Abstract
Context
Patch use can illustrate the contributions that forage quality and foraging risk make in shaping foraging decisions made under the risk of being killed.
Objectives
We aimed to determine whether forage selection by European bison Bison bonasus was state-dependent in being shaped by risk (wolf predation or human poaching/culling avoidance) or nutrient quality at two time periods—early winter (when resources are abundant) and at the end of winter (when resources are scarce)—in Białowieża Primeval Forest (Poland).
Methods
We used a giving-up density framework using the proportional consumption of hay provided by humans to measure the perceptions of risk in European bison.
Results
European bison resource selection was primarily driven by minimising the distance they travel over open ground in early winter (i.e. avoiding humans), however by the end of winter, when resources are scarce, bison selected haystacks for their nutrient quality (low fibre, high Mg and energy).
Conclusions
This study illustrates how resource selection varies according to the condition of animals (i.e. state-dependency affects the marginal value theorem). It also indicates conservation managers using hay quality may actively manage human–bison conflicts in trouble spots and that bison are currently free from top-down limitation in Białowieża as they do not perceive wolves as threatening enough to alter their foraging behaviour.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Optimal foraging theory provides a framework for studying the perceptions of wildlife to the risks inherent in their environment (Brown 1988; Brown and Kotler 2004; Druce et al. 2006). Measuring the use of foraging patches can reveal the costs and benefits of foraging for wildlife (Brown 1988). Notably, patch use can illustrate the contribution of energy, nutrients and predation risk to a species’ fitness (Kotler et al. 1994; Hochman and Kotler 2006). Where the risk of predation is high, foragers extract more nutrients from safe patches than they do from risky patches (Brown 1988, 1992; Kotler et al. 1994; Fortin and Fortin 2009).
When harvest rates in a patch are a diminishing function of food density, giving-up densities (GUDs) are quantifiable and provide an estimate of foraging costs inter alia (Tadese and Kotler 2013). Diminishing returns in exploiting patches should drive foragers to exploit them until harvest rates decline to the level of the energetic, predation and missed opportunity costs of foraging (Brown 1988, 1992; Kotler et al. 1994).
We used GUDs to test whether forage selection by European bison (Bison bonasus)—the largest terrestrial mammal of Europe—is driven by risk (wolf predation, road accident risk or human conflict/poaching/culling avoidance) or nutrient quality. European bison were reintroduced to forest habitats after their extinction in the wild at the beginning of the twentieth century. They increasingly utilise open areas in the vicinity of forest habitats, especially in winter, to compensate for scarcity of graze within forest habitats due to the bisons’ adaptations for open or mixed habitats and its hypothesised ‘refugee’ status in forests (Kerley et al. 2012; Bocherens et al. 2015). This also reduces bison density at feeding sites and thereby reduces parasite loads (Radwan et al. 2010; Pyziel et al. 2011). In effect, this leads to human–wildlife conflict and bison disturbance because bison damage farm crops, such as winter green rape and cereals, and hay stored by farmers on meadows that were mown to obtain European Union (EU) subsidies. The amount of hay available for bison in the northern vicinity of the Białowieża Forest—an area of regular winter bison movements (Kowalczyk et al. 2013)—increased substantially after EU subsidies became available (after Poland’s accession to the EU in 2004) (Hofman-Kamińska and Kowalczyk 2012). These artificial forage patches provided an ideal GUD measurement as they are foraged at different rates and are situated at varying distances from different potential sources of risk for bison.
