Abstract
Since Hans Asperger’s first description (Arch Psych Nervenkrankh 117:76–136, 1944), through Lorna Wing’s translation and definition (Psychol Med 11:115–129, 1981), to its introduction in the fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM, 1994), Asperger Syndrome has always aroused huge interest and debate, until vanishing in the DSM fifth edition (2013). The debate regarded its diagnostic validity and its differentiation from high functioning autism (HFA). The present study aimed to examine whether AS differed from HFA in clinical profiles and to analyze the impact of DSM-5’s innovation. Differences in cognitive, language, school functioning and comorbidities, were revealed when 80 AS and 70 HFA patients (3–18 years) were compared. Results suggested that an AS empirical distinction within autism spectrum disorder should be clinically useful.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A Concise History of Asperger Syndrome
In the same years of Leo Kanner’s description of Infantile Autism (Kanner 1943), Hans Asperger wrote a case report on Autistic Psychopathy (Asperger 1944), describing children with social-communication impairment, eccentric manners, unusual interests and cognitive domains of hyper-functioning. The American Psychiatric Association (APA) did not immediately recognize Autism as a distinct category: it was introduced as Infantile Autism in the third edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-III, APA 1980) and included within Pervasive Developmental Disorders (PDD) in the DSM-III-R (APA 1987). In 1981, Wing (1981) resumed Asperger’s researches, renaming the Autistic Psychopathy as Asperger Syndrome (AS). In 1989, the first diagnostic criteria for AS were proposed (Gillberg and Gillberg 1989; Szatmari et al. 1989) and in the 1990s, AS appeared in the DSM-IV within PDD (APA 1994).
Asperger Syndrome and High Functioning Autism
The diagnosis of AS required at least two symptoms of social interaction impairment and one symptom of behavioral and interest restriction, a normal cognitive functioning and the absence of significant general delay in language. Moreover, diagnostic criteria for Autistic Disorder should not be met (otherwise, Autistic diagnosis should have precedence). This implied a differential diagnosis between AS and Autism, especially the type without cognitive delay, also known as High Functioning Autism (HFA) (Klin et al. 2000, 2005). HFA is not a term used in the DSM, but it is commonly used to identify patients diagnosed with Autistic Disorder (AD) or PDD-Not Other Specified (PDD-NOS), with average or above average intellectual abilities (Intelligent Quotient, IQ, higher than 70). HFA differs from low-functioning autism (IQ lower than 70) in terms of clinical presentation, prognosis and need of support and assistance in daily life. Since AS and HFA are both characterized by a normal cognitive functioning, there has been considerable debate over whether AS and HFA are distinct conditions, suggesting different etiological and neurobiological mechanism, or share a similar underlying neuropsychological functioning and should therefore be regarded as variants of a single disorder (Gilchrist et al. 2001; Howlin 2003; Ghaziuddin and Mountain-Kimchi 2004; Macintosh and Dissanayake 2004; Walker et al. 2004; Koyama et al. 2007; Saulnier and Kin 2007; Bennett et al. 2008; Ghaziuddin 2008; Witwer and Lecavalier 2008; Sanders 2009; Woodbury-Smith and Volkmar 2009; Rinehart et al. 2010; Spek et al. 2010; Speirs et al. 2011; Nayate et al. 2012; Planche and Lemonnier 2012; Sharma et al. 2012; Lai et al. 2013; Tsai and Ghaziuddin 2014; Volkmar and McPartland 2014; Wilson et al. 2014; Barahona-Corréa and Filipe 2016; Montgomery et al. 2016; Prior et al. 1998).
According to Hans Asperger original description, his patients differ from those described by Kanner (Asperger 1944). Instead, Lorna Wing, translating Asperger’s work, named the syndrome and Kanner’s autism both part of an autistic continuum (Wing 1981).
AS definition and in its boundaries with HFA have been the topic of a growing body of literature, published from the 1980s till today, providing contradictory results: most researches have highlighted the significant similarities between AS and HFA (Howlin 2003; Macintosh and Dissanayake 2004; Witwer and Lecavalier 2008; Sanders 2009; Nayate et al. 2012; Wilson et al. 2014), while other authors continued to stress the importance to consider them as different clinical entities (Gilchrist et al. 2001; Ghaziuddin and Mountain-Kimchi 2004; Bennett et al. 2008; Ghaziuddin 2008; Speirs et al. 2011; Planche and Lemonnier 2012; Lai et al. 2013; Tsai and Ghaziuddin 2014; Barahona-Corréa and Filipe 2016; Montgomery et al. 2016).
Although a substantial overlap between AS and autism criteria does exist, a review of 69 studies showed that some fine differences between the two disorders can be recognized in terms of social interaction, motor skills and speech patterns; moreover, all these aspects seem to be relevant for designing clinical and intervention strategies (Sharma et al. 2012).
Tsai and Ghaziuddin (2014) examined 125 comparative studies between AS and HFA (autistic disorder and PDD-NOS); 30 studies concluded that the two conditions were similar, while 95 found quantitative and qualitative differences between them.
Taken together, the main evidences underline more quantitative rather than qualitative differences between AS to HFA, most of them regarding superior linguistic, cognitive and social functioning (Sanders 2009; Volkmar and McPartland 2014).
Recent studies largely focused on biological markers producing controversial results. Some of them found little support for a discrimination between AS and HFA, both in genetic and neuroimaging fields (Barahona-Corréa and Filipe 2016). Other studies underlined qualitative differences in the grey matter distribution between AS and HFA; these findings not only support the hypothesis that the two disorders might have different neurobiological basis, but also the evidence that mixing individuals with autism and AS may sometimes obscure important characteristics of one or the other condition alone (Yu et al. 2011; Bi et al. 2018).
Asperger Syndrome and High Functioning Autism in the Age of DSM-5
Ultimately, the unsolved confusion in defining AS criteria and the clinical overlap between HFA and AS led to its merging into one unifying category, on the assumption that they cannot be reliably differentiated from one another; the latest edition of the DSM, the fifth, incorporated AS into Autistic Spectrum Disorder (ASD), removing the previously discrete diagnostic presentation of PDD (APA 2013). Furthermore, in order to better characterize the diagnosis, specifiers about the presence of cognitive and language impairment and severity levels have been added.
