Abstract
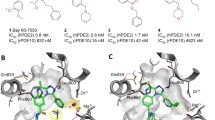
Bacterial β-ketoacyl-acyl carrier protein synthase III (FabH) has become an attractive target for the development of new antibacterial agents which can overcome the increased resistance of these pathogens to antibiotics in clinical use. Despite several efforts have been dedicated to find inhibitors for this enzyme, it is not a straightforward task, mainly due its high flexibility which makes difficult the structure-based design of FabH inhibitors. Here, we present for the first time a Molecular Dynamics (MD) study of the E. colil FabH enzyme to explore its conformational space. We compare the flexibility of this enzyme for the unliganded protein and an enzyme-inhibitor complex and find a correspondence between our modeling results and the experimental evidence previously reported for this enzyme. Furthermore, through a 100 ns MD simulation of the unliganded enzyme we extract useful information related to the concerted motions that take place along the principal components of displacement. We also establish a relation between the presence of water molecules in the oxyanion hole with the conformational stability of structural important loops. Representative conformations of the binding pocket along the whole trajectory of the unliganded protein are selected through cluster analysis and we find that they contain a conformational diversity which is not provided by the X-ray structures of ecFabH. As a proof of this last hypothesis, we use a set of 10 FabH inhibitors and show that they cannot be correctly modeled in any available X-ray structure, while by using our set of conformations extracted from the MD simulations, this task can be accomplish. Finally, we show the ability of short MD simulations for the refinement of the docking binding poses and for MM-PBSA calculations to predict stable protein-inhibitor complexes in this enzyme.















Similar content being viewed by others
References
Monaghan RL, Barrett JF (2006) Antibacterial drug discovery–then, now and the genomics future. Biochem Pharmacol 71(7):901–909
Levy SB, Marshall B (2004) Antibacterial resistance worldwide: causes, challenges and responses. Nat Med 10(12 Suppl):S122–S129
Nikaido H (2009) Multidrug resistance in bacteria. Annu Rev Biochem 78(1):119–146
Livermore DM (2004) The need for new antibiotics. Clin Microbiol Infect 10(s4):1–9
Pucci MJ (2006) Use of genomics to select antibacterial targets. Biochem Pharmacol 71(7):1066–1072
Conly J, Johnston B (2005) Where are all the new antibiotics? The new antibiotic paradox. Can J Infect Dis Med Microbiol 16(3):159–160
Barbachyn MR, Ford CW (2003) Oxazolidinone structure-activity relationships leading to linezolid. Angew Chem Int Ed Engl 42(18):2010–2023
Fischbach MA, Walsh CT (2009) Antibiotics for emerging pathogens. Science 325(5944):1089–1093
Kern WV (2006) Daptomycin: first in a new class of antibiotics for complicated skin and soft-tissue infections. Int J Clin Pract 60(3):370–378
Heath RJ, Rock CO (2004) Fatty acid biosynthesis as a target for novel antibacterial. Curr Opin Investig Drugs 5(2):146–153
Jayakumar A, Tai MH, Huang WY, al-Feel W, Hsu M, Abu-Elheiga L, Chirala SS, Wakil SJ (1995) Human fatty acid synthase: properties and molecular cloning. Proc Natl Acad Sci USA 92(19):8695–8699
