Abstract
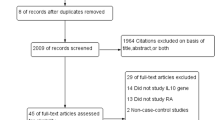
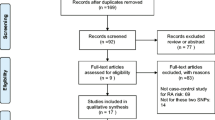
The aim of this study was to determine whether interleukin-23 receptor (IL-23R) polymorphisms confer susceptibility to rheumatoid arthritis (RA). A meta-analysis was conducted on the associations between the IL-23R rs1343151, rs10489629, rs7517847, rs11209026, rs1004819, and rs2201841 polymorphisms and RA using (1) allele contrast, (2) the recessive model, (3) the dominant model, and (4) the additive model. A total of 13 studies from eight articles involving 10,016 RA patients and 11,967 controls were considered in the meta-analysis. Meta-analysis identified a significant association between RA and the A allele of the rs1343151 polymorphism in the overall population (OR = 1.110, 95 % CI = 1.056–1.168, p = 4.7 × 10−6). Stratification by ethnicity identified a significant association between this polymorphism and RA in Europeans (OR = 1.105, 95 % CI = 1.049–1.163, p = 1.4 × 10−5). An association was also found between RA and the A allele carrier of the rs1343151 polymorphism in Europeans (OR = 1.135, 95 % CI = 1.058–1.217, p = 4.0 × 10−5). Meta-analysis revealed a significant association between RA and the A allele of the rs10489629 polymorphism in the overall population (OR = 1.079, 95 % CI = 1.029–1.131, p = 0.002) and in Europeans (OR = 1.092, 95 % CI = 1.038–1.149, p = 0.001). Meta-analyses of recessive, dominant, and additive models showed the same pattern as the meta-analysis of the A allele of the rs10489629 polymorphism, that is, a significant association with RA in Europeans. However, no association was found between the IL-23R rs7517847, rs11209026, rs1004819, and rs2201841 polymorphisms and RA susceptibility. This meta-analysis shows that the IL-23R rs1343151 and rs10489629 polymorphisms are associated with the development of RA in Europeans. These findings suggest that the IL-23R genes confer susceptibility to RA in the European population, but further study of this association is required in other ethnic groups.


Similar content being viewed by others
References
Harris ED Jr (1990) Rheumatoid arthritis. Pathophysiology and implications for therapy. N Engl J Med 322:1277–1289
Kim HR, Park MK, Cho ML, Kim KW, Oh HJ, Park JS, Heo YM, Lee SH, Kim HY, Park SH (2010) Induction of macrophage migration inhibitory factor in ConA-stimulated rheumatoid arthritis synovial fibroblasts through the P38 map kinase-dependent signaling pathway. Korean J Intern Med 25:317–326
Deighton CM, Walker DJ, Griffiths ID, Roberts DF (1989) The contribution of HLA to rheumatoid arthritis. Clin Genet 36:178–182
Choi SJ, Rho YH, Ji JD, Song GG, Lee YH (2006) Genome scan meta-analysis of rheumatoid arthritis. Rheumatology (Oxford) 45:166–170
Brown MA (2008) Breakthroughs in genetic studies of ankylosing spondylitis. Rheumatology (Oxford) 47:132–137
Murphy CA, Langrish CL, Chen Y, Blumenschein W, McClanahan T, Kastelein RA, Sedgwick JD, Cua DJ (2003) Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J Exp Med 198:1951–1957
Steinman L (2007) A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat Med 13:139–145