Multiple studies raised concerns about the number of individuals diagnosed with PDD (according to DSM-IV-TR) who may no longer match the new diagnostic criteria of DSM-5. Available literature shows contradictory results on this topic; in fact, some studies indicate that between 50 and 75% of individuals with a prior DSM-IV diagnosis of PDD will maintain the diagnosis, finding the greatest decreases among high functioning population (previous diagnosis of PDD-NOS or AS) (Kulage et al. 2014; Young and Rodi 2014; Smith et al. 2015). Other studies reveal similar percentage of HFA maintaining diagnosis (63%) and higher percentage only for AS (92%), concluding the absence of support for the concerns of losing the diagnosis with the advent of DSM-5 (Kim et al. 2015).
Another debated question is if the term of AS should be mentioned in the future version of the DSM as a “useful” label for a specific group of patients within ASD who differ in terms of clinical features, prognosis and treatment needs.
Aims
The present study re-evaluated and compared subjects diagnosed according to DSM-IV-TR criteria with Asperger Syndrome (henceforth named AS group) and subjects diagnosed with PDD-NOS or AD with an IQ of average or above (henceforth named HFA group), and it aimed to: examine whether subjects with AS versus subjects with HFA differ in clinical profiles; evaluate the effect of the application of DSM-5 criteria and severity levels in terms of concordance between DSM-IV-TR and DSM-5; reflect about the utility and the clinical reliability of the merging subtypes of PDD in the broader diagnostic category of ASD.
Methods
Participants
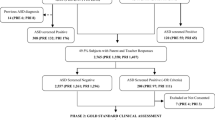
Two groups of patients were sampled for the present study: a group with a diagnosis of AS (n = 80; age range 5–18); a group with HFA (n = 70, age range 3–18, including 7 patients with a diagnosis of Autistic Disorder and 63 patients with a diagnosis of PDD-Not Otherwise Specified).
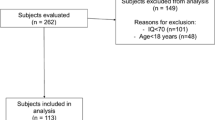
Participants were recruited from a consecutive case series of children and adolescents (aged 3–18) assessed at the Neuropsychiatric Child Unit of the University of Bari, over the course of a 2-year period (2012–2013). Inclusion criteria: a clinical diagnosis of PDD, according to DSM-IV-TR criteria (APA 2000); intelligent quotient (IQ) average or above.
The total number of families invited to participate was 165 and 150 (90.9%) gave informed consent to participate.
Materials
Australian Scale for Asperger’s Syndrome (ASAS) (Attwood 2006) is a questionnaire designed to identify behaviors and abilities indicative of AS in children, divided in five domains (social emotional abilities, communication skills, cognitive skills, specific interests, movement skills). Each domain consists of different questions, ranging from one (if the indicative behavior is rarely present) to six (if the indicative behavior is frequently present). Additionally, ASAS comprises a section of “other characteristics” that investigates the presence/absence of atypical sensitivity, stereotypic movements and language delay. ASAS results do not yield cut-off scores but give important information to be used in a clinical assessment. To provide a detailed account of the clinical profile of the subjects, we have chosen some specific items of ASAS as representative measures of clinical presentation. Responses to each question are in the form of “yes” (positive score) if the behavior is present (ASAS’s score 4–6), or “not” (not scored) if it is not (ASAS’s score 0–3).
Michigan Autism Spectrum Questionnaire (MASQ) (Ghaziuddin and Welch et al. 2013) is a scale designed on the basis of the clinical characteristics of HFA and AS. It focuses on two main areas: quality of social interaction and form/content of communication. It includes 10 questions, each has four responses ranging from 0 to 3, yielding a total score of 30. The highest total scores (> 22) predict AS, the intermediate scores (14 through 21) predict HFA and the lowest scores (< 14) predict other psychiatric disorders.
Intellectual functioning evaluation included the Wechsler Scales (WISC-IV in 69 patients, WISC-III in 49 patients, WPPSI-III in 32 patients); academic achievement of school-aged individuals (n = 117) has been evaluated with a battery of tests validated for the Italian language (Cornoldi and Colpo 1995, 1998; Sartori et al. 1995; Cornoldi et al. 2002, 2010; Cornoldi and Cazzola 2003); comorbidities assessment included the Italian version of the Child Behavior Checklist questionnaire (Achenbach and Edelbrock 1983; Achenbach 1991).
Procedures
In the first step of the study, research team reviewed all the medical records of the participants, obtaining information about diagnoses and previous assessment (intellectual functioning, academic achievement and comorbidities). The research team was composed by one Child Psychologist and five Neuro-Psychiatrists.
In the second step of the study, research team administered to caregivers two rating scales designed to measure specific core features of AS and HFA: the Australian Scale for Asperger’s Syndrome (Attwood 2006) and the Michigan Autism Spectrum Questionnaire (Ghaziuddin and Welch 2013).
In the last step of the study a comparison of the two groups in term of cognitive functioning, core clinical features, school learning abilities and comorbidities was conducted. Moreover, an analysis of the consequences of the application of DSM-5 criteria in terms of concordance and severity levels was performed.
Statistical Analysis
Demographic data, including age and sex, between the two groups, underwent statistical analysis (Table 1).
Quantitative data were summarized as mean and standard deviation. Because quantitative data resulted normally distributed, comparisons between independent groups have been performed with t student test. Multiple evaluations of scales were compared between AS and HFA groups with a multiple testing approach, using a t test and adjusting p values with a permutational false discovery rate (FDR) method.
Analysis of variance for repeated measure was applied to evaluate difference between AS and HFA groups in two score methods (paired measure).
Qualitative variables are summarized as count and percentage, comparisons among two or more group were performed with Chi square test or Fisher exact test, as appropriate. Comparisons of percentage of answers to questionnaires between AS and HFA groups were performed by a multiple testing approach with a permutational FDR adjustment of p values. Difference in percentage and its 95% confidence interval was determined for each single comparison accounting the binomial method.
Concordance between DSM-IV (AS and HFA) and DSM-5 (level 1, level 2, level 3) was evaluated with McNemar test, the strength of the concordance was described by Cohen’s K.
