Abstract
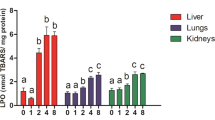
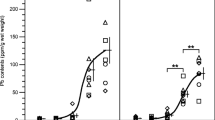
Biomarker responses and histopathological lesions have been documented in laboratory mammals exposed to elevated concentrations of lead and cadmium. The exposure of white-footed mice (Peromyscus leucopus) to these metals and the potential associated toxic effects were examined at three contaminated sites in the Southeast Missouri Lead Mining District and at a reference site in MO, USA. Mice from the contaminated sites showed evidence of oxidative stress and reduced activity of red blood cell δ-aminolevulinic acid dehydratase (ALAD). Histological examinations of the liver and kidney, cytologic examination of blood smears, and biomarkers of lipid peroxidation and DNA damage failed to show indications of toxic effects from lead. The biomagnification factor of cadmium (hepatic concentration/soil concentration) at a site with a strongly acid soil was 44 times the average of the biomagnification factors at two sites with slightly alkaline soils. The elevated concentrations of cadmium in the mice did not cause observable toxicity, but were associated with about a 50% decrease in expected tissue lead concentrations and greater ALAD activity compared to the activity at the reference site. Lead was associated with a decrease in concentrations of hepatic glutathione and thiols, whereas cadmium was associated with an increase. In addition, to support risk assessment efforts, we developed linear regression models relating both tissue lead dosages (based on a previously published a laboratory study) and tissue lead concentrations in Peromyscus to soil lead concentrations.

Similar content being viewed by others
References
Agency for Toxic Substances and Disease Registry. (2004). Interaction profile for: arsenic, cadmium, chromium, and lead. Atlanta: U.S. Department of Health & Human Services.
Agency for Toxic Substances and Disease Registry. (2007). Toxicological profile for lead. Atlanta: U.S. Department of Health and Human Services.
Agency for Toxic Substances and Disease Registry. (2012). Toxicological profile for cadmium. Atlanta: U.S. Department of Health & Human Services.
Agrawal, S., Flora, G., Bhatnagar, P., & Flora, S. (2014). Comparative oxidative stress, metallothionein induction and organ toxicity following chronic exposure to arsenic, lead and mercury in rats. Cellular and Molecular Biology (Noisy-Le-Grand), 60(2), 13–21.
Ahamed, M., & Siddiqui, M. K. J. (2007). Low level lead exposure and oxidative stress: current opinions. Clinica Chimica Acta, 383(1–2), 57–64. https://doi.org/10.1016/j.cca.2007.04.024.
Allert, A. L., Fairchild, J. F., Di Stefano, R. J., Schmitt, C. J., Besser, J. M., Brumbaugh, W. G., & Poulton, B. C. (2008). Effects of lead-zinc mining on crayfish (Orconectes hylas) in Black River watershed, Missouri, USA. Freshwater Crayfish, 16, 97–111.
Alya, A., Bini Ines, D., Montassar, L., Najoua, G., & Saloua, E. F. (2015). Oxidative stress, biochemical alterations, and hyperlipidemia in female rats induced by lead chronic toxicity during puberty and post puberty periods. Iranian Journal of Basic Medical Sciences, 18(10), 1034–1043.
Berlin, A., & Schaller, K. (1974). European standardized method for the determination of delta-aminolevulinic acid dehydratase activity in blood. Zeitschrift für Klinische Chemie und Klinische Biochemie, 12(8), 389–390.
Besser, J., Brumbaugh, W., May, T., & Schmitt, C. (2007). Biomonitoring of lead, zinc, and cadmium in streams draining lead-mining and non-mining areas, southeast Missouri, USA. Environmental Monitoring and Assessment, 129(1), 227–241.
