Abstract
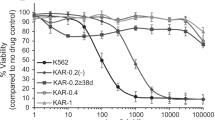
The multidrug-resistant (MDR) phenotype is multifactorial, and cell lines presenting multiple resistance mechanisms might be good models to understand the importance of the various pathways involved. The present work characterized a MDR chronic myeloid leukemia cell line, derived from K562 through a selective process using daunorubicin. This MDR cell line was shown to be resistant to vincristine, daunorubicin, and partially resistant to imatinib. It showed a slower duplication rate. Overexpression of ABCB1 and ABCC1 was observed at the protein and functional levels and the expression of CD95, a molecule related to cell death, was reduced in the MDR cell line. Conversely, no differences were observed related to the anti-apoptotic molecule Bcl-2 or p53 expression. The activation antigen CD69 was reduced in the MDR cell line and treatment with imatinib further decreased the expressed levels. Furthermore, secretion of IL-8 was diminished in the MDR cell line. When daunorubicin-selected cells were compared to another MDR cell line, Lucena 1, derived from the same parental line K562, and selected with vincristine, a different profile was observed in relation to most aspects studied. When both cell lines were silenced for ABCB1, differences in CD69 and CD95 were maintained, despite resistance reversal. These results reinforce the idea that cell lines selected in vitro may display multiple resistance strategies that may vary with the selective agent used as well as during different steps of the selection process.












Similar content being viewed by others
Abbreviations
- CML:
-
Chronic myeloid leukemia
- ABC transporters:
-
ATP binding cassette transporters
- P gp:
-
P glycoprotein
- BCRP:
-
Breast cancer resistant protein
- MDR:
-
Multidrug resistance
- MRP1:
-
Multidrug resistance related protein 1
- IM:
-
Imatinibe mesylate
- FCS:
-
Fetal calf serum
- VCR:
-
Vincristine sulfate
- DNR:
-
Daunorubicin hydrochloride
- CDDP:
-
Cisplatin
- VP:
-
Verapamil
- DMSO:
-
Dimethyl sulfoxide
- CFDA:
-
Carboxy fluorescein diacetate
- Rho:
-
Rhodamine 123
- INDO:
-
Indomethacin
- PRB:
-
Probenicid
- FACS:
-
Fluorescence cell sorter
- PBS:
-
Phosphate buffered saline
- HBSS:
-
Hank’s balanced salt solution
References
Deininger MW, Goldman JM, Melo JV (2000) The molecular biology of chronic myeloid leukemia. Blood 96(10):3343–3356
Deininger M, Buchdunger E, Druker BJ (2005) The development of imatinib as a therapeutic agent for chronic myeloid leukemia. Blood 105(7):2640–2653. doi:10.1182/blood-2004-08-3097
Hamada A, Miyano H, Watanabe H, Saito H (2003) Interaction of imatinib mesilate with human P-glycoprotein. J Pharmacol Exp Ther 307(2):824–828. doi:10.1124/jpet.103.055574
Mahon FX, Belloc F, Lagarde V, Chollet C, Moreau-Gaudry F, Reiffers J, Goldman JM, Melo JV (2003) MDR1 gene overexpression confers resistance to imatinib mesylate in leukemia cell line models. Blood 101(6):2368–2373. doi:10.1182/blood.V101.6.2368
Widmer N, Colombo S, Buclin T, Decosterd LA (2003) Functional consequence of MDR1 expression on imatinib intracellular concentrations. Blood 102(3):1142. doi:10.1182/blood-2003-03-0993
Burger H, van Tol H, Boersma AW, Brok M, Wiemer EA, Stoter G, Nooter K (2004) Imatinib mesylate (STI571) is a substrate for the breast cancer resistance protein (BCRP)/ABCG2 drug pump. Blood 104(9):2940–2942. doi:10.1182/blood-2004-04-1398
