Abstract
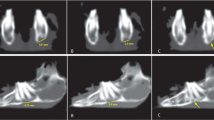
Mandibular defects, caused by congenital, pathological or iatrogenic insults, can significantly affect patient quality of life. The reconstruction of mandible has recently gained the interest of clinical and tissue engineering researchers. The purpose of this study was to evaluate the effectiveness of three-dimensional (3-D) cultured autologous grafts prepared using bone marrow-derived mesenchymal stem cells (BMSCs) combined with demineralized bone matrix (DBM) scaffolds for the restoration of mandibular defects. Cylindrical defects were created in the mandibular body of minipigs and filled with 3D-cultured BMSCs/DBM autografts, 2D-cultured BMSCs/DBM autografts, DBM material (without cells), or were left unfilled (blank). Using computed tomographic (CT) imaging and histological staining, we found that treatment of mandibular defects using 3-D cultured BMSCs/DBM autografts offered improvements in bone formation over both 2-D cultured autografts and cell-free DBM scaffolds. We found increased osteoid formation in 3D and 2D cultures, with more osteogenic cells present in the 3D constructs. We suggest that 3-D cultured homograft BMSCs combined with DBM scaffolds represents a new strategy for bone reconstruction, with potential future clinical applicability.






Similar content being viewed by others
References
Salgado AJ, Oliveira JT, Pedro AJ, Reis RL. Adult stem cells in bone and cartilage tissue engineering. Curr Stem Cell Res Ther. 2006;1:345–64.
Bhattacharjee P, Kundu B, Naskar D, Kim HW, Maiti TK, Bhattacharya D. et al. Silk scaffolds in bone tissue engineering: an overview. Acta Biomater. 2017;63:1–17.
Ciuluvica R, Gradinaru S, Popescu M, Piticescu RM, Cergan R. Computer tomograph (CT) imaging of mandibular anatomical substrate in animal model restored with nanostructured hydroxyapatite compounds. J Med Life. 2015;8:99–102.
No YJ, Li JJ, Zreiqat H. Doped calcium silicate ceramics: a new class of candidates for synthetic bone substitutes. Materials (Basel). 2017;10:2.
Xu Y, Li C, Zhou T, Su Y, He X, Fan X, et al. Treatment of aseptic necrosis of the lunate bone (Kienbock disease) using a nickel-titanium memory alloy arthrodesis concentrator: a series of 24 cases. Med (Baltim). 2015;94:e1760.
Chen B, Lin H, Wang J, Zhao Y, Wang B, Zhao W, et al. Homogeneous osteogenesis and bone regeneration by demineralized bone matrix loading with collagen-targeting bone morphogenetic protein-2. Biomaterials. 2007;28:1027–35.
Huber E, Pobloth AM, Bormann N, Kolarczik N, Schmidt-Bleek K, Schell H, et al. Demineralized bone matrix as a carrier for bone morphogenetic protein-2: burst release combined with long-term binding and osteoinductive activity evaluated in vitro and in vivo. Tissue Eng Part A. 2017.
Jiang XR, Yang HY, Zhang XX, Lin GD, Meng YC, Zhang PX, et al. Repair of bone defects with prefabricated vascularized bone grafts and double-labeled bone marrow-derived mesenchymal stem cells in a rat model. Sci Rep. 2017;7:39431.
Hsiao HY, Yang SR, Brey EM, Chu IM, Cheng MH. Hydrogel delivery of mesenchymal stem cell-expressing bone morphogenetic protein-2 enhances bone defect repair. Plast Reconstr Surg Glob Open. 2016;4:e838.
Han S, Zhao Y, Xiao Z, Han J, Chen B, Chen L, et al. The three-dimensional collagen scaffold improves the stemness of rat bone marrow mesenchymal stem cells. J Genet Genom. 2012;39:633–41.
