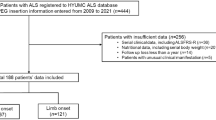
Abstract
Dysphagia and progressive swallowing problems due to motoneuron death is one of amyotrophic lateral sclerosis (ALS) symptoms. Malnutrition and body weight loss result in immunological disturbances, fatigability and increase risk of secondary complications in ALS patients, percutaneous endoscopic gastrostomy tube (PEG) placement representing a well-recognized method for malnutrition correction and potentially increasing life expectancy. However, despite nutritional correction, occasional rapid neurological deterioration may develop after PEG placement. We have hypothesized that this decline can be a result of exteroceptive stress during PEG placement and promote neurodegeneration in ALS patients. Intravenous sedation may decrease stress during invasive procedures and it is safe during PEG placement in ALS patients. The aim of the study was comparing different PEG placement protocols of anesthesia (local anesthesia or local anesthesia plus intravenous sedation) in ALS from perspectives of stress load and neurological deterioration profile. During 1.5 years 94 ALS patients were admitted; gastrostomy was performed in 79 patients. After screening according to inclusion and exclusion criteria, 30 patients were included in the prospective consecutive study. All patients were divided in two groups, with local anesthesia and with combination of local anesthesia and intravenous sedation. Routine biochemical indices, neurodegeneration and stress markers were measured. The age of ALS patients was 61 ± 10 years; 20 patients were included at stage 4A and 10 at stage 4B (King’s College staging). PEG was placed at average14 months after the diagnosis and 2.2 years after first symptoms. Mean ALS Functional Rating Scale-Revised was 27.8, mean forced vital capacity of lung 46.3% (19—91%). After one year of observation only 8 patients survived. Mean life duration after PEG was 5 months (5 days—20 months). Comparison of two PEG placement protocols did not reveal differences in survival time, stress load and inflammation level. Higher saliva cortisol levels, serum cortisol, glucose, C-reactive protein and interleukin-6 were detected after PEG placement, confirming considerable stress response. PEG is a stressful factor for ALS patients, PEG placement representing a natural model of exteroceptive stress. Stress response was detected as increased cortisol, C-reactive protein, interleukin-6, and glucose levels. Intravenous sedation did not increase the risk of PEG placement procedure, however, sedation protocol did not affect stress load.








Similar content being viewed by others
Availability of data and material
Due to the nature of this research, participants of this study did not agree for their data to be shared publicly, so supporting data is not available.
Code availability
Not applicable.
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis;
- ALSFRS-R:
-
ALS Functional Rating Scale-Revised;
- BDNF:
-
Brain-derived neurotrophic factor;
- BMI:
-
Body mass index;
- CRP:
-
C-reactive protein;
- ECAS:
-
Edinburgh Cognitive and Behavioral ALS Screen;
- FVC:
-
Forced vital capacity;
- Il-1β:
-
Interleukin-1β;
- Il-6:
-
Interleukin-6;
- PEG:
-
Percutaneous endoscopic gastrostomy tube;
- pNfh:
-
Phosphorylated neurofilament heavy chains
References
Abrahams S, Newton J, Niven E, Foley J, Bak T (2014) Screening for cognition and behaviour changes in ALS. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration15(1–2):9–14 https://doi.org/10.3109/21678421.2013.805784
Aguinis H, Gottfredson R, Joo H (2013) Best-Practice Recommendations for Defining, Identifying, and Handling Outliers. Organ Res Methods 16(2):270–301. https://doi.org/10.1177/1094428112470848
Andersen PM et al (2012) EFNS Task Force on Diagnosis and Management of Amyotrophic Lateral Sclerosis: et al. EFNS guidelines on the clinical management of amyotrophic lateral sclerosis (MALS)–revised report of an EFNS task force. European journal of neurology 19: 360–375. https://doi.org/10.1111/j.1468-1331.2005.01351.x
Atassi N, Cudkowicz M, Schoenfeld D (2011) Advanced Statistical Methods to Study the Effects of Gastric Tube and Non-Invasive Ventilation on Functional Decline and Survival in Amyotrophic Lateral Sclerosis 12(4):272–277. https://doi.org/10.3109/17482968.2011.577786
Aust H, Rüsch D, Schuster M, Sturm T, Brehm F, Nestoriuc Y (2016) Coping strategies in anxious surgical patients. BMC Health Serv Res 16:250. https://doi.org/10.1186/s12913-016-1492-5
Ben-Eliyahu S (2020) Tumor Excision as a Metastatic Russian Roulette: Perioperative Interventions to Improve Long-Term Survival of Cancer Patients. Trends Cancer9:S2405–8033(20)30188–6. https://doi.org/10.1016/j.trecan.2020.06.004.