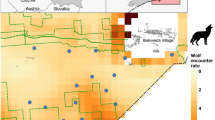
We predicted that (1) European bison in Białowieża would have higher GUDs where the risk of predation was high (closer to where wolves Canis lupus occur, as they are almost entirely restricted to the forest and therefore potentially increase their predation success through proximity, as shown in other risk-perception studies on wolves and their prey; Fig. 1a) (Altendorf et al. 2001). While wolves rarely kill bison in Białowieża (0–3 recorded kills annually or 2.6 % of all bison mortalities; Jędrzejewski et al. 2012; Krasińska and Krasiński 2013), they more frequently take bison in North America (Smith et al. 2000) to the extent that they can drive populations to near extinction (Carbyn et al. 1993). We hypothesised that (2) if disturbance/poaching/culling was a threat to bison, they would show higher GUDs at sites closer to human habitation having associated humans with risk following an increase in human–wildlife conflict with the recent expansion of bison beyond the Białowieża forest (Hofman-Kamińska and Kowalczyk 2012) (Fig. 1b). We hypothesised that (3) roads may be linear corridors of fear for bison in the region due to the risk of road kill/vehicle contact (car collisions are the most important anthropogenic factor responsible for 8 % of all mortality cases; Krasińska and Krasiński 2013), and so we predicted higher GUDs at sites close to roads (Fig. 1c). (4) We hypothesised that higher nutrient quality of the hay within the stacks would lead to lower GUDs as the rate of energy intake was higher (Fig. 1d). (5) in resource-scarce periods, low nutrients would be compensated for by high through-put that would require increased water and so close proximity to water would lead to lower GUDs (Fig. 1e; Shrader et al. 2008). Finally, we hypothesised that (6) haystacks closer to the range centre of the nearest herd would be foraged upon more heavily than those further from the range centre if accessing patches was limiting (Fig. 1f). We measured the GUDs (% of haystack remaining) twice (at the start and end of winter in early December 2007 and the end of March 2008) to test whether the factors described above alter the habitat quality or landscape of fear of European bison when they are under different conditional states.
Methods
The study site was a 6 × 5 km2 area at the north of Białowieża Primeval Forest (Białowieża) in eastern Poland (Fig. 2). Białowieża is an example of the original ecosystem of continental Europe with largely intact predator and large herbivore guilds and a ‘strict reserve’ that has not been impacted by humans for logging for over 100 years (Jędrzejewska and Jędrzejewski 1998). European bison were saved from extinction at Białowieża (Krasińska and Krasiński 2007).
Farmers surrounding Białowieża are paid by the European Union to maintain the meadows. Mowed hay is then left by farmers on their meadows over winter to supplementary feed bison. Farmers use four haystack designs to store the hay: (1) a random pile of cut hay; (2) a more structured pile of hay; (3) hay piled on a wooden tee-pee structure; and (4) roofed haystacks. Although moose Alces alces, red deer Cervus elaphus and roe deer Capreolus capreolus are present in Białowieża, bison are the only bulk grazer that regularly leaves the forest to forage on open areas, and this was verified by footprints on the ground. The proportional amount of hay consumed from each haystack was estimated by comparing the original volume of hay against that remaining. The hay volume at the beginning and end of winter was estimated on the basis of height, diameter and shape of haystacks (that is equations used to estimate the volumes were those of a sphere for flat-topped haystacks or cone for pointy topped haystacks). Haystacks were measured by tape at the start and end of winter 2007/2008, when climatic conditions were considered average (Kowalczyk et al. 2011).
We tested whether there were any differences in the use of hay between haystack designs using an ANOVA. We ran two types of models. Firstly, we ran binary logistic regression using whether a haystack was foraged upon (1) or not (0) as the dependent variable to test which factors determined the selection of forage by bison. Secondly, we used linear regression with the percentage of hay forage consumed (GUD) as the dependent variable to determine which factors determined the relative amount foraged by bison. Such modelling can distinguish between the causes of resource selection and consumption (Hayward et al. 2007). We first ran the model including the time period (start/end of winter) variable to determine if there were differences in the foraging decisions made by bison at these time periods. We then ran these models again separately for the two different time periods (start/end of winter; n = 91 for all) to test whether natural resource scarcity (i.e. at the end of winter) altered the foraging decisions of bison. Early winter was defined as the period prior to the seasonal food shortages (i.e. data collected early December), while the end of winter is a period of seasonal food shortages within the forest and data was collected in March. Thus we are testing how state-dependency (nutritional status here, but also predation risk as described below) affects the marginal value theorem (Nonacs 2001). We had no direct data on body condition of bison during our study period (due the a low number of bison culled that year), but long-term data on body mass change throughout winter (December to March) based on between two and eight adult females culled annually between 2000 and 2013 indicate a significant decline in condition (10.1 % decline in body mass over winter; r 2 = 0.102, n = 92, p = 0.002; R. Kowalczyk unpubl. data).