Burger H, van Tol H, Brok M, Wiemer EA, de Bruijn EA, Guetens G, de Boeck G, Sparreboom A, Verweij J, Nooter K (2005) Chronic imatinib mesylate exposure leads to reduced intracellular drug accumulation by induction of the ABCG2 (BCRP) and ABCB1 (MDR1) drug transport pumps. Cancer Biol Ther 4(7):747–752
Eechoute K, Sparreboom A, Burger H, Franke RM, Schiavon G, Verweij J, Loos WJ, Wiemer EA, Mathijssen RH (2011) Drug transporters and imatinib treatment: implications for clinical practice. Clin Cancer Res 17(3):406–415. doi:10.1158/1078-0432.CCR-10-2250
Juliano RL, Ling V (1976) A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim Biophys Acta 455(1):152–162
Cole SP, Bhardwaj G, Gerlach JH, Mackie JE, Grant CE, Almquist KC, Stewart AJ, Kurz EU, Duncan AM, Deeley RG (1992) Overexpression of a transporter gene in a multidrug-resistant human lung cancer cell line. Science 258(5088):1650–1654. doi:10.1126/science.1360704
Doyle LA, Yang W, Abruzzo LV, Krogmann T, Gao Y, Rishi AK, Ross DD (1998) A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc Natl Acad Sci USA 95(26):15665–15670. doi:10.1073/pnas.95.26.15665
Gottesman MM, Fojo T, Bates SE (2002) Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer 2(1):48–58. doi:10.1038/nrc706
Vaidya S, Ghosh K, Vundinti BR (2011) Recent developments in drug resistance mechanism in chronic myeloid leukemia: a review. Eur J Haematol 87(5):381–393. doi:10.1111/j.1600-0609.2011.01689.x
Mahon FX, Deininger MW, Schultheis B, Chabrol J, Reiffers J, Goldman JM, Melo JV (2000) Selection and characterization of BCR-ABL positive cell lines with differential sensitivity to the tyrosine kinase inhibitor STI571: diverse mechanisms of resistance. Blood 96(3):1070–1079
Rumjanek VM, Trindade GS, Wagner-Souza K, de-Oliveira MC, Marques-Santos LF, Maia RC, Capella MA (2001) Multidrug resistance in tumour cells: characterization of the multidrug resistant cell line K562-Lucena 1. An Acad Bras Cienc 73(1):57–69
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63
Hall MD, Okabe M, Shen DW, Liang XJ, Gottesman MM (2008) The role of cellular accumulation in determining sensitivity to platinum-based chemotherapy. Annu Rev Pharmacol Toxicol 48:495–535. doi:10.1146/annurev.pharmtox
Eiring AM, Khorashad JS, Morley K, Deininger MW (2011) Advances in the treatment of chronic myeloid leukemia. BMC Med 9:99. doi:10.1186/1741-7015-9-99
Assef Y, Rubio F, Colo G, del Monaco S, Costas MA, Kotsias BA (2009) Imatinib resistance in multidrug-resistant K562 human leukemic cells. Leuk Res 33(5):710–716. doi:10.1016/j.leukres.2008.09.024
Kotaki M, Motoji T, Takanashi M, Wang YH, Mizoguchi H (2003) Anti-proliferative effect of the abl tyrosine kinase inhibitor STI571 on the P-glycoprotein positive K562/ADM cell line. Cancer Lett 199(1):61–68
Rumpold H, Wolf AM, Gruenewald K, Gastl G, Gunsilius E, Wolf D (2005) RNAi-mediated knockdown of P-glycoprotein using a transposon-based vector system durably restores imatinib sensitivity in imatinib-resistant CML cell lines. Exp Hematol 33(7):767–775. doi:10.1016/j.exphem.2005.03.014
Hall MD, Handley MD, Gottesman MM (2009) Is resistance useless? Multidrug resistance and collateral sensitivity. Trends Pharmacol Sci 30(10):546–556. doi:10.1016/j.tips.2009.07.003
Trompier D, Chang XB, Barattin R, du Moulinet D’Hardemare A, Di Pietro A, Baubichon-Cortay H (2004) Verapamil and its derivative trigger apoptosis through glutathione extrusion by multidrug resistance protein MRP1. Cancer Res 64(14):4950–4956. doi:10.1158/0008-5472.CAN-04-0143
Warr JR, Brewer F, Anderson M, Fergusson J (1986) Verapamil hypersensitivity of vincristine resistant Chinese hamster ovary cell lines. Cell Biol Int Rep 10(5):389–399
