Abstract
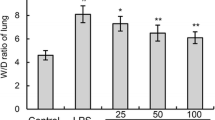
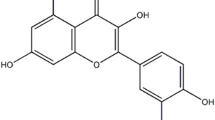
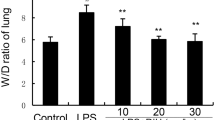
Magnolol, a tradition Chinese herb, displays an array of activities including antifungal, antibacterial, and antioxidant effects. To investigate the protective effect of magnolol on lipopolysaccharide (LPS)-induced acute lung injury (ALI) in mice. ALI was induced in mice by intratracheal instillation of LPS (1 mg/kg). The mice received intratracheal instillation of magnolol (5 μg/kg) 30 min before LPS administration. Pulmonary histological changes were evaluated by hematoxylin-eosin stain and lung wet/dry weight ratios were observed. Concentrations of tumor necrosis factor (TNF)-α and interleukin (IL)-1β, and myeloperoxidase (MPO) activity were measured by enzyme-linked immunosorbent assay. Expression of cyclooxygenase (COX)-2 in lung tissues was determined by Western blot analysis. Magnolol pretreatment significantly attenuated the severity of lung injury and inhibited the production of TNF-α and IL-1β in mice with ALI. After LPS administration, the lung wet/dry weight ratios, as an index of lung edema, and MPO activity were also markedly reduced by magnolol pretreatment. The expression of COX-2 was significantly suppressed by magnolol pretreatment. Magnolol potently protected against LPS-induced ALI and the protective effects of magnolol may attribute partly to the suppression of COX-2 expression.





Similar content being viewed by others
References
Kollef, M.H., and D.P. Schuster. 1995. The acute respiratory distress syndrome. New England Journal of Medicine 332: 27–37.
Worthen, G.S., C. Haslett, A.J. Rees, R.S. Gumbay, J.E. Henson, and P.M. Henson. 1987. Neutrophil-mediated pulmonary vascular injury: synergistic effect of trace amounts of lipopolysaccharide and neutrophil stimuli on vascular permeability and neutrophil sequestration in the lung. The American Review of Respiratory Disease 136: 19–28.
Rubenfeld, G.D., E. Caldwell, E. Peabody, J. Weaver, D.P. Martin, M. Neff, E.J. Stern, and L.D. Hudson. 2005. Incidence and outcomes of acute lung injury. New England Journal of Medicine 353: 1685–1693.
Hla, T., and K. Neilson. 1992. Human cyclooxygenase-2 cDNA. Proceedings of the National Academy of Sciences of the United States of America 89: 7384–7388.
Masferrer, J.L., B.S. Zweifel, P.T. Manning, S.D. Hauser, K.M. Leahy, W.G. Smith, P.C. Isakson, and K. Seibert. 1994. Selective inhibition of inducible cyclooxygenase 2 in vivo is antiinflammatory and nonulcerogenic. Proceedings of the National Academy of Sciences of the United States of America 91: 3228–3232.
Diaz, A., K.P. Chepenik, J.H. Korn, A.M. Reginato, and S.A. Jimenez. 1998. Differential regulation of cyclooxygenases 1 and 2 by interleukin-1 beta, tumor necrosis factor-alpha, and transforming growth factor-beta 1 in human lung fibroblasts. Experimental Cell Research 241: 222–229.
Serou, M.J., M.A. DeCoster, and N.G. Bazan. 1999. Interleukin-1 beta activates expression of cyclooxygenase-2 and inducible nitric oxide synthase in primary hippocampal neuronal culture: platelet-activating factor as a preferential mediator of cyclooxygenase-2 expression. Journal of Neuroscience Research 58: 593–598.
Cuzzocrea, S., E. Mazzon, L. Sautebin, L. Dugo, I. Serraino, A. De Sarro, and A.P. Caputi. 2002. Protective effects of Celecoxib on lung injury and red blood cells modification induced by carrageenan in the rat. Biochemical Pharmacology 63: 785–795.
Fukunaga, K., P. Kohli, C. Bonnans, L.E. Fredenburgh, and B.D. Levy. 2005. Cyclooxygenase 2 plays a pivotal role in the resolution of acute lung injury. The Journal of Immunology 174: 5033–5039.
Ohara, M., T. Sawa, K. Kurahashi, J.P. Wiener-Kronish, V. Doshi, I. Kudoh, and M.A. Gropper. 1998. Induction of cyclooxygenase-2 in alveolar macrophages after acid aspiration: Selective cyclooxygenase-2 blockade reduces interleukin-6 production. Anesthesiology 88: 1014–1022.
Bang, K.H., Y.K. Kim, B.S. Min, M.K. Na, Y.H. Rhee, J.P. Lee, and K.H. Bae. 2000. Antifungal activity of magnolol and honokiol. Archives of Pharmacal Research 23: 46–49.
Lo, Y.C., C.M. Teng, C.F. Chen, C.C. Chen, and C.Y. Hong. 1994. Magnolol and honokiol isolated from Magnolia officinalis protect rat heart mitochondria against lipid peroxidation. Biochemical Pharmacology 47: 549–553.
Matsuda, H., T. Kageura, M. Oda, T. Morikawa, Y. Sakamoto, and M. Yoshikawa. 2001. Effects of constituents from the bark of Magnolia obovata on nitric oxide production in lipopolysaccharide-activated macrophages. Chemical & Pharmaceutical Bulletin 49: 716–720.