To evaluate the discriminating power of MASQ a ROC curve analysis was performed, reporting area under the curve (AUC) and its 95% confidence interval.
Main comparisons were considered as statistically significant for p value < 0.05. Analysis was performed with SAS 9.4 for PC and R version 3.2.3. The PROC MULTTEST was applied to account for multiple testing, using the permutational FDR approach. ROC curve analysis was conducted with MedCalc Statistical Software version 18 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2018).
Results
Intelligence Quotient (Table 2)
The mean (SD) of FSIQ was 114.1(15.9) in AS and 92.1(14.5) in HFA, showing a statistically significant higher value in the former group (t test = 8.78, raw p value < 0.0001, adjusted p value < 0.0001). Moreover, all the IQ sub-values resulted statistically higher in AS than in HFA (Table 2).
Core Clinical Features, Obtained from ASAS Scores (Table 3)
Among social emotional abilities, there was no significant difference between the two groups, nevertheless HFA appeared more compromised than AS subjects, especially in avoiding social contact.
Among communication skills, over-precise or pedantic speech was significantly more common in AS than HFA subjects (raw p value = 0.0122, adjusted p value = 0.0112); on the contrary literal interpretation was significantly more common in HFA than AS subjects (raw p value = 0.0114, adjusted p value = 0.0431).
Among cognitive skills, there was no significant difference between the two groups, though exceptional long-term memory and fascination by a particular topic was more common in AS group.
No statistically significant difference in clumsiness (both raw and adjusted p value = 1), atypical sensitivity (raw p value = 0.605; p value = 1) and motor mannerism was observed between the two groups.
Delay in acquiring speech was significantly more common in HFA compared to AS group (raw p value = 0.0051, adjusted p value = 0.0234).
MASQ Scores (Fig. 1)
The mean (SD) score was 22.2 (5.1) in AS group and 15.5 (5.9) in HFA group, showing a statistically significant higher value in the former group (t test = 7.42, p value < 0.0001). In the ROC analysis, AUC resulted 0.85 (95% CI 0.72–0.98). The suggested threshold is a score of 18 that provides a sensitivity of 84.6% (95% CI 54.6–98.1) and specificity of 75.7% (95% CI 64–85.2).
School Learning Abilities (Table 4)
Analyzing the subsample of 117 school-aged subjects, learning difficulties were significantly more frequent in HFA than in AS. In particular, the frequency of impairment in reading (adjusted p values: time, p value = 0.015; accuracy, p value = 0.0299; comprehension, p value = 0.0074), mathematics (time, p value = 0.0431; accuracy, p value = 0.0033) and writing (accuracy, p value = 0.0033) was significantly higher in HFA respect to AS group; only the handwriting impairment was not significantly different between the two groups (p value = 0.1754).
Comorbidities (Table 5)
The presence of at least one comorbidity was significantly higher in AS than HFA group (Chi square = 11.14; p value = 0.0013). In both groups, the most frequent comorbidity was ADHD, but comparing AS and HFA, its prevalence did not statistically differ (p value = 0.3992). Depressive and anxiety disorders were significantly more common in AS than in HFA group (Chi square = 4.71; p value = 0.0299; Chi square = 17.87; p value < 0.0001); none of other comorbidities showed a statistically significant difference between the two groups.
Application of DSM-5 Criteria and Severity Levels
When DSM-5 criteria for ASD were applied, these were met by the 97.3% of the total sample 100% of HFA (70/70) and 95% of AS (76/80).
Applying DSM-5 severity levels, 95% of AS subjects (76/80) were classified as Level 1 (the reminder 5% was not classified in ASD); 55.7% of HFA subjects were classified as Level 1 and 44.3% as Level 2. The concordance measured with Cohen’s K was 0.42 (95% CI 0.29–0.54, Mc Nemar statistic = 43, df = 3, p < 0.0001).
Discussion
In DSM-5, the diagnostic label of Asperger’s syndrome (AS) was removed and it was included in a more general category of Autism Spectrum Disorder (ASD). So, its clinical validity, already rather debated, became more ambiguous and its differentiation from High Functioning Autism (HFA) remained unsolved or, at least, equivocal.
Irrespective of the DSM-5 changes, several studies (also published later than APA 2013) continue to debate about AS and its conceptualization within ASD (Chiang et al. 2014; Volkmar and McPartland 2014; Tsai and Ghaziuddin 2014; Wilson et al. 2014; Lai et al. 2015; Helles et al. 2015; Montgomery et al. 2016; Faridi and Khosrowabadi 2017). In this unclear perspective, this study provided a detailed description of clinical profiles of 150 AS/HFA subjects, aged 3–18 years, to examine whether it has been useful to merge them in a unique diagnostic category or if it would be more meaningful to differentiate them in distinct diagnostic categories.
Asperger Syndrome Versus High Functioning Autism
First, in the present study, the results suggest that in both AS and HFA, autistic core features (i.e. deficits in social communication and social interaction and restricted, repetitive patterns of behavior, interests, or activities) are present, even if with subtle differences.
Communication
About the communication features, the present study revealed a significant difference in the presence of a history of delay in language development, prior to 3 years of age, between subjects with AS and subjects with HFA, even if also in AS a mild language delay was reported. In the DSM-IV-TR, the “absence of clinically significant general delay in language (e.g., single words used by age 2 years, communicative phrases used by age 3 years)” was a relevant criterion for AS diagnosis. In contrast with this theoretical construct, extensive literature data showed that language delay could not be considered as a diagnostic exclusion criterion for AS because subtle language problems are expected in AS and also because the recognition of a language delay is not always enough reliable, since it is often dependent on parents recall (Ghaziuddin and Welch 2013). In the DSM-5, the decision to merge the single diagnostic entities in a unique broader category of ASD and the introduction of language impairment as a “specifier” solved this controversy. The present study suggests that even if language delay may not be a differentiating feature of AS/HFA, it can be useful to differentiate developmental language profiles in terms of clinical approach. In the present study, communication skills resulted overall overlapping between AS and HFA, revealing statistically significant differences only in “over-precise or pedantic speech” (characterized by overly formal speech, similar to an in-depth monologue about a topic of special interest, verbose and tangential, lack of the normal prosody), more frequent in AS, and in “literal interpretation of comments”, more common in HFA. The first finding (“over-precise or pedantic speech”) is unsurprising and represents one of the most typical clinical feature of AS, firstly described by Hans Asperger’s original work (1944, in which AS were called “little professors” for their way of talking), then included in the following proposed diagnostic criteria sets (Gillberg and Gillberg 1989; Szatmari et al. 1989) and finally confirmed by subsequent studies (Ghaziuddin and Gerstein 1996; Gilchrist et al. 2001; Macintosh and Dissanayake 2004; Woodbury-Smith and Volkmar 2009). The second finding (“literal interpretation of comments”) may be related to the HFA worse cognitive and comprehension abilities, which made them unable in recognizing the speaker’s communicative intention and in social understanding, unlike patients with AS, who adopted intellectual strategies of compensation.