Cavalcanti GB Jr, Vasconcelos FC, Pinto de Faria G, Scheiner MA, de Almeida Dobbin J, Klumb CE, Maia RC (2004) Coexpression of p53 protein and MDR functional phenotype in leukemias: the predominant association in chronic myeloid leukemia. Cytometry B Clin Cytom 61(1):1–8. doi:10.1002/cyto.b.20013
Waring P, Mullbacher A (1999) Cell death induced by the Fas/Fas ligand pathway and its role in pathology. Immunol Cell Biol 77(4):312–317. doi:10.1046/j.1440-1711.1999.00837.x
Smeets ME, Raymakers RA, Vierwinden G, Pennings AH, Wessels H, de Witte T (1999) Triggering noncycling hematopoietic progenitors and leukemic blasts to proliferate increases anthracycline retention and toxicity by downregulating multidrug resistance. Blood 94(7):2414–2423
Marzio R, Mauel J, Betz-Corradin S (1999) CD69 and regulation of the immune function. Immunopharmacol Immunotoxicol 21(3):565–582. doi:10.3109/08923979909007126
Hantschel O, Gstoettenbauer A, Colinge J, Kaupe I, Bilban M, Burkard TR, Valent P, Superti-Furga G (2008) The chemokine interleukin-8 and the surface activation protein CD69 are markers for Bcr-Abl activity in chronic myeloid leukemia. Mol Oncol 2(3):272–281. doi:10.1016/j.molonc.2008.07.003
Vasconcelos FC, Cavalcanti GB Jr, Silva KL, de Meis E, Kwee JK, Rumjanek VM, Maia RC (2007) Contrasting features of MDR phenotype in leukemias by using two fluorochromes: implications for clinical practice. Leuk Res 31(4):445–454. doi:10.1016/j.leukres.2006.07.016
Vasconcelos FC, Gattass CR, Rumjanek VM, Maia RC (2007) Pomolic acid-induced apoptosis in cells from patients with chronic myeloid leukemia exhibiting different drug resistance profile. Invest New Drugs 25(6):525–533. doi:10.1007/s10637-007-9064-5
Reis FR, Vasconcelos FC, Pereira DL, Moellman-Coelho A, Silva KL, Maia RC (2011) Survivin and P-glycoprotein are associated and highly expressed in late phase chronic myeloid leukemia. Oncol Rep 26(2):471–478. doi:10.3892/or.2011.1296
Vasconcelos FC, Silva KL, Souza PS, Silva LF, Moellmann-Coelho A, Klumb CE, Maia RC (2011) Variation of MDR proteins expression and activity levels according to clinical status and evolution of CML patients. Cytometry B Clin Cytom 80(3):158–166. doi:10.1002/cyto.b.20580
Grandjean F, Bremaud L, Verdier M, Robert J, Ratinaud MH (2001) Sequential gene expression of P-glycoprotein (P-gp), multidrug resistance-associated protein (MRP) and lung resistance protein: functional activity of P-gp and MRP present in the doxorubicin-resistant human K562 cell lines. Anticancer Drugs 12(3):247–258
Zhou DC, Ramond S, Viguie F, Faussat AM, Zittoun R, Marie JP (1996) Sequential emergence of MRP- and MDR1-gene over-expression as well as MDR1-gene translocation in homoharringtonine-selected K562 human leukemia cell lines. Int J Cancer 65(3):365–371. doi:10.1002/(SICI)1097-0215(19960126)65:3<365:AID-IJC15>3.0.CO;2-9
Ertel A, Verghese A, Byers SW, Ochs M, Tozeren A (2006) Pathway-specific differences between tumor cell lines and normal and tumor tissue cells. Mol Cancer 5(1):55. doi:10.1186/1476-4598-5-55
Karwatsky J, Lincoln MC, Georges E (2003) A mechanism for P-glycoprotein-mediated apoptosis as revealed by verapamil hypersensitivity. Biochemistry 42(42):12163–12173. doi:10.1021/bi034149+
Barattin R, Perrotton T, Trompier D, Lorendeau D, Di Pietro A, d’Hardemare Adu M, Baubichon-Cortay H (2010) Iodination of verapamil for a stronger induction of death, through GSH efflux, of cancer cells overexpressing MRP1. Bioorg Med Chem 18(17):6265–6274. doi:10.1016/j.bmc.2010.07.031