Beyer, W., Pattee, O., Sileo, L., Hoffman, D., & Mulhern, B. (1985). Metal contamination in wildlife living near two zinc smelters. Environmental Pollution Series A, Ecological and Biological, 38(1), 63–86. https://doi.org/10.1016/0143-1471(85)90094-7.
Beyer, W., Connnor, E., & Gerould, S. (1994). Estimates of soil ingestion by wildlife. The Journal of Wildlife Management, 58(2), 375–382. https://doi.org/10.2307/3809405.
Beyer, W., Franson, J., French, J., May, T., Rattner, B., Shearn-Bochsler, V., et al. (2013). Toxic exposure of songbirds to lead in the Southeast Missouri lead mining district. Archives of Environmental Contamination and Toxicology, 65(3), 598–610. https://doi.org/10.1007/s00244-013-9923-3.
Beyer, W., Basta, N. T., Chaney, R. L., Henry, P. F., Mosby, D. E., Rattner, B. A., et al. (2016). Bioaccessibility tests accurately estimate bioavailability of lead to quail. Environmental Toxicology and Chemistry, 35(9), 2311–2319. https://doi.org/10.1002/etc.3399.
Burch, H., & Siegel, A. (1971). Improved method for measurement of delta-aminolevulinic acid dehydratase activity in human erythrocytes. Clinical Chemistry, 17(10), 1038–1041.
Casteel, S., Weis, C. P., Henningsen, G. M., & Brattin, W. J. (2006). Estimation of relative bioavailability of lead in soil and soil-like materials using young swine. Environmental Health Perspectives, 114(8), 1162–1171. https://doi.org/10.1289/ehp.8852.
Cooke, J. (2011). Cadmium in small mammals. In W. Beyer & J. Meador (Eds.), Environmental contaminants in biota: interpreting tissue concentrations (2nd ed., pp. 627–642). Boca Raton: Taylor and Francis. https://doi.org/10.1201/b10598-21.
Czarnezki, J. M. (1985). Accumulation of lead in fish from missouri streams impacted by lead mining. Bulletin of Environmental Contamination and Toxicology, 34(1), 736–745
Damek-Poprawa, M., & Sawicka-Kapusta, K. (2003). Damage to the liver, kidney, and testis with reference to burden of heavy metals in yellow-necked mice from areas around steelworks and zinc smelters in Poland. Toxicology, 186(1), 1–10. https://doi.org/10.1016/S0300-483X(02)00595-4.
Davis, J. R., & Avram, M. J. (1978). A comparison of the stimulatory effects of cadmium and zinc on normal and lead-inhibited human erythrocytic δ-aminolevulinic acid dehydratase activity in vitro. Toxicology and Applied Pharmacology, 44(1), 181–190. https://doi.org/10.1016/0041-008X(78)90297-1.
Ercal, N., Gurer-Orhan, H., & Aykin-Burns, N. (2001). Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Current Topics in Medicinal Chemistry, 1(6), 529–539. https://doi.org/10.2174/1568026013394831.
Flora, S. J., Pande, M., & Mehta, A. (2003). Beneficial effect of combined administration of some naturally occurring antioxidants (vitamins) and thiol chelators in the treatment of chronic lead intoxication. Chemico-Biological Interactions, 145(3), 267–280. https://doi.org/10.1016/S0009-2797(03)00025-5.
Gale, N., & Wixson, B. (1979). Cadmium in forest ecosystems around lead smelters in Missouri. Environmental Health Perspectives, 28, 23–37. https://doi.org/10.1289/ehp.792823.
George, J. W., & Duncan, J. R. (1979). The hematology of lead poisoning in man and animals. Veterinary Clinical Pathology, 8(1), 23–30. https://doi.org/10.1111/j.1939-165X.1979.tb00878.x.
Goering, P. L., & Fowler, B. A. (1987). Metal constitution of metallothionein influences inhibition of δ-aminolaevulinic acid dehydratase (porphobilinogen synthase) by lead. Biochemical Journal, 245(2), 339–345. https://doi.org/10.1042/bj2450339.
