Abstract
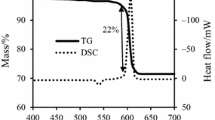
In order to investigate relative reactivity of different oxidants in solid-state reactions of pyrotechnic mixtures, thermal properties of Sn + Sr(NO3)2, Sn + Ba(NO3)2, and Sn + KNO3 pyrotechnic systems have been studied by means of TG, DTA, and DSC methods and the results compared with those of pure oxidants. The apparent activation energy (E), ΔG #, ΔH #, and ΔS # of the combustion processes were obtained from the DSC experiments. The results showed that the nature of oxidant has a significant effect on ignition temperature, and the kinetic of the pyrotechnic mixtures’ reactions, and the relative reactivity of these mixtures was found to obey in the following order: Sn + Sr(NO3)2 > Sn + Ba(NO3)2 > Sn + KNO3.



Similar content being viewed by others
References
Brown ME. Some thermal studies on pyrotechnic compositions. J Therm Anal Calorim. 2001;65:323–34.
Sivapirakasam SP, Surianarayanan M, Chandrasekaran F, Swaminathan G. Thermal hazards of cracker mixture using DSC. J Therm Anal Calorim. 2004;78:799–808.
Roduit B, Borgeat C, Berger B, Folly P, Andres H, Schädeli U, Vogelsanger B. UP-scaling of DSC DATA of high energetic materials, simulation of cook-off experiments. J Therm Anal Calorim. 2006;85:195–202.
Berger B, Brammer AJ, Charsley EL, Rooney JJ, Warrington SB. Thermal analysis studies on the boron–potassium perchlorate–nitrocellulose pyrotechnic system. J Therm Anal. 1997;49:1327–55.
Hosseini SG, Pourmortazavi SM, Hajimirsadeghi SS. Thermal decomposition of pyrotechnic mixtures containing sucrose with either potassium chlorate or potassium perchlorate. Combust Flame. 2005;14:322–6.
Roduit B, Borgeat C, Berger B, Folly P, Alonso B, Aebischer JN, Stoessel F. Advanced kinetic tools for the evaluation of decomposition reactions, determination of thermal stability of energetic materials. J Therm Anal Calorim. 2005;80:229–36.
Charsley EL, Laye PG, Brown ME. Handbook of thermal analysis and calorimetry: pyrotechnics. 1st ed. Amsterdam: Elsevier; 2003. p. 777–815.
Akhavan J. The chemistry of explosives. 2nd ed. London: The Royal Society of Chemistry; 2004.
Ianoş R, Lazău I, Păcurariu C. Metal nitrate/fuel mixture reactivity and its influence on the solution combustion synthesis of γ-LiAlO2. J Therm Anal Calorim. 2007;97:209–14.
Patnaik P. Handbook of inorganic chemicals. New York: McGraw-Hill Companies, Inc; 2003.
Cashdollar KL, Cashdollar I, Zlochower A. Explosion temperatures and pressures of metals and other elemental dust clouds. J Loss Prev Proc. 2007;20:337–48.
Cashdollar KL. Flammability of metals and other elemental dust clouds. Process Saf Prog. 1994;13:139–45.
Yoganarasimhan SR, Josyulu OS. Reactivity of the ternary pyrotechnic system red lead–silicon-ferric oxide, Defense. Sci J. 1987;37:73–83.
Knowlton GD, Ludwig CP. Auto-ignition composition, WO1997/045294, 12 Apr 1997.
Pourmortazavi SM, Hajimirsadeghi SS, Hosseini SG. Characterization of the aluminum/potassium chlorate mixtures by simultaneous TG-DTA. J Therm Anal Calorim. 2006;84:557–61.
Stets VP, Karlov VP, Butuzov GN, Ryabukha AA. Heat treatment of tin powder. Powder Metall Met C+. 1989;28:837–9.
de Klerk WPC, Krabbendam-LaHaye ELM, Berger B, Brechbuhl H, Popescu C. Thermal studies to determine the accelerated ageing of flares. J Therm Anal Calorim. 2005;80:529–36.
Kosanke KB, Kubota N, Sturman B, Jennings-White C. Pyrotechnic chemistry. Pyrotechnic reference series no.4. USA: Journal of pyrotechnics, Inc.; 2004.
Ellern H. Military and civilian pyrotechnic. New York: Chemical Publishing Company Inc; 1968.
McLain JH. Pyrotechnics from the viewpoint of solid state chemistry. Philadelphia, Penna: The Franklin Institute Press; 1980.
Rugunanan RA, Brown ME. Reactions of powdered silicon with some pyrotechnic oxidants. J Therm Anal Calorim. 1991;37:1193–211.
Turcotte R, Fouchard RC, Turcotte A-M, Jones DEG. Thermal analysis of black powder. J Therm Anal Calorim. 2003;73:105–18.
Freeman ES. The kinetics of the thermal decomposition of potassium nitrate and of the reaction between potassium nitrite and oxygen. J Am Chem Soc. 1957;79:838–42.
Shimizu T. Fireworks. The art science and technique. USA: Pyrotechnica Publications; 1981.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem.1957;29:1702–6.
Lehmann B, Karger-Kocsis J. Isothermal and non-isothermal crystallisation kinetics of pCBT and PBT. J Therm Anal Calorim. 2009;95:221–6.
Hatakeyama T, Quinn FX. Thermal analysis, fundamentals and applications to polymer science fundamentals and applications to polymer science. New York: Wiley; 1994.
ASTM E698-05. Standard test method for Arrhenius kinetic constants for thermally unstable materials. doi:10.1520/E0698-05.
Criado JM, Perez-Maqueda LA, Sanchez-Jimenez PE. Dependence of the preexponential factor on temperature. J Therm Anal Calorim. 2005;82:671–5.
Eslami A, Hosseini SG, Pourmortazavi SM. Thermoanalytical investigation on some boron-fuelled binary pyrotechnic systems. Fuel. 2008;87:3339–43.
Wang T, Lu YX, Zhu ML, Zhang JS, Ji SJ. DSC research on critical temperature in thermal explosion synthesis reaction Ti+ 3Al→TiAl3. J Therm Anal Calorim. 2002;67:605–11.
Zhang TL, Hu RZ, Xie Y, Li FP. The estimation of critical temperatures of thermal explosion for energetic materials using non-isothermal DSC. Thermochim Acta. 1994;244:171–6.
Missler GL, Tarr A. Inorganic chemistry. 4th ed. Upper Saddle River: Pearson Prentice Hall; 2010.
Markowitz MM, Boryta DA. The differential thermal analysis of perchlorate. J Phys Chem. 1964;68:2282–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hosseini, S.G., Eslami, A. Thermoanalytical investigation of relative reactivity of some nitrate oxidants in tin-fueled pyrotechnic systems. J Therm Anal Calorim 101, 1111–1119 (2010). https://doi.org/10.1007/s10973-010-0813-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-010-0813-x