Motor Functions
Clumsiness has been originally considered a distinctive feature of AS (Ghaziuddin et al. 1994); in our experience, it appeared in both groups, without significant difference, as reported by more recent studies (Fournier et al. 2010; Nayate et al. 2012).
Sensory System
Even if sensory abnormalities were well-represented in our sample, no significant difference between subjects with AS and subjects with HFA was observed. Sensory abnormalities were included in DSM-5 within the second cluster of core symptoms (previously omitted). Sensory overload can be experienced as painful and correlated with lower participation in leisure activities and lower performance at school, therefore considering sensory features in intervention is essential (Grapel et al. 2015).
Intellectual Functioning
In this study, Full Scale Intelligence Quotient mean values were statistically significant higher in AS than HFA. This result is in accordance with previous literature data (Ghaziuddin and Mountain-Kimchi 2004; Macintosh and Dissanayake 2004; Koyama et al. 2007; Ghaziuddin 2008; Witwer and Lecavalier 2008; Noterdaeme et al. 2010; Chiang et al. 2014), although, on this topic, conflicting results were reported. Wilson et al. compared cognitive characteristics of individuals with AS and HFA reporting no significant differences on the cognitive performance (2014). Most reports suggested that patients with AS have a distinct cognitive profile, characterized by a higher verbal IQ and a lower performance IQ, whereas in most cases with HFA, the pattern is reversed (Ghaziuddin and Gerstein 1996; Saulnier and Kin 2007; Volkmar et al. 2009; Noterdaeme et al. 2010; Planche and Lemonnier 2012; Chiang et al. 2014). This profile has been linked to the better verbal abilities of AS patients. Other studies did not confirm the existence of these typical profiles, reporting mixed cognitive patterns that are not consistent enough to enable diagnostic differentiation between the two groups (Ghaziuddin and Mountain-Kimchi 2004; Macintosh and Dissanayake 2004; Ghaziuddin 2008). In the present study, on the basis of the cognitive evaluation, it is not possible to distinguish the two groups: all the IQ sub-values resulted statistically higher in AS than in HFA and differences between verbal and performance sub-values are not statistically significant within the two groups. These results support the hypothesis that the two conditions could be distinguished at a quantitative cognitive level, represented by FSIQ, rather than at a qualitative level, represented by specific cognitive profile. Nevertheless, as for intellectual disability, the presence of very different IQs could denote distinct levels of severity and clinical profile, in the same way, a large discrepancy between AS and HFA intellectual scores could have a qualitative effect on general function.
Academic Achievement
In the present study, another remarkable topic of differentiation between HFA and AS is the prevalence of learning disorders that resulted significantly more frequent in HFA, except to dysgraphia that emerged frequently in both groups, without a significant difference. Low performances in hand-writing, in terms of forming letters or using the paper space were noted in the original description of AS (Asperger 1944) and in recent literature (Jansiewicz et al. 2006; Fuentes et al. 2009). To our knowledge, until today, academic functioning and education needs in HFA have still not been sufficiently explored in literature. In accordance with our findings, Grimm et al. (2015) reported schooling of 45 patients exhibiting ASD without mental retardation, finding that patients with childhood autism and atypical autism respect to those with AS were more likely to receive the support of a special educational assistant and underwent a higher number of consultations and treatment episodes.
Comorbidities
Rates of comorbid disorders are significantly higher in AS than HFA. The most prevalent comorbidity in both groups is ADHD, but comparing AS and HFA, its prevalence did not statistically differ. An association between ASD and ADHD and other externalizing disorders have been reported and supported by clinical population research and twin studies, suggesting substantial genetic overlap (Taylor et al. 2013, 2015; Cooper et al. 2014; Salazar et al. 2015; Antshel et al. 2016). Considering that comorbidity between these disorders was a relevant and frequent occurrence, differently from the previous edition, DSM-5 allowed the diagnosis of ADHD in the context of an ASD. People with AS displayed more anxiety and depressive symptoms compared to HFA, consistently with previous literature (Mazzone et al. 2012; Simonoff et al. 2013; Ung et al. 2013; Chiang and Gau 2015; De-la-Iglesia and Olivar 2015; Roy et al. 2015; Gillberg et al. 2016).
Clinical and Treatment Implications
Overall, the results of this study suggested a complex aged-related different developmental profile between HFA and AS, characterized by quantitative and qualitative differences such as language development and communication style, academic achievement, comorbidities profile and cognitive functioning. Since language development and academic abilities play a central role in global functioning during the first years of life, HFA require more support in terms of rehabilitative treatments and school educational needs than AS. Consistently, in this study, when DSM-5 specifiers of severity levels were applied to the sample, HFA were more frequently classified in level 2 (“requiring substantial support”) than AS, while AS were more frequently classified in level 1 (“requiring support”) than HFA.
Nevertheless, despite this aged-related definite pattern of differentiation and severity level classification, in middle childhood and adolescence, AS presents higher internalizing comorbidities rate (overall anxiety and depressive disorders) than HFA, as resulted in this study. The presence of high rate of comorbidity is also underlined in previous literature (Mazzone et al. 2012; Simonoff et al. 2013; Ung et al. 2013; Roy et al. 2015; Gillberg et al. 2016). This could be explained with AS higher cognitive and communicative levels, that made them more able in introspection, more “active but odd” (Ghaziuddin 2008; Wing et al. 2011) in relations attempts, more conscious of social difficulties and so more vulnerable to affective disorders (De-la-Iglesias and Olivar 2015).