There are three assumptions of the marginal value theorem that GUDs are based on (Charnov 1976). Firstly, individuals must be free to choose when to leave a patch to maximise the ratio between food intake and time. Secondly, the rate of intake of food in a patch decreases as a function of time, and finally, patch quality varies throughout the landscape. Clearly, bison were free to move throughout the landscape and haystack quality varied. Food intake decreased over time (albeit non-linearly) as haystacks became eaten down and trodden upon. Most studies using GUDs rely on the repeated experimental provisioning of feed (e.g., seed trays or mealworms) daily to measure the amount of food given up (remaining) (Brown and Kotler 2004). Natural GUD studies are more challenging because the initial quantity of available food and its quality is hard to measure (Brown and Kotler 2004). Tadese and Kotler (2013) used the diameter of vegetation at the point of browse by mountain nyala Tragelaphus buxtoni as a measure of GUD (a one-off measure at each foraging point), while ignoring variation in nutrient quality of different plant species. Olsson et al. (1999) used the beetle larvae remaining within branches after woodpeckers had foraged on them as a measure of GUD—these beetles varied in size (hence quality), and were not replenished (the branches were chopped off) and were very unlikely to be equal from the start (all stations were not equal). The haystacks we used did not consist of the same volume, but the change in volume (GUD) was measureable (as described above). Furthermore, we measured the quality of the haystacks to investigate how this issue affects the marginal value theorem. Hence, we believe our experimental design fits within a GUD framework.
We assessed the bivariate correlations between the percentage of hay consumed and the potential predictor variables: distance to nearest forest (Forest); distance to nearest house (Human); distance to nearest source of water (Water); distance to nearest road (Road); and distance to home range centre (Home Range). These habitat variables were measured directly in the field in metres using a Leica rangefinder or on digital maps using the MapInfo GIS program. Distance to the home range centre was measured comparing haystack location and the estimated center of minimum convex polygon range based on GIS-collared bison tracking of three individuals (two females aged 4 and 6 years, and one male aged 11; 24 GPS-localisations a day). The range was estimated using Biotas™ v. 1.03 (Ecological Software Solutions LLC). There were 70–80 bison in the area moving in different sized groups (from 2 to 60 individuals, mean 23.1), with completely overlapping home ranges (Kowalczyk et al. unpubl. data). We ran the models using Akaike’s Information Criteria corrected for small sample size (AICc) to select the most supported model. Models with ΔAICc < 2 showed strong support, those with ΔAICc between 2 and 7 showed some support, and those with ΔAICc > 7 were not supported (Burnham and Anderson 2001). We present the results of the most supported models (ΔAICc < 2).
To analyse nutrient content of the hay, we sampled a handful of hay from the centre of randomly selected haystacks (n = 38) and determined: energy (kJ/g); % dry matter DM; macronutrients: crude fat (% DM); crude protein (% DM); starch (% DM); glucose (% DM); sucrose (% DM); fructose (% DM); neutral detergent fibre (NDF; % DM); acid detergent fibre (ADF; % DM); acid detergent lignin (ADL; % DM); cellulose (% DM); hemi-cellulose (% DM); crude fibre (% DM); crude ash (% DM); minerals: Ca; Cu; Fe; Zn; Mg; Mn; Na and K (each mg/kg DM). We excluded variables that were significantly correlated to minimise the number of variables modelled in accordance with Harrell’s (2001) ‘rule of thumb’ and so modelled the habitat variable with the strongest support alongside these nutrient variables to test whether nutrients were more important in driving forage selection than habitat features. For hay nutrient analyses, the following standard methods were used: to determine dry matter a subsample of the hay was dried at 105 °C until constant weight, energy was determined via bomb calorimetry and crude fat via ethyl ether extraction (Soxhlett method). For crude protein analysis, a subsample was burned at high temperature (about 950 °C) in pure oxygen (Dumas combustion). This provides the nitrogen content of the sample and crude protein was calculated after CP = N × 6.25. Starch, sucrose, glucose, and fructose were determined with the help of enzymatic tests and crude ash by burning dried sample in a muffle furnace for at least 2 h at 550 °C, after which only inorganic matter remains and amount of ash was obtained by difference in weights. The detergent system of fiber analysis after Van Soest (1994) was used to estimate the amount of indigestible carbohydrates in samples. This method consists of dissolving successively soluble cell components, hemicelluloses and celluloses. We obtain sequentially NDF, ADF and ADL or lignin. Mineral analysis samples were microwave digested and analyzed by atom-absorption-spectroscopy (AAS).