Goyer, R. A., Leonard, D. L., Moore, J. F., Rhyne, B., & Krigman, M. R. (1970). Lead dosage and the role of the intranuclear inclusion body. Archives of Environmental Health: An International Journal, 20(6), 705–711. https://doi.org/10.1080/00039896.1970.10665647.
Gurer, H., & Ercal, N. (2000). Can antioxidants be beneficial in the treatment of lead poisoning? Free Radical Biology & Medicine, 29(10), 927–945. https://doi.org/10.1016/S0891-5849(00)00413-5.
Haneef, S., Swarup, D., Dwivedi, S., & Dash, P. (1998). Effects of concurrent exposure to lead and cadmium on renal function in goats. Small Ruminant Research, 28(3), 257–261. https://doi.org/10.1016/S0921-4488(97)00083-7.
Hogan, G., & Razniak, S. (1992). Split dose studies on the erythropoietic effects of cadmium. Bulletin of Environmental Contamination and Toxicology, 48(6), 857–864.
Hooper, M. J., Cobb, G. P., & McMurry, S. T. (2002). Wildlife biomonitoring at the anaconda smelter site. Montana: Deer Lodge County http://www.fws.gov/montanafieldoffice/environmental_contaminants/abr/abr_final_report.pdf. Accessed 25 November 2017.
Isaksson, C. (2010). Pollution and its impact on wild animals: a meta-analysis on oxidative stress. EcoHealth, 7(3), 342–350. https://doi.org/10.1007/s10393-010-0345-7.
Kabata-Pendias, A., & Mukherjee, A. B. (2007). Trace elements from soil to human. New York: Springer. https://doi.org/10.1007/978-3-540-32714-1.
Kamiyama, T., Miyakawa, H., Li, J. P., Akiba, T., Liu, J. H., Liu, J.-H., et al. (1995). Effects of one-year cadmium exposure on livers and kidneys and their relation to glutathione levels. Research Communications in Molecular Pathology and Pharmacology, 88(2), 177–186.
Kaneko, J. J., Harvey, J. W., & Bruss, M. L. (2008). Clinical biochemistry of domestic animals. Burlington: Academic Press.
Kasperczyk, S., Blaszczyk, I., Dobrakowski, M., Romuk, E., Kapka-Skrzypczak, L., Adamek, M., et al. (2013). Exposure to lead affects male biothiols metabolism. Annals of Agricultural and Environmental Medicine, 20(4), 721–725.
Kempe, D. S., Lang, P. A., Eisele, K., Klarl, B. A., Wieder, T., Huber, S. M., et al. (2005). Stimulation of erythrocyte phosphatidylserine exposure by lead ions. American Journal of Physiology-Cell Physiology, 288(2), C396–C402.
Leary, S., Underwood, W., Anthony, R., Cartner, S., Corey, D., Grandin, T., et al. (2013). AVMA guidelines for the euthanasia of animals: 2013 edition. http://works.bepress.com/cheryl_greenacre/14/. Accessed 25 Nov 2017.
Levengood, J. M., & Heske, E. J. (2008). Heavy metal exposure, reproductive activity, and demographic patterns in white-footed mice (Peromyscus leucopus) inhabiting a contaminated floodplain wetland. Science of the Total Environment, 389(2), 320–328. https://doi.org/10.1016/j.scitotenv.2007.08.050.
Liu, C.-M., Ma, J.-Q., & Sun, Y.-Z. (2012). Puerarin protects the rat liver against oxidative stress-mediated DNA damage and apoptosis induced by lead. Experimental and Toxicologic Pathology, 64(6), 575–582. https://doi.org/10.1016/j.etp.2010.11.016.
Ma, W.-C. (1989). Effect of soil pollution with metallic lead pellets on lead bioaccumulation and organ/body weight alterations in small mammals. Archives of Environmental Contamination and Toxicology, 18(4), 617–622. https://doi.org/10.1007/BF01055030.