Blasco H, Veyrat-Durebex C, Bocca C et al (2017) Lipidomics Reveals Cerebrospinal-Fluid Signatures of ALS. Sci Rep 7(1):17652. https://doi.org/10.1038/s41598-017-17389-9
Bond L, Ganguly P, Khamankar N et al (2019) A Comprehensive Examination of Percutaneous Endoscopic Gastrostomy and Its Association with Amyotrophic Lateral Sclerosis Patient Outcomes. Brain Sci 9(9):223. https://doi.org/10.3390/brainsci9090223
Brooks B, Miller R, Swash M, Munsat T and World Federation of Neurology Research Group on Motor Neuron Diseases (2000) El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotrophic lateral sclerosis and other motor neuron disorders 1(5): 293–299.https://doi.org/10.1080/146608200300079536
Chełstowska B, Kuźma-Kozakiewicz M (2020) Biochemical parameters in determination of nutritional status in amyotrophic lateral sclerosis. Neurol Sci 41(5):1115–1124. https://doi.org/10.1007/s10072-019-04201-x
Chernenkaya V, Gorbachev K, Gorbachev D, Ataulina A, Fominykh V, Brylev L, Guekht A (2018) The Edinburgh Cognitive and Behavioral ALS Screen (ECAS): a Russian version. Zh Nevrol Psikhiatr Im S S Korsakova 118(12):36–39. https://doi.org/10.17116/jnevro201811812136
Chiò A, Calvo A, Bovio G et al (2014) Piemonte and Valle d’Aosta Register for Amyotrophic Lateral Sclerosis. Amyotrophic lateral sclerosis outcome measures and the role of albumin and creatinine: a population-based study. JAMA Neurol 71(9):1134–42. https://doi.org/10.1001/jamaneurol.2014.1129.
Choi S, Hong Y, Shin J et al (2017) Takotsubo cardiomyopathy in amyotrophic lateral sclerosis. J Neurol Sci 375:289–293. https://doi.org/10.1016/j.jns.2017.02.012
Crockford C, Newton J, Lonergan K et al (2018) ALS-specific cognitive and behavior changes associated with advancing disease stage in ALS. Neurology 91(15):e1370–e1380. https://doi.org/10.1212/WNL.0000000000006317
Finsterer J, Wahbi K (2014) CNS disease triggering Takotsubo stress cardiomyopathy. Int J Cardiol 177(2):322–329. https://doi.org/10.1016/j.ijcard.2014.08.101
Fominykh V, Vorobyeva A, Onufriev MV, Brylev L, Zakharova MN, Gulyaeva NV (2018) Interleukin-6, S-Nitrosothiols, and Neurodegeneration in Different CentralNervous System Demyelinating Disorders: Is There a Relationship? J Clin Neurol 14(3):327–332. https://doi.org/10.3988/jcn.2018.14.3.327
Gulyaeva N (2019) Biochemical Mechanisms and Translational Relevance of Hippocampal Vulnerability to Distant Focal Brain Injury: The Price of Stress Response. Biochemistry (mosc) 84(11):1306–1328. https://doi.org/10.1134/S0006297919110087
Heffernan C et al (2004) Nutritional management in MND/ALS patients: an evidence based review. Amyotroph Lateral Scler Other Motor Neuron Disord 5(2):72–83. https://doi.org/10.1080/14660820410020349
Hill CM, Walker RV (2001) Salivary cortisol determinations and self-rating scales in the assessment of stress in patients undergoing the extraction of wisdom teeth. Br Dent J 191(9):513–515. https://doi.org/10.1038/sj.bdj.4801220
Hodgins F, Mulhern S, Abrahams S (2020) The clinical impact of the Edinburgh Cognitive and Behavioural ALS Screen (ECAS) and neuropsychological intervention in routine ALS care. Amyotrophic Lateral Sclerosis & Frontotemporal Degeneration 21(1–2):92–99. https://doi.org/10.1080/21678421.2019.1674874
Hoeper A, Barbara D, Watson J et al (2019) Amyotrophic lateral sclerosis and anesthesia: a case series and review of the literature. J Anesth 33(2):257–265. https://doi.org/10.1007/s00540-019-02611-x
Kalava A, Clendenen S, McKinney J, Bojaxhi E, Greengrass R (2016) Bilateral thoracic paravertebral nerve blocks for placement of percutaneous radiologic gastrostomy in patients with amyotrophic lateral sclerosis: a case series. Romanian journal of anaesthesia and intensive care 23(2):149–153. https://doi.org/10.21454/rjaic.7518/232.scl
Larauche M, Mulak A, Taché Y (2012) Stress and visceral pain: from animal models to clinical therapies. Exp Neurol 233(1):49–67. https://doi.org/10.1016/j.expneurol.2011.04.020
Lu C, Petzold A, Topping J et al (2015) Plasma neurofilament heavy chain levels and disease progression in amyotrophic lateral sclerosis: insights from a longitudinal study. J Neurol Neurosurg Psychiatry 86(5):565–573. https://doi.org/10.1136/jnnp-2014-307672