Genoux-Bastide E, Lorendeau D, Nicolle E, Yahiaoui S, Magnard S, Di Pietro A, Baubichon-Cortay H, Boumendjel A (2011) Identification of xanthones as selective killers of cancer cells overexpressing the ABC transporter MRP1. ChemMedChem 6(8):1478–1484. doi:10.1002/cmdc.201100102
Pluchino KM, Hall MD, Goldsborough AS, Callaghan R, Gottesman MM (2012) Collateral sensitivity as a strategy against cancer multidrug resistance. Drug Resist Updat 15(1–2):98–105. doi:10.1016/j.drup.2012.03.002
Pohl A, Lage H, Muller P, Pomorski T, Herrmann A (2002) Transport of phosphatidylserine via MDR1 (multidrug resistance 1) P-glycoprotein in a human gastric carcinoma cell line. Biochem J 365(Pt 1):259–268. doi:10.1042/BJ20011880
Wartenberg M, Frey C, Diedershagen H, Ritgen J, Hescheler J, Sauer H (1998) Development of an intrinsic P-glycoprotein-mediated doxorubicin resistance in quiescent cell layers of large, multicellular prostate tumor spheroids. Int J Cancer 75(6):855–863. doi:10.1002/(SICI)1097-0215(19980316)75:6<855:AID-IJC7>3.0.CO;2-U
Myc A, DeAngelis P, Kimmel M, Melamed MR, Darzynkiewicz Z (1991) Retention of the mitochondrial probe rhodamine 123 in normal lymphocytes and leukemic cells in relation to the cell cycle. Exp Cell Res 192(1):198–202
Ruefli AA, Tainton KM, Darcy PK, Smyth MJ, Johnstone RW (2002) P-glycoprotein inhibits caspase-8 activation but not formation of the death inducing signal complex (disc) following Fas ligation. Cell Death Differ 9(11):1266–1272. doi:10.1038/sj.cdd.4401081
Smyth MJ, Krasovskis E, Sutton VR, Johnstone RW (1998) The drug efflux protein, P-glycoprotein, additionally protects drug-resistant tumor cells from multiple forms of caspase-dependent apoptosis. Proc Natl Acad Sci USA 95(12):7024–7029
Cullen KV, Davey RA, Davey MW (2001) Drug resistance does not correlate with resistance to Fas-mediated apoptosis. Leuk Res 25(1):69–75
Belloc F, Cotteret S, Labroille G, Schmit V, Jaloustre C, Dumain P, Durrieu F, Reiffers J, Boisseau MR, Bernard P, Lacombe F (1997) Bcr-abl translocation can occur during the induction of multidrug resistance and confers apoptosis resistance on myeloid leukemic cell lines. Cell Death Differ 4(8):806–814. doi:10.1038/sj.cdd.4400303
Cai Z, Stancou R, Korner M, Chouaib S (1996) Impairment of Fas-antigen expression in adriamycin-resistant but not TNF-resistant MCF7 tumor cells. Int J Cancer 68(4):535–546. doi:10.1002/(SICI)1097-0215(19961115)68:4<535:AID-IJC21>3.0.CO;2-2
Notarbartolo M, Cervello M, Dusonchet L, Cusimano A, D’Alessandro N (2002) Resistance to diverse apoptotic triggers in multidrug resistant HL60 cells and its possible relationship to the expression of P-glycoprotein, Fas and of the novel anti-apoptosis factors IAP (inhibitory of apoptosis proteins). Cancer Lett 180(1):91–101
Acknowledgments
We thank Thais M. Gameiro Marques and Bruno Paredes for their assistance in performing the IL-8 measurements. We also thank the Brazilian agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq, Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro—FAPERJ and FAPERJ/PP SUS, and INCT-Controle do Cancer for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Nathalia Daflon-Yunes and Flavio Eduardo Pinto-Silva have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Daflon-Yunes, N., Pinto-Silva, F.E., Vidal, R.S. et al. Characterization of a multidrug-resistant chronic myeloid leukemia cell line presenting multiple resistance mechanisms. Mol Cell Biochem 383, 123–135 (2013). https://doi.org/10.1007/s11010-013-1761-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1761-0