Jayakumar A, Huang WY, Raetz B, Chirala SS, Wakil SJ (1996) Cloning and expression of the multifunctional human fatty acid synthase and its subdomains in Escherichia coli. Proc Natl Acad Sci USA 93(25):14509–14514
Wright HT, Reynolds KA (2007) Antibacterial targets in fatty acid biosynthesis. Curr Opin Microbiol 10(5):447–453
Tsay JT, Oh W, Larson TJ, Jackowski S, Rock CO (1992) Isolation and characterization of the beta-ketoacyl-acyl carrier protein synthase III gene (fabH) from Escherichia coli K-12. J Biol Chem 267(10):6807–6814
Heath RJ, Rock CO (1996) Regulation of fatty acid elongation and initiation by acyl-acyl carrier protein in Escherichia coli. J Biol Chem 271(4):1833–1836
Lai CY, Cronan JE (2003) Beta-ketoacyl-acyl carrier protein synthase III (FabH) is essential for bacterial fatty acid synthesis. J Biol Chem 278(51):51494–51503
Revill WP, Bibb MJ, Scheu AK, Kieser HJ, Hopwood DA (2001) Beta-ketoacyl acyl carrier protein synthase III (FabH) is essential for fatty acid biosynthesis in Streptomyces coelicolor A3(2). J Bacteriol 183(11):3526–3530
Khandekar SS, Gentry DR, Van Aller GS, Warren P, Xiang H, Silverman C, Doyle ML, Chambers PA, Konstantinidis AK, Brandt M, Daines RA, Lonsdale JT (2001) Identification, substrate specificity, and inhibition of the Streptococcus pneumoniae beta-ketoacyl-acyl carrier protein synthase III (FabH). J Biol Chem 276(32):30024–30030
Fleischmann RD, Adams MD, White O, Clayton RA, Kirkness EF, Kerlavage AR, Bult CJ, Tomb JF, Dougherty BA, Merrick JM et al (1995) Whole-genome random sequencing and assembly of Haemophilus influenzae Rd. Science 269(5223):496–512
Cole ST, Brosch R, Parkhill J, Garnier T, Churcher C, Harris D, Gordon SV, Eiglmeier K, Gas S, Barry CE 3rd, Tekaia F, Badcock K, Basham D, Brown D, Chillingworth T, Connor R, Davies R, Devlin K, Feltwell T, Gentles S, Hamlin N, Holroyd S, Hornsby T, Jagels K, Krogh A, McLean J, Moule S, Murphy L, Oliver K, Osborne J, Quail MA, Rajandream MA, Rogers J, Rutter S, Seeger K, Skelton J, Squares R, Squares S, Sulston JE, Taylor K, Whitehead S, Barrell BG (1998) Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393(6685):537–544
Kuroda M, Ohta T, Uchiyama I, Baba T, Yuzawa H, Kobayashi I, Cui L, Oguchi A, Aoki K, Nagai Y, Lian J, Ito T, Kanamori M, Matsumaru H, Maruyama A, Murakami H, Hosoyama A, Mizutani-Ui Y, Takahashi NK, Sawano T, Inoue R, Kaito C, Sekimizu K, Hirakawa H, Kuhara S, Goto S, Yabuzaki J, Kanehisa M, Yamashita A, Oshima K, Furuya K, Yoshino C, Shiba T, Hattori M, Ogasawara N, Hayashi H, Hiramatsu K (2001) Whole genome sequencing of meticillin-resistant Staphylococcus aureus. Lancet 357(9264):1225–1240
Stover CK, Pham XQ, Erwin AL, Mizoguchi SD, Warrener P, Hickey MJ, Brinkman FS, Hufnagle WO, Kowalik DJ, Lagrou M, Garber RL, Goltry L, Tolentino E, Westbrock-Wadman S, Yuan Y, Brody LL, Coulter SN, Folger KR, Kas A, Larbig K, Lim R, Smith K, Spencer D, Wong GK, Wu Z, Paulsen IT, Reizer J, Saier MH, Hancock RE, Lory S, Olson MV (2000) Complete genome sequence of Pseudomonas aeruginosa PA01, an opportunistic pathogen. Nature 406(6799):959–964
Heath RJ, Rock CO (1996) Inhibition of beta-ketoacyl-acyl carrier protein synthase III (FabH) by acyl-acyl carrier protein in Escherichia coli. J Biol Chem 271(18):10996–11000
Alhamadsheh MM, Waters NC, Sachdeva S, Lee P, Reynolds KA (2008) Synthesis and biological evaluation of novel sulfonyl-naphthalene-1, 4-diols as FabH inhibitors. Bioorg Med Chem Lett 18(24):6402–6405
