Abstract
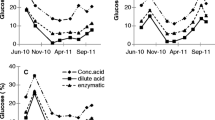
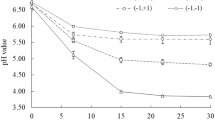
Cultivated kelps (brown algae) are a sustainable biomass resource and a potential feedstock for conversion to biofuels and chemicals. Due to seasonal variations, and a short period with optimal biomass composition, preservation and storage of the biomass is required for a year-round operation of a seaweed processing plant. For use of the biomass as a carbon source for fermentation to biofuels, only low-cost preservation methods are feasible. Preservation of Saccharina latissima (sugar kelp) by sulphuric and formic acid has been evaluated as a method to maintain the fermentable carbohydrates laminaran and mannitol. In milled biomass, stored anaerobically for up to 6.5 months at different pH values, laminaran and mannitol were efficiently preserved in samples stored at pH below 3.7, obtained by addition of sulphuric acid. When a combination of sulphuric and formic acid was used, no sugar loss could be detected up to pH 4. The content of free glucose increased during the storage period in the well-preserved samples without loss of sugars. The free glucose levels were highest at the highest storage pH, providing strong evidence for the presence of endogenous β-glucanases that hydrolyse laminaran to glucose. Our work was primarily aimed at preservation of the biomass for application as a carbon source for fermentation. However, the method will be equally suited for other applications of the biomass, such as extraction of valuable compounds for use in functional food, feed or other areas.






Similar content being viewed by others
References
Black W (1955) The preservation of seaweed by ensiling and bactericides. J Sci Food Agric 6:14–23
Førde H, Forbord S, Handå A, Fossberg J, Arff J, Johnsen G, Reitan KI (2016) Development of bryozoan fouling on cultivated kelp (Saccharina latissima) in Norway. J Appl Phycol 28:1225–1234
Haug A (1961) Dissociation of alginic acid. Acta Chem Scand 15:950–952
Herrmann C, Fitz Gerald J, O’Shea R, Xia A, O’Kiely P, Murphy JD (2015) Ensiling of seaweed for a seaweed biofuel industry. Bioresour Technol 196:301–313
Horn S, Aasen I, Østgaard K (2000) Ethanol production from seaweed extract. J Ind Microbiol Biotechnol 25:249–254
Hou X, Hansen JH, Bjerre A-B (2015) Integrated bioethanol and protein production from brown seaweed Laminaria digitata. Bioresour Technol 197:310–317
Kennedy SJ (1990) Comparison of the fermentation quality and nutritive value of sulphuric and formic acid-treated silages fed to beef cattle. Grass Forage Sci 45:17–28
Kraan S (2013) Mass-cultivation of carbohydrate rich macroalgae, a possible solution for sustainable biofuel production. Mitig Adapt Strat Glob Chang 18:27–46
Marquez GPB, Santiañez WJE, Trono GC, de la Rama SRB, Takeuchi H, Hasegawa T (2015) Seaweeds: a sustainable fuel source. In: Tiwari B, Troy D (eds) Seaweed sustainability: food and non-food applications, Elsevier, Amsterdam, pp 421–458
Mayne C (1993) The effect of formic acid, sulphuric acid and a bacterial inoculant on silage fermentation and the food intake and milk production of lactating dairy cows. Anim Sci 56:29–42
McFeeters RF, Chen K-H (1986) Utilization of electron acceptors for anaerobic mannitol metabolism by Lactobacillus plantarum. Compounds which serve as electron acceptors. Food Microbiol 3:73–81
Milledge JJ, Harvey PJ (2016a) Ensilage and anaerobic digestion of Sargassum muticum. J Appl Phycol 28:3021–3030
Milledge JJ, Harvey PJ (2016b) Potential process ‘hurdles’ in the use of macroalgae as feedstock for biofuel production in the British Isles. J Chem Technol Biotechnol 91:2221–2234
Redden H, Milledge JJ, Greenwell HC, Dyer PW, Harvey PJ (2016) Changes in higher heating value and ash content of seaweed during ensiling. J Appl Phycol 29:1037–1046
Schiener P, Black KD, Stanley MS, Green DH (2015) The seasonal variation in the chemical composition of the kelp species Laminaria digitata, Laminaria hyperborea, Saccharina latissima and Alaria esculenta. J Appl Phycol 27:363–373
Schiener P, Stanley MS, Black KD, Green DH (2016) Assessment of saccharification and fermentation of brown seaweeds to identify the seasonal effect on bioethanol production. J Appl Phycol 28:3009–3020
Stévant P, Marfaing H, Duinker A, Fleurence J, Rustad T, Sandbakken I, Chapman A (2017a) Biomass soaking treatments to reduce potentially undesirable compounds in the edible seaweeds sugar kelp (Saccharina latissima) and winged kelp (Alaria esculenta) and health risk estimation for human consumption. J Appl Phycol. https://doi.org/10.1007/s10811-017-1343-8
Stévant P, Rebours C, Chapman A (2017b) Seaweed aquaculture in Norway: recent industrial developments and future perspectives. Aquac Int 25:1373–1390
Theron MM, Rykers Lues JF (2010) Organic acids and food preservation. CRC Press, Boca Raton, Florida
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 105 kb)
Rights and permissions
About this article
Cite this article
Sandbakken, I.S., Sæther, M., Funderud, J. et al. Acid preservation of Saccharina latissima for application as a carbon source for fermentation to biofuels and chemicals. J Appl Phycol 30, 3581–3588 (2018). https://doi.org/10.1007/s10811-018-1489-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-018-1489-z