Antebi B, Zhang Z, Wang Y, Lu Z, Chen XD, Ling J. Stromal-cell-derived extracellular matrix promotes the proliferation and retains the osteogenic differentiation capacity of mesenchymal stem cells on three-dimensional scaffolds. Tissue Eng Part C Methods. 2015;21:171–81.
Green JA, Yamada KM. Three-dimensional microenvironments modulate fibroblast signaling responses. Adv Drug Deliv Rev. 2007;59:1293–8.
Yamada KM, Cukierman E. Modeling tissue morphogenesis and cancer in 3D. Cell. 2007;130:601–10.
Opiela J, Lipinski D, Romanek J, Juzwa W, Bochenek M, Wilczek P. MMP-2, TIMP-2, TAZ and MEF2a transcript expression in osteogenic and adipogenic differentiation of porcine mesenchymal stem cells. Ann Anim Sci. 2016;16:369–85.
Bosch P, Pratt SL, Stice SL. Isolation, characterization, gene modification, and nuclear reprogramming of porcine mesenchymal stem cells. Biol Reprod. 2006;74:46–57.
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–7.
Schouten CC, Hartman EH, Spauwen PH, Jansen JA. DBM induced ectopic bone formation in the rat: the importance of surface area. J Mater Sci Mater Med. 2005;16:149–52.
Yamaguchi Y, Ohno J, Sato A, Kido H, Fukushima T. Mesenchymal stem cell spheroids exhibit enhanced in-vitro and in-vivo osteoregenerative potential. BMC Biotechnol. 2014;14:105.
Bueno EM, Glowacki J. Cell-free and cell-based approaches for bone regeneration. Nat Rev Rheumatol. 2009;5:685–97.
Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–84.
Gruskin E, Doll BA, Futrell FW, Schmitz JP, Hollinger JO. Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev. 2012;64:1063–77.
Dinopoulos HT, Giannoudis PV. Safety and efficacy of use of demineralised bone matrix in orthopaedic and trauma surgery. Expert Opin Drug Saf. 2006;5:847–66.
Zhao Y, Lin H, Zhang J, Chen B, Sun W, Wang X, et al. Crosslinked three-dimensional demineralized bone matrix for the adipose-derived stromal cell proliferation and differentiation. Tissue Eng Part A. 2009;15:13–21.
Katz JM, Nataraj C, Jaw R, Deigl E, Bursac P. Demineralized bone matrix as an osteoinductive biomaterial and in vitro predictors of its biological potential. J Biomed Mater Res B Appl Biomater. 2009;89:127–34.
Mosekilde L, Kragstrup J, Richards A. Compressive strength, ash weight, and volume of vertebral trabecular bone in experimental fluorosis in pigs. Calcif Tissue Int. 1987;40:318–22.
Aerssens J, Boonen S, Lowet G, Dequeker J. Interspecies differences in bone composition, density, and quality: potential implications for in vivo bone research. Endocrinology. 1998;139:663–70.
Thorwarth M, Schultze-Mosgau S, Kessler P, Wiltfang J, Schlegel KA. Bone regeneration in osseous defects using a resorbable nanoparticular hydroxyapatite. J Oral Maxillofac Surg. 2005;63:1626–33.
Acknowledgements
This work was supported by National Key R&D Program of China (2016YFC1000800, 2016YFC1000806, 2016YFC1101500, 2017YFA0104700), National Research Institute for Family Planning (2016GJZ06), the National Science Foundation of China (81601084), Strategic Priority Research Program of the Chinese Academy of Sciences (XDA04020000). We thank Rebecca Jackson, PhD, from Liwen Bianji, Edanz Group China (www.liwenbianji.cn/ac), for editing the English text of a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Cui, Y., Lu, C., Chen, B. et al. Restoration of mandibular bone defects with demineralized bone matrix combined with three-dimensional cultured bone marrow-derived mesenchymal stem cells in minipig models. J Mater Sci: Mater Med 29, 147 (2018). https://doi.org/10.1007/s10856-018-6152-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-018-6152-3