Gajiwala KS, Margosiak S, Lu J, Cortez J, Su Y, Nie Z, Appelt K (2009) Crystal structures of bacterial FabH suggest a molecular basis for the substrate specificity of the enzyme. FEBS Lett 583(17):2939–2946
He X, Reeve AM, Desai UR, Kellogg GE, Reynolds KA (2004) 1, 2-dithiole-3-ones as potent inhibitors of the bacterial 3-ketoacyl acyl carrier protein synthase III (FabH). Antimicrob Agents Chemother 48(8):3093–3102
Li HQ, Luo Y, Lv PC, Shi L, Liu CH, Zhu HL (2010) Design and synthesis of novel deoxybenzoin derivatives as FabH inhibitors and anti-inflammatory agents. Bioorg Med Chem Lett 20(6):2025–2028
Li HQ, Shi L, Li QS, Liu PG, Luo Y, Zhao J, Zhu HL (2009) Synthesis of C(7) modified chrysin derivatives designing to inhibit beta-ketoacyl-acyl carrier protein synthase III (FabH) as antibiotics. Bioorg Med Chem 17(17):6264–6269
LV PC, Wang KR, Yang Y, Mao WJ, Chen J, Xiong J, Zhu HL (2009) Design, synthesis and biological evaluation of novel thiazole derivatives as potent FabH inhibitors. Bioorg Med Chem Lett 19(23):6750–6754
Senior SJ, Illarionov PA, Gurcha SS, Campbell IB, Schaeffer ML, Minnikin DE, Besra GS (2004) Acetylene-based analogues of thiolactomycin, active against Mycobacterium tuberculosis mtFabH fatty acid condensing enzyme. Bioorg Med Chem Lett 14(2):373–376
Senior SJ, Illarionov PA, Gurcha SS, Campbell IB, Schaeffer ML, Minnikin DE, Besra GS (2003) Biphenyl-based analogues of thiolactomycin, active against Mycobacterium tuberculosis mtFabH fatty acid condensing enzyme. Bioorg Med Chem Lett 13(21):3685–3688
Nie Z, Perretta C, Lu J, Su Y, Margosiak S, Gajiwala KS, Cortez J, Nikulin V, Yager KM, Appelt K, Chu S (2005) Structure-based design, synthesis, and study of potent inhibitors of beta-ketoacyl-acyl carrier protein synthase III as potential antimicrobial agents. J Med Chem 48(5):1596–1609
Alhamadsheh MM, Waters NC, Huddler DP, Kreishman-Deitrick M, Florova G, Reynolds KA (2007) Synthesis and biological evaluation of thiazolidine-2-one 1, 1-dioxide as inhibitors of Escherichia coli beta-ketoacyl-ACP-synthase III (FabH). Bioorg Med Chem Lett 17(4):879–883
He X, Reynolds KA (2002) Purification, characterization, and identification of novel inhibitors of the beta-ketoacyl-acyl carrier protein synthase III (FabH) from Staphylococcus aureus. Antimicrob Agents Chemother 46(5):1310–1318
Daines RA, Pendrak I, Sham K, Van Aller GS, Konstantinidis AK, Lonsdale JT, Janson CA, Qiu X, Brandt M, Khandekar SS, Silverman C, Head MS (2003) First X-ray cocrystal structure of a bacterial FabH condensing enzyme and a small molecule inhibitor achieved using rational design and homology modeling. J Med Chem 46(1):5–8
Perez-Castillo Y, Perez MA (2008) Bacterial beta-ketoacyl-acyl carrier protein synthase III (FabH): an attractive target for the design of new broad-spectrum antimicrobial agents. Mini Rev Med Chem 8(1):36–45
Cornell W, Nam K (2009) Steroid hormone binding receptors: application of homology modeling, induced fit docking, and molecular dynamics to study structure-function relationships. Curr Top Med Chem 9(9):844–853
Rosales-Hernandez MC, Bermudez-Lugo J, Garcia J, Trujillo-Ferrara J, Correa-Basurto J (2009) Molecular modeling applied to anti-cancer drug development. Anticancer Agents Med Chem 9(2):230–238