Kim HR, Cho ML, Kim KW, Juhn JY, Hwang SY, Yoon CH, Park SH, Lee SH, Kim HY (2007) Up-regulation of IL-23p19 expression in rheumatoid arthritis synovial fibroblasts by IL-17 through PI3-kinase-, NF-kappaB- and p38 MAPK-dependent signalling pathways. Rheumatology (Oxford) 46:57–64
Cargill M, Schrodi SJ, Chang M, Garcia VE, Brandon R, Callis KP, Matsunami N, Ardlie KG, Civello D, Catanese JJ, Leong DU, Panko JM, McAllister LB, Hansen CB, Papenfuss J, Prescott SM, White TJ, Leppert MF, Krueger GG, Begovich AB (2007) A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am J Hum Genet 80:273–290
Safrany E, Melegh B (2009) Functional variants of the interleukin-23 receptor gene in non-gastrointestinal autoimmune diseases. Curr Med Chem 16:3766–3774
Chen-Xu M, Topless R, McKinney C, Merriman ME, Phipps-Green A, Dalbeth N, Gow PJ, Harrison AA, Highton J, Jones PB, Nissen M, Smith MD, van Rij A, Jones GT, Rodriguez-Rodriguez L, Fernandez-Gutierrez B, Teruel M, Balsa A, Pascual-Salcedo D, Ortiz AM, Gonzalez-Gay MA, Steer S, Maehlen M, Lie B, Wordsworth BP, Stamp LK, Martin J, Merriman TR (2012) Replication of association of the interleukin-23 receptor rs1343151 variant with rheumatoid arthritis in Caucasian sample sets. Ann Rheum Dis 71:155–157
Varade J, Ramon Lamas J, Rodriguez L, Fernandez-Arquero M, Loza-Santamaria E, Jover JA, de la Concha EG, Fernandez-Gutierrez B, Urcelay E, Martinez A (2009) IL23R and IL12B genes: susceptibility analysis in rheumatoid arthritis. Ann Rheum Dis 68:1230–1232
Park JH, Kim YJ, Park BL, Bae JS, Shin HD, Bae SC (2009) Lack of association between interleukin-23 receptor gene polymorphisms and rheumatoid arthritis susceptibility. Rheumatol Int 29:781–786
Hollis-Moffatt JE, Merriman ME, Rodger RA, Rowley KA, Chapman PT, Dalbeth N, Gow PJ, Harrison AA, Highton J, Jones PB, O’Donnell JL, Stamp LK, Merriman TR (2009) Evidence for association of an interleukin-23 receptor variant independent of the R381Q variant with rheumatoid arthritis. Ann Rheum Dis 68:1340–1344
Chang M, Saiki RK, Cantanese JJ, Lew D, van der Helm-van Mil AH, Toes RE, Huizinga TW, Ardlie KG, Criswell LA, Seldin MF, Amos CI, Kastner DL, Gregersen PK, Schrodi SJ, Begovich AB (2008) The inflammatory disease-associated variants in IL12B and IL23R are not associated with rheumatoid arthritis. Arthritis Rheum 58:1877–1881
Farago B, Magyari L, Safrany E, Csongei V, Jaromi L, Horvatovich K, Sipeky C, Maasz A, Radics J, Gyetvai A, Szekanecz Z, Czirjak L, Melegh B (2008) Functional variants of interleukin-23 receptor gene confer risk for rheumatoid arthritis but not for systemic sclerosis. Ann Rheum Dis 67:248–250
Orozco G, Rueda B, Robledo G, Garcia A, Martin J (2007) Investigation of the IL23R gene in a Spanish rheumatoid arthritis cohort. Hum Immunol 68:681–684
Wellcome Trust Case Control Consortium (2007) Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447:661–678
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. Br Med J 315:629–634
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Egger M, Smith GD, Phillips AN (1997) Meta-analysis: principles and procedures. Br Med J 315:1533–1537
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Hazlett J, Stamp LK, Merriman T, Highton J, Hessian PA (2012) IL-23R rs11209026 polymorphism modulates IL-17A expression in patients with rheumatoid arthritis. Genes Immun 13:282–287