Manou-Stathopoulou V, Korbonits M, Ackland GL (2019) Redefining the perioperative stress response: a narrative review. Br J Anaesth 123(5):570–583. https://doi.org/10.1016/j.bja.2019.08.011
Prabhakar A, Owen CP, Kaye AD (2013) Anesthetic management of the patient with amyotrophic lateral sclerosis. J Anesth 27(6):909–918. https://doi.org/10.1007/s00540-013-1644-2
ProGas Study Group (2015) Gastrostomy in patients with amyotrophic lateral sclerosis (ProGas): a prospective cohort study. The Lancet Neurology 14(7):702–709. https://doi.org/10.1016/S1474-4422(15)00104-0
Steinthorsdottir K, Kehlet H, Aasvang E (2017) Surgical stress response and the potential role of preoperative glucocorticoids on post-anesthesia care unit recovery. Minerva anestesiologica 83(12):1324–1331. https://doi.org/10.23736/S0375-9393.17.11878-X
Strijbos D, Hofstede J, Keszthelyi D, Masclee A, Gilissen L (2017) Percutaneous endoscopic gastrostomy under conscious sedation in patients with amyotrophic lateral sclerosis is safe: an observational study. Eur J Gastroenterol Hepatol 29(11):1303–1308. https://doi.org/10.1097/MEG.0000000000000959
Toone R, Peacock O, Smith A, Thompson D, Drawer S, Cook C, Stokes KA (2013) Measurement of steroid hormones in saliva: Effects of sample storage condition. Scand J Clin Lab Invest 73(8):615–621. https://doi.org/10.3109/00365513.2013.835862
Vergonjeanne M, Fayemendy P, Marin B et al (2020) Predictive factors for gastrostomy at time of diagnosis and impact on survival in patients with amyotrophic lateral sclerosis. Clin Nutr 39(10):3112–3118. https://doi.org/10.1016/j.clnu.2020.01.018
Funding
This study was performed with financial and organizational support by "Live Now" Charity Foundation for supporting people with ALS and other neuromuscular disorders (Moscow, Russia, https://alsfund.ru) and RFBR grant № 18–315-00228 mol_a.
Author information
Authors and Affiliations
Contributions
Brylev L. – design and idea of the study, interpretation of data, manuscript preparation.
Fominykh V. – design the study, patient management, data analysis, interpretation of data, manuscript preparation and submission.
Chernenkaia V. – patients treatment and management, draft preparation, design of figures.
Chernenkiy I., Yakovlev A. – statistical support and analysis.
Gorbachev K., Ataulina A. Parshikov V., Demeshonok V. – patients management and technical works, biological fluids management and analysis.
Izvekov A., Monakhov M, Olenichev A, Orlov S, Turin I. – surgical procedures, PEG placement, data management.
Loginov M., Rautbart S. – anesthesiological management of patients, data management.
Baymukanov A. – analysis of cardiac function, design of cardiac evaluation.
Druzhkova T. – analysis of biomarkers, laboratory management.
Guekht A. – supervising, critical revising of intellectual content.
Gulyaeva N. – experimental design, supervising, critical revising of intellectual content.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethics approval
This study was approved by the local Ethical Committee of Bujanov Moscow City Clinical Hospital. This study was performed in line with the principles of the Declaration of Helsinki.
Consent to participate, consent for publication
Informed consent was obtained from all individual participants included in the study.
All patients signed informed concern for data publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• PEG placement is a stress load for ALS patients
• Stress response can be detected by augmented cortisol, CRP and Il-6 levels
• After PEG placement, markers of stress response return to baseline
• Stress load does not depend on type of anesthesia
• Intravenous sedation does not increase the risk of PEG placement
• Normalization of total cholesterol after PEG confirms stabilization of nutritional status
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Brylev, L., Fominykh, V., Chernenkaia, V. et al. Stress load and neurodegeneration after gastrostomy tube placement in amyotrophic lateral sclerosis patients. Metab Brain Dis 36, 2473–2482 (2021). https://doi.org/10.1007/s11011-021-00837-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00837-x