Alonso H, Bliznyuk AA, Gready JE (2006) Combining docking and molecular dynamic simulations in drug design. Med Res Rev 26(5):531–568
Talele TT, Khedkar SA, Rigby AC (2009) Successful applications of computer aided drug discovery: moving drugs from concept to the clinic. Curr Top Med Chem 10(1):127–141
Qiu X, Janson CA, Smith WW, Head M, Lonsdale J, Konstantinidis AK (2001) Refined structures of beta-ketoacyl-acyl carrier protein synthase III. J Mol Biol 307(1):341–356
Scarsdale JN, Kazanina G, He X, Reynolds KA, Wright HT (2001) Crystal structure of the Mycobacterium tuberculosis beta-ketoacyl-acyl carrier protein synthase III. J Biol Chem 276(23):20516–20522
Qiu X, Janson CA, Konstantinidis AK, Nwagwu S, Silverman C, Smith WW, Khandekar S, Lonsdale J, Abdel-Meguid SS (1999) Crystal structure of beta-ketoacyl-acyl carrier protein synthase III. A key condensing enzyme in bacterial fatty acid biosynthesis. J Biol Chem 274(51):36465–36471
Alhamadsheh MM, Musayev F, Komissarov AA, Sachdeva S, Wright HT, Scarsdale N, Florova G, Reynolds KA (2007) Alkyl-CoA disulfides as inhibitors and mechanistic probes for FabH enzymes. Chem Biol 14(5):513–524
Musayev F, Sachdeva S, Scarsdale JN, Reynolds KA, Wright HT (2005) Crystal structure of a substrate complex of Mycobacterium tuberculosis beta-ketoacyl-acyl carrier protein synthase III (FabH) with lauroyl-coenzyme A. J Mol Biol 346(5):1313–1321
Qiu X, Choudhry AE, Janson CA, Grooms M, Daines RA, Lonsdale JT, Khandekar SS (2005) Crystal structure and substrate specificity of the beta-ketoacyl-acyl carrier protein synthase III (FabH) from Staphylococcus aureus. Protein Sci 14(8):2087–2094
Sachdeva S, Musayev FN, Alhamadsheh MM, Scarsdale JN, Wright HT, Reynolds KA (2008) Separate entrance and exit portals for ligand traffic in Mycobacterium tuberculosis FabH. Chem Biol 15(4):402–412
Marco E, Gago F (2007) Overcoming the inadequacies or limitations of experimental structures as drug targets by using computational modeling tools and molecular dynamics simulations. Chem Med Chem 2(10):1388–1401
Durrant JD, de Oliveira CA, McCammon JA (2010) Including receptor flexibility and induced fit effects into the design of MMP-2 inhibitors. J Mol Recognit 23(2):173–182
Liu M, Yuan M, Luo M, Bu X, Luo HB, Hu X (2010) Binding of curcumin with glyoxalase I: molecular docking, molecular dynamics simulations, and kinetics analysis. Biophys Chem 147(1–2):28–34
Case DA, Darden TA, Cheatham III TE, Simmerling CL, Wang J, Duke RE, Luo R, Crowley M, Walker RC, Zhang W, Merz KM, Wang B, Hayik S, Roitberg A, Seabra G, Kolossváry I, Wong KF, Paesani F, Vanicek J, Wu X, Brozell SR, Steinbrecher T, Gohlke H, Yang L, Tan C, Mongan J, Hornak V, Cui G, Mathews DH, Seetin MG, Sagui C, Babin V, Kollman PA (2008) AMBER 10. University of California, San Francisco
Mongan J (2004) J Comput Aided Mol Des 18(6):433–436
Humphrey W, Dalke A, Schulten K (1996) VMD–visual molecular dynamics. J Mol Graph 14:33–38
MATLAB (2009) R2009a edn. The MathWorks Inc
OpenEye Scientific Software I (2008) OMEGA, 2.3.2 edn. OpenEye Scientific Software, Inc., Santa Fe
OpenEye Scientific Software I (2008) QUACPAC, 1.3.1 edn. OpenEye Scientific Software, Inc., Santa Fe
Lang PT, Brozell SR, Mukherjee S, Pettersen EF, Meng EC, Thomas V, Rizzo RC, Case DA, James TL, Kuntz ID (2009) DOCK 6: combining techniques to model RNA-small molecule complexes. RNA 15(6):1219–1230