The variables percentage hay consumed, distance to forage, water, road and home range centre, glucose, sucrose, fructose, crude ash, iron and potassium were all log transformed to improve normality based on inspections of Q–Q plots in R. All analyses were conducted in R (R Core Development Team 2008), and model selection was via the MuMin package (Barton 2013). Means are presented ±1 SE.
Results
There was no significant difference in the percentage of forage eaten within each haystack type early in winter or at the end of winter (one-way ANOVA: early F 3,87 = 1.246, p = 0.298; end F 3,87 = 0.553, p = 0.648). Consequently, haystack type was excluded from modelling. Generalised linear models revealed time (early or late winter) was the primary driver of variability in the global model (∑Timew i = 0.96) and was almost 40 % more important than the next most important variable and we subsequently analysed the data separately for early and late winter.
Energy (kJ/g), NDF (fibre) and magnesium (Mg) were correlated with all other variables (Table 1) and were selected for the final nutrient modelling. Distance to forest edge was significantly correlated with distance to the nearest house (R = 0.393, n = 91, p > 0.05), so the following habitat variables were modelled: log Distance to Forest; log Distance to Water; log Distance to Road; and log Distance to Home Range Centre. There was also a significant correlation between the percentage of hay consumed in early winter with that at the end of winter (R = 0.628, n = 91; p < 0.001).
Forty-two (42) of the 91 haystacks (46 %) had been fed upon in early winter, and 14 more (56) were fed on by the end of winter (62 %). There was an estimated 90 ± 2 % of hay in the haystacks remaining in early winter, while 74 ± 3 % remained at the end of winter. If we restrict this to only haystacks that were foraged upon, they had 80 ± 4 % remaining at the start of winter and 58 ± 4 % remaining at the end of winter.
The binary logistic regression models of the drivers of whether a haystack was foraged upon or not at the start of winter revealed substantial support for six models with the most supported including distance to forest and water (AICc = 121.17; Table 2a). Akaike’s weights showed that proximity to forest was the primary variable with almost 35 % more support than the next variable (∑forestw i = 0.81; ∑roadw i = 0.53; ∑Waterw i = 0.47; ∑Home Range Centrew i = 0.31). Haystacks were more likely to be eaten if they were closer to the forest edge and therefore further from human habitation (Fig. 3), rejecting hypothesis 1 but supporting hypothesis 2. Similar binary logistic models run at the end of winter revealed substantial support for proximity to forest as well as distance from home range centre (AICc = 113.70; Table 2b). Akaike’s weights supported this with close proximity to forest and long distances from home range centre the primary variables with up to 47 % more support than the other variables (∑Forestw i = 0.88; ∑Home Range Centrew i = 0.74; ∑Roadw i = 0.50; ∑Waterw i = 0.47; Fig. 3). These results again reject hypothesis 1 and provide no support for the other hypotheses.
When reanalysed with nutrient data alongside proximity to forest for the start of winter data, eight binary logistic models showed substantial support, but with the most supported model including proximity to forest and energy content of the hay in the haystack (AICc = 35.32; Table 2c). Akaike’s weights showed that proximity to forest was still the primary driver of which haystacks bison sample early in winter with 45–81 % more support than the other variables (∑Forestw i = 0.81; ∑Energyw i = 0.52; ∑Mgw i = 0.47; ∑NDFw i = 0.45; Fig. 4). These results again reject hypothesis 1 and provide no support for the other hypotheses. By the end of winter, bison selected haystacks based on proximity to forest and home range centres, plus a mix of nutrient variables (AICc = 32.46; Table 2d). In concordance with previous results, Akaike’s weights showed that proximity to forest was still the primary driver of which haystacks bison sampled by the end of winter with 19–48 % more support than the other variables (∑Forestw i = 0.79; ∑Energyw i = 0.64; ∑NDFw i = 0.54; ∑Home Range Centrew i = 0.52; ∑Mgw i = 0.41).