Paradowska-Gorycka A, Grzybowska-Kowalczyk A, Wojtecka-Lukasik E, Maslinski S (2010) IL-23 in the pathogenesis of rheumatoid arthritis. Scand J Immunol 71:134–145
Safrany E, Hobor R, Jakab L, Tarr T, Csongei V, Jaromi L, Sipeky C, Valasek A, Zeher M, Fust G, Czirjak L, Melegh B (2010) Interleukin-23 receptor gene variants in Hungarian systemic lupus erythematosus patients. Inflamm Res 59:159–164
Lee HS, Park H, Yang S, Kim D, Park Y (2008) STAT4 polymorphism is associated with early-onset type 1 diabetes, but not with late-onset type 1 diabetes. Ann N Y Acad Sci 1150:93–98
Ban Y, Tozaki T, Taniyama M, Nakano Y, Yoneyama K, Hirano T (2009) Association studies of the IL-23R gene in autoimmune thyroid disease in the Japanese population. Autoimmunity 42:126–130
Martinez A, Varade J, Marquez A, Cenit MC, Espino L, Perdigones N, Santiago JL, Fernandez-Arquero M, de la Calle H, Arroyo R, Mendoza JL, Fernandez-Gutierrez B, de la Concha EG, Urcelay E (2008) Association of the STAT4 gene with increased susceptibility for some immune-mediated diseases. Arthritis Rheum 58:2598–2602
Korman BD, Kastner DL, Gregersen PK, Remmers EF (2008) STAT4: genetics, mechanisms, and implications for autoimmunity. Curr Allergy Asthma Rep 8:398–403
Asarch A, Barak O, Loo DS, Gottlieb AB (2008) Th17 cells: a new therapeutic target in inflammatory dermatoses. J Dermatolog Treat 19:318–326
Huber AK, Jacobson EM, Jazdzewski K, Concepcion ES, Tomer Y (2008) Interleukin (IL)-23 receptor is a major susceptibility gene for Graves’ ophthalmopathy: the IL-23/T-helper 17 axis extends to thyroid autoimmunity. J Clin Endocrinol Metab 93:1077–1081
Iwakura Y, Ishigame H (2006) The IL-23/IL-17 axis in inflammation. J Clin Invest 116:1218–1222
Burton PR, Clayton DG, Cardon LR, Craddock N, Deloukas P, Duncanson A, Kwiatkowski DP, McCarthy MI, Ouwehand WH, Samani NJ, Todd JA, Donnelly P, Barrett JC, Davison D, Easton D, Evans DM, Leung HT, Marchini JL, Morris AP, Spencer CC, Tobin MD, Attwood AP, Boorman JP, Cant B, Everson U, Hussey JM, Jolley JD, Knight AS, Koch K, Meech E, Nutland S, Prowse CV, Stevens HE, Taylor NC, Walters GR, Walker NM, Watkins NA, Winzer T, Jones RW, McArdle WL, Ring SM, Strachan DP, Pembrey M, Breen G, St Clair D, Caesar S, Gordon-Smith K, Jones L, Fraser C, Green EK, Grozeva D, Hamshere ML, Holmans PA, Jones IR, Kirov G, Moskivina V, Nikolov I, O’Donovan MC, Owen MJ, Collier DA, Elkin A, Farmer A, Williamson R, McGuffin P, Young AH, Ferrier IN, Ball SG, Balmforth AJ, Barrett JH, Bishop TD, Iles MM, Maqbool A, Yuldasheva N, Hall AS, Braund PS, Dixon RJ, Mangino M, Stevens S, Thompson JR, Bredin F, Tremelling M, Parkes M, Drummond H, Lees CW, Nimmo ER, Satsangi J, Fisher SA, Forbes A, Lewis CM, Onnie CM, Prescott NJ, Sanderson J, Matthew CG, Barbour J, Mohiuddin MK, Todhunter CE, Mansfield JC, Ahmad T, Cummings FR, Jewell DP, Webster J, Brown MJ, Lathrop MG, Connell J, Dominiczak A, Marcano CA, Burke B, Dobson R, Gungadoo J, Lee KL, Munroe PB, Newhouse SJ, Onipinla A, Wallace C, Xue M, Caulfield M, Farrall M, Barton A, Bruce IN, Donovan H, Eyre S, Gilbert PD, Hilder SL, Hinks AM, John SL, Potter