Current and Proposed Specifier
At present, DSM-5 allows to use specifiers of language impairment, intellectual impairment and severity levels to distinguish clinical profiles of ASD broader category. Nevertheless, considering the results obtained from the clinical profiles comparison and from the DSM-5 severity levels distinction, it is necessary to consider the risk that, in the severity specifier “Level 1” of the DSM-5, many phenotypic variances could be squeezed making this level very heterogeneous. Because of this, any attempts to predict clinical outcomes, develop individualized treatment targets or identify etiological factors may become unreal (Kim et al. 2015). Lai et al. (2013) underlined that defining autism using the umbrella term ASD, could hide the evident heterogeneity. In order to make progress in autism research and ultimately improve clinical practice, there is the necessity to move forward in the identification of subtypes within the autism spectrum. Maybe, a “subtype specifier” for HFA and AS could be useful to direct specific age-related paths of treatment (i.e. services of traditional rehabilitation, such as speech therapy and psychomotricity, and of assistance tools for school targeted to HFA and social training and psychotherapy targeted to AS).
Clinical judgement of qualified professional experts and the use of specific scales, designed on the clinical characteristics of HFA and AS, could provide data for this subtle distinction. In the present study, MASQ gave significantly different scores in HFA and AS, so that its application could help in discriminating AS from HFA subjects. In its original description, MASQ was used to distinguish, beyond AS and autism, high functioning spectrum disorders from other psychiatric disorders. Authors found a threshold of 22 for AS and intermediate scores for autism/PDD NOS (Ghaziuddin and Welch 2013).
In the present study, the suggested threshold is lower (18 with a sensitivity of 84.6% and specificity of 75.7%).
Concordance Between DSM-IV-TR and DSM-5
The second aim of this study was to verify the percentage of individuals maintaining diagnoses when DSM-5 diagnostic criteria were applied. Several reviewed studies found that DSM-5 criteria for ASD have a higher specificity but lower sensitivity as compared to the DSM-IV-TR (Gibbs et al. 2012; Worley and Matson 2012; Koh et al. 2014; McPartland et al. 2014). Recent studies indicated a percentage between 50 and 75% of individuals maintaining diagnosis under DSM-5 criteria, with the greatest decreases among high-functioning populations or previous diagnoses of PDD-NOS or AS (Kulage et al. 2014; Young and Rodi 2014; Smith et al. 2015). In the present study, 97.3% of the total sample met DSM-5 criteria for ASD; the remainder, who did not meet the full criteria, consisted exclusively of AS subjects. These AS subject, even if in presence of cognitive-emotional-social features representative of AS, lacked, at the age of re-examination, of significant clinical global impairment, indispensable to justify the diagnosis. In fact, recent theories of neurodiversity that consider AS a normal human difference rather than a disorder (Baron-Cohen 2002; Jaarsma and Welin 2012; Lorenz and Heinitz 2014), their phenotypes may be considered as a sub-clinical AS forms. Nevertheless, also approving the hypothesis of sub-clinical phenotype, in most cases, these subjects also need a sort of support (cognitive-behavioral treatment, parent/teacher psycho-education, personalization of school program). As in ADHD coding of DSM-5, regarding level 1 ASD patients, it could be suggested the use of a “partial remission” specifier, for those patients who do not meet currently the full diagnostic criteria.
As mentioned above, in the present study, the severity levels application showed a distribution of HFA in level 1 and 2, while AS were totally placed in level 1. The concordance between HFA/AS groups and level 1/level 2 groups, measured with Cohen’s K, could be considered poor. Indeed, the choice of evaluating the concordance using McNemar test was forced, since the matched classifications were not concordant: HFA/AS groups pertained to a clinical perspective, while level1/level 2 to an assistential one. In daily practice, clinicians do not strictly address type and intensity of support only to diagnoses, but they have to consider lots of variables, depending on age, comorbidities and environment.
Conclusion
Considering the available evidences and our results, even continuing to use the broad category of ASD, as Wing (who firstly rescued Asperger’s work) stated, “a number of changes need to be made in order that the future version of DSM might be used reliably and validly in clinical practice and research” (Wing et al. 2011). ASD category of DSM-5 had the merit to have solved lots of previous classification discordances and “splitting” approaches (Volkmar and McPartland 2014). Nevertheless, it could be meaningful to introduce an additional “subtype specifiers” (i.e., Autistic Disorder or Asperger’s disorders), in order to grant the recognition of different “nuances” of the spectrum and determine better individual clinical, assistential and research addresses.
Limitations and Future Directions
The sample of the present study is characterized by a preponderance in HFA group of PDD-NOS that tend to be rather heterogeneous. In previous research, comparisons were made with samples that include larger number of AD. Therefore, it is unknown whether our results may be extended to the broader ASD category. Lastly, in the evaluation of academic achievement, the higher rates of learning problems in children with HFA might be attributed or at least influenced by the lower IQ scores of HFA than AS. Future research about the efficacy of these prospective specifiers could be beneficial.
References
Achenbach, T. M., & Edelbrock, C. (1983). Manual for the child behavior checklist and revised child behavior profile. Burlington: University of Vermont Press.
Achenbach, T. M. (1991). Integrative guide for the 1991 CBCL/4-18, YSR, and TRF profiles. Burlington: Department of Psychiatry, University of Vermont.
American Psychiatric Association. (1980). Diagnostic and statistical manual of mental disorders (3rd ed.). Washington, DC: American Psychiatric Publication, DSM-III.
American Psychiatric Association. (1987). Diagnostic and statistical manual of mental disorders (3rd ed.). Washington, DC: American Psychiatric Publication, DSM-III-R.
American Psychiatric Association. (1994). Diagnostic and statistical manual of mental disorders (4th ed.). Washington, DC: American Psychiatric Association, DSM-IV.
American Psychiatric Association. (2000). Diagnostic and statistical manual of mental disorders (in text rev.), DSM-IV-TR (4th ed.). Washington, DC: American Psychiatric Association.
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Arlington: American Psychiatric Association, DSM-5.