The linear regression model of habitat features driving the amount of forage eaten revealed most support for proximity to home range centre as the primary driver of the amount of hay eaten by European bison at the start of winter (Akaike’s weight w i = 0.36; Table 3a). There was also support for models that included distances to forest and water alongside distance to home range centre (Table 3a). The sum of Akaike’s weights revealed home range centre (∑w i = 0.90) as the most important variable, with other variables offering less than one-third of the importance (∑waterw i = 0.30; ∑forestw i = 0.28; ∑roadw i = 0.22). Bison consumed more hay further from their home range centre (Fig. 5), refuting hypothesis 6. By the end of winter, bison were far less selective with no variable clearly driving selection and all top models exhibiting strong support (Table 3b). Sum of Akaike’s weights support this (∑waterw i = 0.52;∑home range centrew i = 0.38; ∑roadw i = 0.38; ∑forestw i = 0.21).
The linear regression model of drivers for the amount of forage eaten for the start of winter that included nutrient features and the previously identified key driver of bison resource use (proximity to home range centre) showed that high fibre content was almost as important as the distance the bison needed to move (∑home range centrew i = 0.69; ∑NDFw i = 0.66), with Mg and energy being less important (∑Mgw i = 0.53; ∑Energyw i = 0.15; Table 3c). Haystacks with more fibre were increasingly consumed (Fig. 6). By the end of winter, bison were far more prepared to move longer distances as trace elements (∑Mgw i = 0.52), fibre (∑NDFw i = 0.48) and energy (∑Energyw i = 0.69) became almost four times more important than proximity to home range centre (∑home range centrew i = 0.14; Table 3d); supporting hypothesis 4. Bison consumed more hay that had lower fibre and higher Mg and energy (Fig. 6). There was no support for hypotheses 3 (roads) or 5 (water).
Discussion
This GUD study reveals that the perception of risk in an animal’s environment varies according to the condition of animals, that is, state-dependent behaviour clearly affects the marginal value theorem (Nonacs 2001). Bison lose condition over winter and, ultimately, select forage resources based on their higher nutrient content reflecting a trade-off whereby they accept higher risk of being killed when forage quality decreases over winter (i.e. starvation risk increases). Proximity to the forest edge was the primary driver of bison sampling haystacks early in winter (Fig. 3), suggesting bison perceived crossing large areas of open grassland near human habitation to access haystacks as risky. Thus there is no support for the predator (wolf) avoidance hypotheses (1), and this supports other research on risk perception in ungulates in Białowieża (Theuerkauf and Rouys 2008). There is however some support for avoidance of humans in reducing the distance they travelled across open areas (hypothesis 2). This makes biological sense as wolves rarely prey on bison in Białowieża (Jędrzejewska and Jędrzejewski 1998; Jędrzejewski et al. 2012; Krasińska and Krasiński 2013), however humans regularly do so by culling sickly individuals (up to 17 % of bison are shot annually for population regulation; Krasińska and Krasiński 2004). Humans also increasingly chase bison from fields in conflict situations. There was no evidence that roads affected bison foraging decisions (refuting hypothesis 3). Thus, predator avoidance (human or wolf), threat of road kill and distance required to access food patches did not drive bison forage selection. At the end of winter, European bison still sampled haystacks based on their proximity to the forest edge and their home range centre suggesting that they perceived the same factors as important (forest/home range centre—no predation risk/optimal foraging). Home ranges of bison using the same parts of the forest/forest surrounds strongly overlap (Krasińska et al. 2000). This was also the case for the three individuals tracked in the study area during the study period (R. Kowalczyk pers. obs.). Tracked animals were often ranging together, so sex or status had no influence on the home range size and movement patterns. The abundance of haystacks within the bison range suggests social foraging (Shrader et al. 2012) did not cause these results as all haystacks would have been equally affected.
Foraging resource selection is driven by optimality theory (Charnov 1976; Pyke et al. 1977; Krebs 1980; Belovsky 1984), however for the bison this meant travelling further to access higher quality hay (reflecting the nutritional state-dependency in this test of the marginal value theorem). This also drove the amount of haystack consumed. Nutrition quality was the primary determinant of hay consumption when resources were scarce at the end of winter (supporting hypothesis 4), however there was no evidence of compensatory drinking to cope with poorer quality forage (refuting hypothesis 5), although water from snow may be taken despite the energetic costs associated with this during cold periods. Distance from home range centre affected haystack selection and consumption, but opposite that predicted based on optimality theory of minimising energy expenditure (refuting hypothesis 6), illustrating that bison were so short of forage within the forest that they would travel further and then consume more once they selected an optimal patch (Courant and Fortin 2010; Babin et al. 2011; Kowalczyk et al. 2011). From spring to summer, European bison remain within forested habitats in Białowieża. This may explain their avoidance of open areas at the beginning of winter, which results in less selective foraging. But, as winter conditions continue the need to selectively forage on higher quality hay overrides avoidance of open habitat. When resources become scarce at the end of winter, bison extended their haystack foraging selection strategy to include nutrients (vitamins, fibre and energy) as bison were more prepared to travel further to select haystacks with these features. More fibrous forage leads to increased processing time, but yields less digestible content and ingestion rate of energy (Tadese and Kotler 2013), illustrating why bison selected against high fibre content of hay. In contrast, less fibrous forage will be processed faster and will yield a higher digestible content and energy ingestion (i.e. lower GUDs) (Courant and Fortin 2010; Babin et al. 2011).