C, Silman AJ, Symmons DP, Thomson W, Worthington J, Dunger DB, Widmer B, Frayling TM, Freathy RM, Lango H, Perry JR, Shields BM, Weedon MN, Hattersley AT, Hitman GA, Walker M, Elliott KS, Groves CJ, Lindgren CM, Rayner NW, Timpson NJ, Zeggini E, Newport M, Sirugo G, Lyons E, Vannberg F, Hill AV, Bradbury LA, Farrar C, Pointon JJ, Wordsworth P, Brown MA, Franklyn JA, Heward JM, Simmonds MJ, Gough SC, Seal S, Stratton MR, Rahman N, Ban M, Goris A, Sawcer SJ, Compston A, Conway D, Jallow M, Rockett KA, Bumpstead SJ, Chaney A, Downes K, Ghori MJ, Gwilliam R, Hunt SE, Inouye M, Keniry A, King E, McGinnis R, Potter S, Ravindrarajah R, Whittaker P, Widden C, Withers D, Cardin NJ, Ferreira T, Pereira-Gale J, Hallgrimsdo’ttir IB, Howie BN, Su Z, Teo YY, Vukcevic D, Bentley D, Mitchell SL, Newby PR, Brand OJ, Carr-Smith J, Pearce SH, Reveille JD, Zhou X, Sims AM, Dowling A, Taylor J, Doan T, Davis JC, Savage L, Ward MM, Learch TL, Weisman MH, Brown M (2007) Association scan of 14,500 nonsynonymous SNPs in four diseases identifies autoimmunity variants. Nat Genet 39:1329–1337
Rueda B, Orozco G, Raya E, Fernandez-Sueiro JL, Mulero J, Blanco FJ, Vilches C, Gonzalez-Gay MA, Martin J (2008) The IL23R Arg381Gln non-synonymous polymorphism confers susceptibility to ankylosing spondylitis. Ann Rheum Dis 67:1451–1454
Pearce SH, Merriman TR (2006) Genetic progress towards the molecular basis of autoimmunity. Trends Mol Med 12:90–98
Duerr RH, Taylor KD, Brant SR, Rioux JD, Silverberg MS, Daly MJ, Steinhart AH, Abraham C, Regueiro M, Griffiths A, Dassopoulos T, Bitton A, Yang H, Targan S, Datta LW, Kistner EO, Schumm LP, Lee AT, Gregersen PK, Barmada MM, Rotter JI, Nicolae DL, Cho JH (2006) A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 314:1461–1463
Zhang XY, Zhang HJ, Zhang Y, Fu YJ, He J, Zhu LP, Wang SH, Liu L (2006) Identification and expression analysis of alternatively spliced isoforms of human interleukin-23 receptor gene in normal lymphoid cells and selected tumor cells. Immunogenetics 57:934–943
Safrany E, Pazar B, Csongei V, Jaromi L, Polgar N, Sipeky C, Horvath IF, Zeher M, Poor G, Melegh B (2009) Variants of the IL23R gene are associated with ankylosing spondylitis but not with Sjogren syndrome in Hungarian population samples. Scand J Immunol 70:68–74
Zwiers A, Kraal L, van de Pouw Kraan TC, Wurdinger T, Bouma G, Kraal G (2012) Cutting edge: a variant of the IL-23R gene associated with inflammatory bowel disease induces loss of microRNA regulation and enhanced protein production. J Immunol 188:1573–1577
Bettelli E, Oukka M, Kuchroo VK (2007) T(H)-17 cells in the circle of immunity and autoimmunity. Nat Immunol 8:345–350
Acknowledgments
This study was supported by the Korean Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (Grant No. A102065).
Conflict of interest
The authors have no financial or non-financial conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, G.G., Bae, SC., Choi, S.J. et al. Associations between interleukin-23 receptor polymorphisms and susceptibility to rheumatoid arthritis: a meta-analysis. Mol Biol Rep 39, 10655–10663 (2012). https://doi.org/10.1007/s11033-012-1955-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-012-1955-7