Antshel, K. M., Zhang-James, Y., Wagner, K. E., Ledesma, A., & Faraone, S. V. (2016). An update on the comorbidity of ADHD and ASD: A focus on clinical management. Expert Review of Neurotherapeutics, 16(3), 279–293.
Asperger, H. (1944). Die autistisehen Psychopathen im Kindesalter. Archiv für Psychiatrie und Nervenkrankheit, 117, 76–136.
Attwood, T. (2006). The complete guide to Asperger’s syndrome. Philadelphia: Jessica Kingsley Publishers.
Barahona-Corréa, J. B., & Filipe, C. N. (2016). A concise history of Asperger Syndrome: The short reign of a troublesome diagnosis. Frontiers in Psychology. https://doi.org/10.3389/fpsyg.2015.02024.
Baron-Cohen, S. (2002). Is Asperger syndrome necessarily viewed as a disability? Focus on Autism and Other Developmental Disabilities, 17(3), 186–191.
Bennett, T., Szatmari, P., Bryson, S., Volden, J., Zwaigenbaum, L., Vaccarella, L., Duku, E., & Boyle, M. (2008). Differentiating autism and Asperger syndrome on the basis of language delay or impairment. Journal of Autism and Developmental Disorders, 38(4), 616–625.
Bi, X., Wang, Y., Shu, Q., Sun, Q., & Xu, Q. (2018). Classification of autism spectrum disorder using random support vector machine cluster. Frontiers in Genetics, 9, 18.
Chiang, H. L., & Gau, S. S. (2015). Comorbid psychiatric conditions as mediators to predict later social adjustments in youths with autism spectrum disorder. Journal of Child Psychology and Psychiatry. https://doi.org/10.1111/jcpp.12450.
Chiang, H. L., Tsai, L. Y., Cheung, Y. K., Brown, A., & Li, H. (2014). A meta-analysis of differences in IQ profiles between individuals with Asperger’s disorder and high-functioning autism. Journal of Autism and Developmental Disorders. https://doi.org/10.1007/s10803-013-2025-2.
Cooper, M., Martin, J., Langley, K., Amshere, M., & Thapar, A. (2014). Autistic traits in children with ADHD index clinical and cognitive problems. European Child and Adolescent Psychiatry, 23(1), 23–34.
Cornoldi, C., & Cazzola, C. (2003). Test di valutazione delle abilità di calcolo e problem solving (11–14 anni). Gardolo-Trento: Edizioni Erickson.
Cornoldi, C., & Colpo, G. (1995). Nuove prove di lettura MT per la scuola media inferiore. Firenze: Organizzazioni Speciali.
Cornoldi, C., & Colpo, G. (1998). Prove di lettura MT per la scuola elementare. Firenze: Organizzazioni Speciali.
Cornoldi, C., Lucangeli, D., & Bellina, M. (2002). Test AC-MT. Test di valutazione delle abilità di calcolo. Gruppo MT. Trento: Centro Studi Erickson.
Cornoldi, C., Pra Baldi, A., Friso, G., Giacomin, A., Giofrè, D., & Zaccaria, S. (2010). Prove MT Avanzate di Lettura e Matematica 2 per il biennio della scuola superiore di II grado. Firenze: Organizzazioni Speciali.
De-la-Iglesia, M., & Olivar, J. S. (2015). Risk factors for depression in children and adolescents with high functioning autism spectrum disorders. Scientific World Journal. https://doi.org/10.1155/2015/127853.
Faridi, F., & Khosrowabadi, R. (2017). Behavioral, cognitive and neural markers of Asperger syndrome. Basic and Clinical Neuroscience, 8(5), 349–359.
Fournier, K. A., Hass, C. J., Naik, S. K., Lodha, N., & Cauraugh, J. H. (2010). Motor coordination in autism spectrum disorders: A synthesis and meta-analysis. Journal of Autism and Developmental Disorders, 40(10), 1227–1240.
Fuentes, C. T., Mostofsky, S. H., & Bastian, A. J. (2009). Children with autism show specific and writing impairments. Neurology, 73(19), 1532–1537.
Ghaziuddin, M. (2008). Defining the behavioral phenotype of Asperger Syndrome. Journal of Autism and Developmental Disorders, 38, 138–142.
Ghaziuddin, M., Butler, E., Tsai, L., & Ghaziuddin, N. (1994). Is clumsiness a marker for Asperger syndrome? Journal Intellectual Disability Research, 38(5), 519–527.
Ghaziuddin, M., & Gerstein, L. (1996). Pedantic speaking style differentiates Asperger syndrome from high-functioning autism. Journal of Autism and Developmental Disorders, 26, 585–595.
Ghaziuddin, M., & Mountain-Kimchi, K. (2004). Defining intellectual profile of Asperger Syndrome: Comparison with HFA. Journal of Autism and Developmental Disorders, 34(3), 279–284.
Ghaziuddin, M., & Welch, K. (2013). The Michigan autism spectrum questionnaire a rating scale for high functioning autism spectrum disorders. Autism Research and Treatment, 2013, 708273.
Gibbs, V., Aldridge, F., Chandler, F., Witzlsperger, E., & Smith, K. (2012). Brief report: An exploratory study comparing diagnostic outcomes for autism spectrum disorders under DSM-IV-TR with the proposed DSM-5 revision. Journal of Autism and Developmental Disorders, 42(8), 1750–1756.
Gilchrist, A., Cox, A., Rutter, M., Green, J., Burton, D., & Le Couteur, A. (2001). Development and current functioning in adolescents with Asperger Syndrome: A comparative study. Journal of Child Psychology and Psychiatry, 42(2), 227–240.
Gillberg, I., & Gillberg, C. (1989). Asperger syndrome: Some epidemiological considerations-a research note. Journal of Child Psychology and Psychiatry, 30, 631–638.
Gillberg, I., Helles, A., Billeted, E., & Gillberg, C. (2016). Boys with Asperger syndrome grow up: Psychiatric and neurodevelopmental disorders 20 years after initial diagnosis. Journal of Autism and Developmental Disorders, 46(1), 74–82.
Grapel, J. N., Cicchetti, D. V., & Volkmar, F. R. (2015). Sensory features as diagnostic criteria for autism: Sensory features in autism. Yale Journal of Biology and Medicine, 88(1), 69–71.