Single snapshot studies using GUDs that ignore changes in condition of the study species may miss crucial information about the perception of risk that animals face in the environment. As animals become nutrient stressed, their resource selection decisions are likely to change leading to greater risks taken to obtain critical resources. Similarly, it has recently been shown that drought alters the ‘landscape of fear’ for African ungulates (Riginos 2015). Hence, we recommend looking at animal risk perception over longer term scales as we have here.
Despite the risk of starvation conservation managers perceive of European bison (Hayward et al. 2011), there was a surprisingly large proportion of hay remaining at the end of winter (~58 % of available hay), even though a large amount is wasted during foraging through trampling etc. This suggests that the meadows surrounding Białowieża Primeval Forest may have provided sufficient food for the local bison herds to persist through the essentially average winter, in regard to temperature, duration and snow depth, of 2007–2008 (see Supplementary Material). We did not monitor frequently enough to determine whether there were distinct periods that bison sought out the haystacks to see them through particularly stressful conditions or whether they foraged on them purely as they came across them in their normal ranging behaviour and this could be an opportunity for future study. The hay may also be critical in harsher winters. Alternatively, the hay may have been too poor in quality for bison to expend the energy in foraging upon rather than remain foraging upon wild foods (Fortin 2003).
Given the managers of Białowieża National Park pay local farmers with European Union subsidies to provide the hay in the haystacks studied here and these subsidies seem unlikely to stop despite the perverse outcomes they are yielding and problems they cause, our results suggest they would improve the value of those haystacks by dictating the quality of hay that is used. Hay quality varies throughout the year and is highest for the first mow and when cut early in the growing season before seeds have set (Snyman and Joubert 1996; Norton et al. 1997; Kallah et al. 1999; Yari et al. 2012). By paying only for early-cut hay, the managers will be ensuring that the quality of hay is attractive to European bison and thereby obtain greater foraging rates on the haystacks. A more sustainable management option would be to manage bison distribution, and therefore human–bison conflict, through the management of food distribution and quality (Hofman-Kamińska and Kowalczyk 2012) or allow for the expansion of the increasing bison population beyond the forest boundary into meadows (possibly reclaimed from disused agricultural land). For example, high quality haystacks should be situated near the forest boundary and away from conflict areas to ensure bison obtain sufficient nutrients over the season of nutrient stress, while also minimising the risk of conflict.
