Abstract
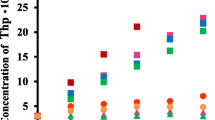
The inclusion complexations of paracyclophane (CP44) and β-cyclodextrin (β-CD) with several anilinonaphthalene sulfonic acids (ANSs) have been characterized via the enhancement of fluorescence spectra upon inclusion. β-CD and CP44 formed 1:1 inclusion complexes with ANSs, and the high stability of inclusion complexes of the latter was demonstrated. The thermodynamic parameters observed from the temperature dependence of the inclusion constants (van’t Hoff analysis) showed that CP44 inclusion complexations with ANSs are enthalpy-driven. Furthermore, the structure of the inclusion complexes was discussed based on 1H- and 2D ROESY-NMR measurements. It was found that CP44 encapsulates the naphthalene moiety of ANSs. On the other hand, differences in the structures of the β-CD inclusion complexes were observed for the 4-OH-substituted ANS. The molecular recognition for the inclusion of β-CD was found to be sensitive compared with that of CP44. In addition, the local polarity inside the CP44 and β-CD cavities was evaluated using ANSs as the fluorescence probe. Based on these results, we have suggested that the local polarity of ANSs in the hydrophobic cavities of CP44 and β-CD plays an important role in quantum yield enhancement upon inclusion.




Similar content being viewed by others
References
Bender, M.L., Komiyama, M.: Cyclodextrin chemistry. Springer, New York (1978)
Junquera, E., Penal, C., Aicart, A.: A conductimetric study of the interaction of β-cyclodextrin or hydroxypropyl-β-cyclodextrin with dodecyltrimethylammonium bromide in water solution. Langmuir 11, 4685–4690 (1995)
Gelb, R.I., Schwartz, L.M., Murray, C.T., Laufer, D.A.: Complexation of 4-biphnyl- carboxylate by cyclohexaamylose. A conductometric and 13C nuclear magnetic resonance spectrometric and analysis. J. Am. Chem. Soc. 100, 3553–3559 (1978)
Odashima, K., Itai, A., Iitaka, Y., Arata, Y.: Inclusion complex formation in a particular geometry by a water-soluble paracyclophane in aqueous solution. Tetrahedron Lett. 21, 4347–4350 (1980)
Odashima, K., Koga, K.: Design and syntheses of paracyclophanes having charged side chains that are soluble in neutral aqueous solution. Heterocycles 15, 1151–1154 (1981)
Berville, M., Karmazin, L., Wytko, J.A., Weiss, J.: Viologen cyclophanes: redox controlled host-guest interactions. Chem. Commun. 51, 15772–15775 (2015)
Dale, E.J., Vermeulen, N.A., Juricek, M., Barnes, J.C., Young, R.M., Wasielewski, M.R., Stoddart, J.F.: Supramolecular explorations: exhibiting the extent of extended cationic cyclophanes. Acc. Chem. Res. 49, 262–273 (2016)
Hayashida, O., Kojima, M., Kusano, S.: Biotinylated cyclophane: synthesis, cyclophane-avidin conjugates, and their enhanced guest-binding affinity. J. Org. Chem. 80, 9722–9727 (2015)
Hayashida, O., Eguchi, C., Kimura, K., Oyama, Y., Nakashima, T., Shioji, K.: Guest binding, cellular uptake, and molecular delivery of water-soluble cyclophanes having a pyrene moiety. Chem. Lett. 39, 1321–1322 (2010)
Kosower, E.M., Dodiuk, H.: Intramolecular donor-acceptor systems. 3. A third type of emitting singlet state for N-alkyl-6-N-arylamino-2-naphthalenesulfonates. Solvent modulation of substituent effects on charge-transfer emissions. J. Am. Chem. Soc. 100, 4173–4179 (1978)
Wagner, B.D., Fitzpatrick, S.J.: A comparison of the host-guest inclusion complexes of 1,8-ANS and 2,6-ANS in parent and modified cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 38, 467–478 (2000)
Sueishi, Y., Fujita, T., Nakatani, S., Inazumi, N., Osawa, Y.: The enhancement of fluorescence quantum yields of anilinonaphthalene sulfonic acids by inclusion of various cyclodextrins and cucurbit[7]uril. Spectroc. Acta A 114, 344–349 (2013)
Odashima, K., Itai, A., Iitaka, Y., Koga, K.: Host-guest complex formation between a water-soluble polyparacyclophene and a hydrophobic guest molecule. J. Am. Chem. Soc. 102, 2501–2502 (1980)
Bucherer, H., Stohmann, A.: Aryl-substituted β-naphthylamines and their preparation by the sulphite method. Zeitschrift fuer Farben- und Textil-Chemie 3, 57–62 and 77–81 (1904)
Velapoldi, R.A., Tonnesen, H.H.: Corrected emission spectra and quantum yields for a series of fluorescent compounds in the visible spectra region. J. Fluoresc. 14, 465–472 (2004)
Job, P.: Formation and stability of inorganic complexes in solution. Ann. Chim. 9, 113–203 (1928)
Scott, R.L.: Some comments on the Benesi-Hildebrand equation. Recueil des Travaux Chimiques des Pays-Bas et de la Belgique 75, 787–789 (1956)
Sueishi, Y., Ohtani, K., Nishimura, N.: The thermal cis-to-trans isomerization of N,N′-diacylindigos. Kinetic pressure, solvent, and substituent effects. Bull. Chem. Soc. Jpn. 58, 810–814 (1985)
Leffler, J.E.: The interpretation of enthalpy and entropy data. J. Org. Chem. 31, 533–537 (1966)
Castronuovo, G., Niccoli, M., Varriale, L.: Complexation forces in aqueous solution. Calorimetric studies of the association of 2-hydroxypropyl-β-cyclodextrin with monocarboxylic acids or cycloalkanols. Tetrahedron 63, 7047–7052 (2007)
Castronuovo, G., Niccoli, M.: Solvent effects on the complexation of 1-alkanols by parent and modified cyclodextrins. Calorimetric studies at 298 K. J. Therm. Anal. Calorim. 103, 641–646 (2011)
Kosower, E.M., Dodiuk, H., Tanizawa, K., Ottolenghi, M., Orbach, N.: Intramolecular donor-acceptor systems. Radiative and nonradiative processes for the excited states of 2-N-arylamino-6-naphthalenesulfonates. J. Am. Chem. Soc. 97, 2167–2178 (1975)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sueishi, Y., Itoh, A., Inazumi, N. et al. A comparative study on inclusion complexation of substituted anilinonaphthalene sulfonic acids with 1,6,20,25-tetraaza[6.1.6.1]-paracyclophane and β-cyclodextrin. J Incl Phenom Macrocycl Chem 91, 1–7 (2018). https://doi.org/10.1007/s10847-018-0790-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-018-0790-4