Grimm, D., Assouline, B., & Piero, A. (2015). Schooling of patients exhibiting autism spectrum disorders without mental retardation. Encephale, 41(6), 499–506.
Helles, A., Gillberg, C. I., Gillberg, C., & Billstedt, E. (2015). Asperger syndrome in males over two decades: Stability and predictors of diagnosis. Journal of Child Psychology and Psychiatry, 56(6), 711–718.
Howlin, P. (2003). Outcome in high functioning adults with autism and without early language delays: Implications for the differentiation between autism and Asperger syndrome. Journal of Autism and Developmental Disorders, 33(1), 3–13.
Jaarsma, P., & Welin, S. (2012). Autism as a natural human variation: Reflections on the claims of the neurodiversity movement. Health Care Analysis, 20, 20–30.
Jansiewicz, E. M., Goldberg, M. C., Newschaffer, C. J., Denckla, M. B., Landa, R., & Mostofsky, S. H. (2006). Motor signs distinguish children with high functioning autism and Asperger’s syndrome from controls. Journal of Autism and Developmental Disorders, 800, 613–621.
Kanner, L. (1943). Autistic disturbances of affective contact. Nervous Child, 2, 217–250.
Kim, S. H., Macari, S., Koller, J., & Chawarska, K. (2015). Examining the phenotypic heterogeneity of early autism spectrum disorder: Subtypes and short-term outcomes. Journal of Child Psychology and Psychiatry. https://doi.org/10.1111/jcpp.12448.
Klin, A., Pauls, D., Schultz, R., & Volkmar, F. (2005). Three diagnostic approaches to Asperger syndrome: Implications for research. Journal of Autism and Developmental Disorders, 35(2), 221–234.
Klin, A., Volkmar, F., & Sparrow, S. (2000). Asperger syndrome, (1st ed.). New York: Guilford Publication.
Koh, Y. J., Kim, S. J., Cheon, K. A., Leventhal, B. L., & Fombonne, E. (2014). A comparison of DSM-IV pervasive developmental disorder and DSM-5 autism spectrum disorder prevalence in an epidemiologic sample. Journal of the American Academy of Child and Adolescent Psychiatry, 53(5), 500–508.
Koyama, T., Tachimori, H., Osada, H., Takeda, T., & Kurita, H. (2007). Cognitive and symptom profiles in Asperger’s syndrome and high-functioning autism. Psychiatry and Clinical Neurosciences, 61, 99–104.
Kulage, K. M., Smaldone, A. M., & Cohn, E. G. (2014). How will DSM-5 affect autism diagnosis? A systematic literature review and meta-analysis. Journal of Autism and Developmental Disorders, 44(8), 1918–1932.
Lai, M. C., Lombardo, M. V., Chakrabarti, B., & Baron-Cohen, S. (2013). Subgrouping the autism “Spectrum”: Reflection on DSM-5. PLOS Biology. https://doi.org/10.1371/journal.pbio.1001544.
Lai, M. C., Lombardo, M. V., Ecker, C., Chakrabarti, B., Sucling, J., Bullmore, E. T., et al. (2015). Neuroanatomy of individual differences in language in adult males with autism. Cerebral Cortex, 25(10), 3613–3628.
Lorenz, T., & Heinitz, K. (2014). Aspergers different, not less: Occupational strengths and job interests of individuals with Asperger’s Syndrome. PLoS ONE, 9(6), e100358.
Macintosh, K. E., & Dissanayake, C. (2004). Annotation: the similarities and differences between autistic disorder and Asperger’s disorder: A review of the empirical evidence. Journal of Child Psychology and Psychiatry, 45(3), 421–434.
Mazzone, L., Ruta, L., & Reale, L. (2012). Psychiatric comorbidities in Asperger syndrome and high functioning autism: Diagnostic challenges. Annals of General Psychiatry, 11, 16.
McPartland, J. C., Klin, A., & Volkmar, F. (2014). Asperger syndrome, second edition. Assessing and treating high-functioning autism spectrum disorders. New York: Guilford Publication.
Montgomery, C. B., Allison, C., Lai, M. C., Cassidy, S., Langdon, P. E., Baron-Cohen, S. (2016). Do adults with high functioning autism or Asperger syndrome differ in empathy and emotion recognition? Journal of Autism and Developmental Disorders, 46(6), 1931–1940.
Nayate, A., Tonga, B. J., Bradshaw, J. L., McGinley, J. L., Iansek, R., & Rinehart, N. J. (2012). Differentiation of High-functioning autism and Asperger’s disorder based on neuromata behavior. Journal of Autism and Developmental Disorders, 42(5), 707–717.
Noterdaeme, M., Wriedt, E., & Hohne, C. (2010). Asperger’s syndrome and high-functioning autism: Language, motor and cognitive profiles. European Child & Adolescent Psychiatry, 19(6), 475–481.
Planche, P., & Lemonnier, E. (2012). Research in autism spectrum disorders children with HFA and Asperger’s syndrome. Research in Autism Spectrum Disorders, 6, 939–948.
Prior, M., Eisenmajer, R., Leekam, S., Wing, L., Gould, J., Ong, B., et al. (1998). Are there subgroups within the autistic spectrum? A cluster analysis of a group of children with autistic spectrum disorders. Journal of Child Psychology and Psychiatry, 39(6), 893–902.
Rinehart, N., Tonge, B., Brereton, A., & Brandshaw, J. (2010) Attentional blink in young people with high functioning autism and Asperger disorder. Autism, 14(1), 47–66.
Roy, M., Prox-Vagedes, V., Ohlmeier, M. D., & Dillo, W. (2015). Beyond childhood: Psychiatric comorbidities and social background of adults with Asperger syndrome. Psychiatria Danubina, 27(1), 50–59.
Salazar, F., Baird, G., Chandler, S., Tseng, E., O’sullivan, T., Howlin, P., Pickles, A., & Simonoff, E. (2015). Co-occurring psychiatric disorders in preschool and elementary school-aged children with autism spectrum disorder. Journal of Autism and Developmental Disorders, 45(8), 2283–2294.
Sanders, J. L. (2009). Qualitative or quantitative differences between Asperger’s disorder and autism? Historical considerations. Journal of Autism and Developmental Disorders, 39(11), 1560–1567.