References
Altendorf KB, Laundre JW, Lopez Gonzalez CA, Brown JS (2001) Assessing effects of predation risk on foraging behavior of mule deer. J Mammal 82:430–439
Babin JS, Fortin D, Wilmshurst JF, Fortin M-J (2011) Energy gains predict the distribution of plains bison across populations and ecosystems. Ecology 92:240–252
Barton K (2013) Package ‘MuMIn’. R Statistics. The Comprehensive R Archive Network (CRAN), Vienna
Belovsky GE (1984) Herbivore optimal foraging: a comparative test of three models. Am Nat 124:97–115
Bocherens H, Hofman-Kamińska E, Drucker DG, Schmölcke U, Kowalczyk R (2015) European bison as a refugee species? Evidence from isotopic data on early Holocene bison and other large herbivores in northern Europe. PLoS ONE 10(2):e0115090
Brown JS (1988) Patch use as an indicator of habitat preference, predation risk, and competition. Behav Ecol Sociobiol 22:37–47
Brown JS (1992) Patch use under predation risk. 1. Models and predictions. Ann Zool Fenn 29(4):301–309
Brown JS, Kotler BP (2004) Hazardous duty pay and the foraging cost of predation. Ecol Lett 7:999–1014
Burnham KP, Anderson DJ (2001) Kullback-Leibler information as a basis for strong inference in ecological studies. Wildl Res 28(2):111–119
Carbyn LN, Oosenbrug SM, Anions DW (1993) Wolves, bison, and the dynamics related to the Peace-Athabasca Delta in Canada’s Wood Buffalo National Park. Circumpolar Res Ser 4:1–270
Charnov EL (1976) Optimal foraging, marginal value theorem. Theor Popul Biol 9(2):129–136
Courant S, Fortin D (2010) Foraging decisions of bison for rapid energy gains can explain the relative risk to neighboring plants in complex swards. Ecology 91:1841–1849
Druce D, Brown JS, Castley JG, Kerley GI, Kotler BP, Slotow R, Knight MH (2006) Scale-dependent foraging costs: habitat use by rock hyraxes (Procavia capensis) determined using giving-up densities. Oikos 115:513–525
Fortin D (2003) Searching behavior and use of sampling information by free-ranging bison (Bos bison). Behav Ecol Sociobiol 54:194–203
Fortin D, Fortin M-J (2009) Group-size-dependent association between food profitability, predation risk and distribution of free-ranging bison. Anim Behav 78(4):887–892
Harrell FE (2001) Regression modeling strategies: with applications to linear models, logistic regression and survival analysis. Springer, New York
Hayward MW, de Tores PJ, Dillon MJ, Banks PB (2007) Predicting the occurrence of the quokka, Setonix brachyurus (Macropodidae: Marsupialia), in Western Australia’s northern jarrah forest. Wildl Res 34:194–199
Hayward MW, Kowalczyk R, Krasiński ZA, Krasińska M, Dackiewicz J, Cornulier T (2011) Restoration and intensive management have no effect on evolutionary strategies. ESR 15:53–61. doi:10.3354/esr00371
Hochman V, Kotler BP (2006) Effects of food quality, diet preference and water on patch use by Nubian ibex. Oikos 112(3):547–554
Hofman-Kamińska E, Kowalczyk R (2012) Farm crops depredation by European bison (Bison bonasus) in the vicinity of forest habitats in northeastern Poland. Environ Manag 50(4):530–541
Jędrzejewska B, Jędrzejewski W (1998) Predation in vertebrate communities: the Bialowieza Primeval Forest as a case study. Springer, Berlin
Jędrzejewski W, Niedzialkowska M, Hayward MW, Goszczyński J, Jędrzejewska B, Borowik T, Bartoń KA, Nowak S, Harmuszkiewicz J, Juszczyk A, Kałamarz T, Kloch A, Koniuch J, Kotiuk K, Mysłajek RW, Nędzyńska M, Olczyk A, Telon M, Wojtulewicz M (2012) Prey choice and diet of wolves related to ungulate communities and wolf subpopulations in Poland. J Mammal 93(6):1480–1492
Kallah MS, Muhammad IR, Baba M, Lawal R (1999) The effect of maturity on the composition of hay and silage made from Columbus grass (Sorghum almum). Trop Grassl 33(1):46–50
Kerley GIH, Kowalczyk R, Cromsigt JPGM (2012) Conservation implications of the refugee species concept and the European bison: king of the forest or refugee in a marginal habitat? Ecography 35(6):519–529
Kotler BP, Gross JE, Mitchell WA (1994) Applying patch use to assess aspects of foraging behavior in Nubian ibex. J Wildl Manag 58(2):299–307
Kowalczyk R, Taberlet P, Coissac E, Valentini A, Miquel C, Kamiński T, Wójcik JM (2011) Influence of management practices on large herbivore diet—case of European bison in Bialowieza Primeval Forest (Poland). For Ecol Manag 261:821–828