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) UCSF Chimera–a visualization system for exploratory research and analysis. J Comput Chem 25(13):1605–1612
Case DA, Cheatham TE III, Darden T, Gohlke H, Luo R, Merz KM Jr, Onufriev A, Simmerling C, Wang B, Woods RJ (2005) The Amber biomolecular simulation programs. J Comput Chem 26(16):1668–1688
Feig M, Im W, Brooks CL 3rd (2004) Implicit solvation based on generalized Born theory in different dielectric environments. J Chem Phys 120(2):903–911
Onufriev A, Case DA, Bashford D (2002) Effective born radii in the generalized born approximation: the importance of being perfect. J Comput Chem 23(14):1297–1304
Li L, Uversky VN, Dunker AK, Meroueh SO (2007) A computational investigation of allostery in the catabolite activator protein. J Am Chem Soc 129(50):15668–15676
Lange OF, Grubmuller H (2006) J Phys Chem B 110(45):22842
Teotico DG, Frazier ML, Ding F, Dokholyan NV, Temple BRS, Redinbo MR (2008) Active nuclear receptors exhibit highly correlated AF-2 domain motions. PLoS Comput Biol 4(7):e1000111
Bradley MJ, Chivers PT, Baker NA (2008) Molecular dynamics simulation of the Escherichia coli NikR protein: equilibrium conformational fluctuations reveal interdomain allosteric communication pathways. J Mol Biol 378(5):1155–1173
Davies C, Heath RJ, White SW, Rock CO (2000) The 1.8 A crystal structure and active-site architecture of beta-ketoacyl-acyl carrier protein synthase III (FabH) from Escherichia coli. Structure 8(2):185–195
Shao J, Tanner SW, Thompson N, Cheatham TE (2007) Clustering molecular dynamics trajectories: 1. characterizing the performance of different clustering algorithms. J Chem Theory Comput 3(6):2312–2334
Mitchell JB (2001) The relationship between the sequence identities of alpha helical proteins in the PDB and the molecular similarities of their ligands. J Chem Inf Comput Sci 41(6):1617–1622
Nobeli I, Spriggs RV, George RA, Thornton JM (2005) A ligand-centric analysis of the diversity and evolution of protein-ligand relationships in E. coli. J Mol Biol 347(2):415–436
Kalinina O, Gelfand M, Russell R (2009) Combining specificity determining and conserved residues improves functional site prediction. BMC Bioinformatics 10(1):174
Acknowledgments
We thank the Vlaams Supercomputer Centrum (VSC) for providing access to the computational resources needed for accomplishing all calculations. Pérez-Castillo Y. thanks the Flemish Interuniversity Council (VLIR) for financial support through the project: “Strengthening research and PhD formation in Computer Sciences and its applications” in the framework of the VLIR-UCLV collaborative program. Cabrera-Pérez M. A. and Pérez-Castillo Y. thank the Spanish Agency of International Cooperation for the Development (AECID) for financial support through the project “Montaje de un laboratorio de química computacional, con fines académicos y científicos, para el diseño racional de nuevos candidatos a fármacos en enfermedades de alto impacto social” (D/024153/09).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pérez-Castillo, Y., Froeyen, M., Cabrera-Pérez, M.Á. et al. Molecular dynamics and docking simulations as a proof of high flexibility in E. coli FabH and its relevance for accurate inhibitor modeling. J Comput Aided Mol Des 25, 371–393 (2011). https://doi.org/10.1007/s10822-011-9427-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-011-9427-z