Sartori, G., Job, R., & Tressoldi, P. E. (1995). Batteria per la valutazione della dislessia e della disortografia in età evolutiva. Firenze: Organizzazioni Speciali.
Saulnier, C. A., & Kin, A. (2007) Brief report: social and communication abilities and disabilities in higher functioning individuals with autism and Asperger syndrome. Journal of Autism and Developmental Disorders, 37(4), 788–793.
Sharma, S., Woolfson, L. M., & Hunter, S. C. (2012). Confusion and inconsistency in diagnosis of Asperger syndrome: A review of studies from 1981 to 2010. Autism, 16(5), 465–486.
Simonoff, E., Jones, C. R., Baird, G., Pickles, A., Happé, F., & Charman, T. (2013). The persistence and stability of psychiatric problems in adolescents with autism spectrum disorders. Journal of Child Psychology and Psychiatry, 54(2), 186–194.
Smith, I. C., Reichow, B., & Volkmar, F. R. (2015). The effects of DSM-5 criteria on numbers of individuals diagnosed with autism spectrum disorder: A systematic review. Journal of Autism and Developmental Disorders, 45(8), 2541–2552.
Speirs, S., Yelland, G., Reinehart, N., & Tonge, B. (2011). Lexical processing in individuals with high functioning autism and Asperger’s disorder. Autism, 15(5), 307–325.
Spek, A. A., Scholte, E. M., & Van Berckelaer-Onnes, I. A. (2010). Theory of mind in adults with HFA and Asperger syndrome. Journal of Autism and Developmental Disorders, 40(3), 280–289.
Szatmari, P., Bartolucci, G., & Bremner, R. (1989). Asperger’s syndrome and autism: Comparison of early history and outcome. Developmental Medicine and Child Neurology, 31, 709–720.
Taylor, M. J., Charman, T., Robinson, E. B., Plomin, R., Happé, F., Asherson, P., & Ronal, A. (2013). Developmental associations between traits of autism spectrum disorder and attention deficit hyperactivity disorder: A genetically informative, longitudinal twin study. Psychological Medicine, 1(1), 1–12.
Taylor, M. J., Charman, T., & Ronald, A. (2015). Where are the strongest associations between autistic traits and traits of ADHD? Evidence from a community-based twin study. European Child and Adolescent Psychiatry, 24(9), 1129–1138.
Tsai, L. Y., & Ghaziuddin, M. (2014). DSM-5 ASD moves forward into the past. Journal of Autism and Developmental Disorders, 44(2), 321–330.
Ung, D., Wood, J. J., Ehrenreich-May, J., Arnold, E. B., Fuji, C., Renno, P., et al. (2013). Clinical characteristics of high-functioning youth with autism spectrum disorder and anxiety. Neuropsychiatry (London). https://doi.org/10.2217/npy.13.9.
Volkmar, F. R., & McPartland, J. C. (2014). From Kanner to DSM-5: Autism as an evolving diagnostic concept. Annual Review of Clinical Psychology, 10, 193–212.
Volkmar, F. R., State, M., & Klin, A. (2009). Autism and autism spectrum disorders: Diagnostic issues for the coming decade. Journal of Child Psychology and Psychiatry, 50(1–2), 108–115.
Walker, D. R., Thompson, A., Zwaigenbaum, L., Goldberg, J., Bryson, S. E., Mahoney, W. J., et al. (2004). Specifying PDD-NOS: A comparison of PDD-NOS, Asperger syndrome, and autism. Journal of the American Academy of Child and Adolescent Psychiatry, 43(2), 172–180.
Wilson, C. E., Happé, F., Wheelwright, S. J., Ecker, C., Lombardo, M. V., Johnston, P., et al. (2014). The neuropsychology of male adults with high-functioning autism or Asperger syndrome. Autism Research, 7(5), 568–581.
Wing, L. (1981). Asperger’s syndrome: A clinical account. Psychological Medicine, 11, 115–129.
Wing, L., Gould, J., & Gillberg, C. (2011). Autism spectrum disorders in the DSM-V: Better or worse than DSM-IV? Research in Developmental Disabilities, 32(2), 768–773.
Witwer, A. N., & Lecavalier, L. (2008). Examining the validity of autism spectrum disorder subtypes. Journal of Autism and Developmental Disorders, 38(9), 1611–1624.
Woodbury-Smith, M. R., & Volkmar, F. R. (2009). Asperger syndrome. European Child and Adolescent Psychiatry, 18(1), 2–11.
Worley, J. A., & Matson, J. L. (2012). Comparing symptoms of autism spectrum disorders using the current DSM-IV-TR diagnostic criteria and the proposed DSM-Vdiagnostic criteria. Research in Autism Spectrum Disorder, 6(2), 965–970.
Young, R. L., & Rodi, M. L. (2014). Redefining autism spectrum disorder using DSM-5: The implications of the proposed DSM-5 criteria for autism spectrum disorders. Journal of Autism and Developmental Disorders, 44(4), 758–765.
Yu, K. K., Cheung, C., Chua, S. E., & Mc Alonan, G. M. (2011). Can Asperger syndrome be distinguished from autism? An anatomic likelihood meta-analysis of MRI studies. Journal of Psychiatry & Neuroscience, 36(16), 412–421.
Author information
Authors and Affiliations
Contributions
CD, PV and LM designed and coordinated the study. MM and RP participated in data collection. PT performed data analysis. All authors participated in data interpretation and helped to draft the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest to disclose.
Ethical Approval
All procedures performed were in accordance with ethical standards of the local Ethics Committee of the Azienda Ospedaliero-Universitaria Consorziale Policlinico di Bari and with the 1964 Helsinky declaration and its later amendments. This article does not contain any studies with animals performed by any of the authors.
Informed Consent
Informed consent from parents and assent from children and adolescents were obtained prior to enrollment.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
de Giambattista, C., Ventura, P., Trerotoli, P. et al. Subtyping the Autism Spectrum Disorder: Comparison of Children with High Functioning Autism and Asperger Syndrome. J Autism Dev Disord 49, 138–150 (2019). https://doi.org/10.1007/s10803-018-3689-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10803-018-3689-4