sensitivity to cisplatin-induced apoptotic death. Mol Biol Rep 47, 4401–4411 (2020). https://doi.org/10.1007/s11033-020-05493-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05493-5