Park, J., J. Lee, E. Jung, Y. Park, K. Kim, B. Park, K. Jung, E. Park, J. Kim, and D. Park. 2004. In vitro antibacterial and anti-inflammatory effects of honokiol and magnolol against Propionibacterium sp. European Journal of Pharmacology 496: 189–195.
Lee, M.M., H.M. Huang, M.T. Hsieh, C.S. Chen, F.T. Yeh, and J.S. Kuo. 2000. Anti-inflammatory and neuroprotective effects of magnolol in chemical hypoxia in rat cultured cortical cells in hypomagnololycemic media. The Chinese Journal of Physiology 43: 61–67.
Chen, Y.H., F.Y. Lin, P.L. Liu, Y.T. Huang, J.H. Chiu, Y.C. Chang, K.M. Man, C.Y. Hong, Y.Y. Ho, and M.T. Lai. 2009. Antioxidative and hepatoprotective effects of magnolol on acetaminophen-induced liver damage in rats. Archives of Pharmacal Research 32: 221–228.
Loong, C.C., J.H. Chiu, R.C. Tiao, Y.Y. Chiu, C.W. Wu, and W.Y. Lui. 2001. Pretreatment with magnolol attenuates ischemia-reperfusion injury in rat small intestine. Transplantation Proceedings 33: 3737–3738.
Parsey, M.V., R. Tuder, and E. Abraham. 1998. Neutrophils are major contributors to intraparenchymal lung IL-1β expression after hemorrhage and endotoxemia. The Journal of Immunology 160: 1007–1101.
Shenkar, R., and E. Abraham. 1999. Mechanisms of lung neutrophil activation after hemorrhage or endotoxemia: Roles of reactive oxygen intermediates, NF-κB, and cyclic AMP response element binding protein. The Journal of Immunology 163: 954–962.
Bradley, P.P., D.A. Priebat, R.D. Christensen, and G. Rothstein. 1982. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. The Journal of Investigative Dermatology 78: 206–209.
Brigham, K.L., and B. Meyrick. 1986. Endotoxin and lung injury. The American Review of Respiratory Disease 133: 913–927.
Goodman, R.B., J. Pugin, J.S. Lee, and M.A. Matthay. 2003. Cytokine-mediated inflammation in acute lung injury. Cytokine & Growth Factor Reviews 14: 523–535.
Chen, J., X. Liu, Q. Shu, S. Li, and F. Luo. 2008. Ghrelin attenuates lipopolysaccharide-induced acute lung injury through NO pathway. Medical Science Monitor 14: 141–146.
Mei, S.H., S.D. McCarter, Y. Deng, C.H. Parker, W.C. Liles, and D.J. Stewart. 2007. Prevention of LPS-induced acute lung injury in mice by mesenchymal stem cells overexpressing angiopoietin 1. PLoS Medicine 4: 1525–1537.
Ejima, K., M.D. Layne, I.M. Carvajal, P.A. Kritek, R.M. Baron, Y.H. Chen, J. Vom Saal, B.D. Levy, S.F. Yet, and M.A. Perrella. 2003. Cyclooxygenase-2-deficient mice are resistant to endotoxin-induced inflammation and death. The FASEB Journal 17: 1325–1327.
Funk, C.D. 2001. Prostamagnololandin and leukotrienes: advances in eicosanoid biology. Science 294: 1871–1875.
Kim, S.O., J.K. Kundu, Y.K. Shin, J.H. Park, M.H. Cho, T.Y. Kim, and Y.J. Surh. 2005. [6]-Gingerol inhibits COX-2 expression by blocking the activation of p38 MAP kinase and NF-kappaB in phorbol ester-stimulated mouse skin. Oncogene 24: 2558–2567.
Gust, R., J.K. Kozlowski, A.H. Stephenson, and D.P. Schuster. 1999. Role of cyclooxygenase-2 in oleic acid-induced acute lung injury. American Journal of Respiratory and Critical Care Medicine 160: 1165–1170.
Chida, M., and N.F. Voelkel. 1996. Effects of acute and chronic hypoxia on rat lung cyclooxygenase. The American Journal of Physiology 270: 872–878.
Feng, L., Y. Xia, G.E. Garcia, D. Hwang, and C.B. Wilson. 1995. Involvement of reactive oxygen intermediates in cyclooxygenase-2 expression induced by interleukin-1, tumor necrosis factor-alpha, and lipopolysaccharide. The Journal of Clinical Investigation 95: 1669–1675.
O'Brien, G., C.J. Shields, D.C. Winter, J.P. Dillon, W.O. Kirwan, and H.P. Redmond. 2005. Cyclooxygenase-2 plays a central role in the genesis of pancreatitis and associated lung injury. Hepatobiliary & Pancreatic Diseases International 4: 126–129.
Sartori, C., and M.A. Matthay. 2002. Alveolar epithelial fluid transport in acute lung injury: new insights. The European Respiratory Journal 20: 1299–1313.
Kong, C.W., K. Tsai, J.H. Chin, W.L. Chan, and C.Y. Hong. 2000. Magnolol attenuates peroxidative damage and improves survival of rats with sepsis. Shock 13: 24–28.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yun Feng Ni and Tao Jiang authors contributed equally to this work and should be regarded as co-first authors.
Rights and permissions
About this article
Cite this article
Ni, Y.F., Jiang, T., Cheng, Q.S. et al. Protective Effect of Magnolol on Lipopolysaccharide-Induced Acute Lung Injury in Mice. Inflammation 35, 1860–1866 (2012). https://doi.org/10.1007/s10753-012-9507-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-012-9507-9