Kowalczyk R, Krasińska M, Kamiński T, Górny M, Struś P, Hofman-Kamińska E, Krasiński ZA (2013) Movements of European bison (Bison bonasus) beyond the Białowieża Forest (NE Poland): range expansion or partial migrations? Acta Theriol 58(4):391–401
Krasińska M, Krasiński ZA (2004) Life of the European bison. In: Jedrzejewska B, Wojcik JM (eds) Essays on mammals of Bialowieza forest. Mammal Research Institute, Polish Academy of Science, Bialowieza, pp 35–42
Krasińska M, Krasiński ZA (2007) The European bison: a nature monograph. Mammal Research Institute, Polish Academy of Science, Bialowieza
Krasińska M, Krasiński ZA (2013) The European bison: the nature monograph. Springer, Berlin
Krasińska M, Krasiński ZA, Bunevich AN (2000) Factors affecting the variability in home range size and distribution in European bison in the Polish and Belarussian parts of the Białowieża Forest. Acta Theriol 45(3):321–334
Krebs JR (1980) Optimal foraging, predation risk and territory defence. Ardea 68:83–90
Nonacs P (2001) State dependent behavior and the Marginal Value Theorem. Behav Ecol 12(1):71–83
Norton NA, Clark RT, Reece PE, Eskridge KM (1997) Quality as a factor in the optimal choice of fertilization and harvest date of meadow hay. J Prod Agric 10(4):551–557
Olsson O, Wiktander U, Holmgren NM, Nilsson SG (1999) Gaining ecological information about Bayesian foragers through their behaviour. II. A field test with woodpeckers. Oikos 87:264–276
Pyke GH, Pulliam HR, Charnov EL (1977) Optimal foraging: a selective review of theory and tests. Q Rev Biol 52(2):137–154
Pyziel AM, Kowalczyk R, Demiaszkiewicz AW (2011) The annual cycle of shedding Eimeria oocysts by European bison (Bison bonasus) in the Bialowieza Primeval Forest, Poland. J Parasitol 97(4):737–739
R Core Development Team (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Radwan J, Demiaszkiewicz AW, Kowalczyk R, Lachowicz J, Kawałko A, Wójcik JM, Pyziel AM, Babik W (2010) An evaluation of two potential risk factors, MHC diversity and host density, for infection by an invasive nematode Ashworthius sidemi in endangered European bison (Bison bonasus). Biol Conserv 143:2049–2053
Riginos C (2015) Climate and the landscape of fear in an African savanna. J Anim Ecol 84(1):124–133
Shrader AP, Kotler B, Brown J, Kerley GIH (2008) Providing water for goats in arid landscapes: effects on feeding effort with regard to time period, herd size and secondary compounds. Oikos 117(3):466–472
Shrader AM, Kerley GIH, Brown JS, Kotler BP (2012) Patch use in free-ranging goats: does a large mammalian herbivore forage like other central place foragers? Ethology 118(10):967–974
Smith DW, Mech LD, Meagher M, Clark WE, Jaffe R, Phillips MK, Mack JA (2000) Wolf-bison interactions in Yellowstone National Park. J Mammal 81(4):1128–1135
Snyman LD, Joubert HW (1996) Effect of maturity stage and method of preservation on the yield and quality of forage sorghum. Anim Feed Sci Technol 57(1–2):63–73
Tadese SA, Kotler BP (2013) Habitat use by mountain nyala (Tragelaphus buxtoni) determined using bite diameters at point of browse, bite rates, and time budgets in the Bale Mountains National Park, Ethiopia. Curr Zool 59:707–717
Theuerkauf J, Rouys S (2008) Habitat selection by ungulates in relation to predation risk by wolves and humans in the Białowieża Forest, Poland. For Ecol Manag 256:1325–1332
Van Soest PJ (1994) Nutritional ecology of the ruminant. Cornell University Press
Yari M, Valizadeh R, Naserian AA, Jonker A, Yu P (2012) Modeling nutrient availability of alfalfa hay harvested at three stages of maturity and in the afternoon and morning in dairy cows. Anim Feed Sci Technol 178(1–2):12–19
Acknowledgments
We thank Graham Kerley and Adrian Shrader for assistance in addressing the referees concerns about our use of a giving-up density framework, and thank the four referees for their efforts. The project was financed by Polish Ministry of Science and Higher Education Grant Nos. 2P04F 011 26 and N N304 253435, and the Mammal Research Institute budget. MWH was funded by BIORESC MTKD-CT-2005-029957 (Marie Curie Transfer of Knowledge Program of the European Union).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hayward, M.W., Ortmann, S. & Kowalczyk, R. Risk perception by endangered European bison Bison bonasus is context (condition) dependent. Landscape Ecol 30, 2079–2093 (2015). https://doi.org/10.1007/s10980-015-0232-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-015-0232-2