Ma, W.-C. (2011). Lead in mammals. In W. Beyer & J. P. Meador (Eds.), Environmental contaminants in biota: interpreting tissue concentrations (2nd ed., pp. 595–607). Boca Raton: Taylor and Francis.
Ma, W.-C., Denneman, W., & Faber, J. (1991). Hazardous exposure of ground-living small mammals to cadmium and lead in contaminated terrestrial ecosystems. Archives of Environmental Contamination and Toxicology, 20(2), 266–270. https://doi.org/10.1007/BF01055914.
Mahaffey, K. R., Capar, S. G., Gladen, B. C., & Fowler, B. A. (1981). Concurrent exposure to lead, cadmium, and arsenic. Effects on toxicity and tissue metal concentrations in the rat. Journal of Laboratory and Clinical Medicine, 98(4), 463–481.
McBride, T. (2007). Influence of metal mixtures on co-occurring toxic metal bioavailability and effects in adult and developing deer mice. PhD, Texas Tech University, Lubbock.
McFarland, C. (2005). Delta-aminolevulinic acid dehydratase (ALAD) characterization and use in metal exposure assessments. PhD, Texas Tech University, Lubbock.
Monaghan, P., Metcalfe, N. B., & Torres, R. (2009). Oxidative stress as a mediator of life history trade-offs: mechanisms, measurements and interpretation. Ecology Letters, 12(1), 75–92. https://doi.org/10.1111/j.1461-0248.2008.01258.x.
National Research Council. (2005). Mineral tolerance of animals (2nd ed.). Washington, DC: National Academy Press.
Niethammer, K., Atkinson, R., Baskett, T., & Samson, F. (1985). Metals in riparian wildlife of the lead mining district of southeastern Missouri. Archives of Environmental Contamination and Toxicology, 14(2), 213–223.
Pain, D. (1987). Hematological parameters as predictors of blood lead and indicators of lead poisoning in the black duck. Environmental Pollution, 60, 67–81.
Pavlowsky, R. T., Owen, M., & Martin, D. (2010). Distribution, geochemistry, and storage of mining sediment in channel and floodplain deposits of the big river system in St. Francois, Washington, and Jefferson Counties, Missouri. Ozarks Environmental and Water Resources Institute (OEWRI), Environmental Data Report, 10–002.
Reynolds, K., Schwarz, M., McFarland, C., McBride, T., Adair, B., Strauss, R., et al. (2006). Northern pocket gophers (Thomomys talpoides) as biomonitors of environmental metal contamination. Environmental Toxicology and Chemistry, 25(2), 458–469. https://doi.org/10.1897/05-130R1.1.
Sakai, T., & Morita, Y. (1996). δ-Aminolevulinic acid in plasma or whole blood as a sensitive indicator of lead effects, and its relation to the other heme-related parameters. International Archives of Occupational and Environmental Health, 68(2), 126–132.
Salińska, A., Włostowski, T., & Zambrzycka, E. (2012). Effect of dietary cadmium and/or lead on histopathological changes in the kidneys and liver of bank voles Myodes glareolus kept in different group densities. Ecotoxicology, 21(8), 2235–2243. https://doi.org/10.1007/s10646-012-0979-z.
Seeger, C. (2008). History of mining in the Southeast Missouri lead district and description of mine processes, regulatory controls, environmental effects, and mine facilities in the viburnum trend subdistrict. In M. Kleeschulte (Ed.), Hydrologic investigations concerning lead mining issues in southeastern Missouri: U.S. Geological Survey Scientific Investigations Report 2008–5140.
Smith, E., Gancarz, D., Rofe, A., Kempson, I., Weber, J., & Juhasz, A. (2012). Antagonistic effects of cadmium on lead accumulation in pregnant and non-pregnant mice. Journal of Hazardous Materials, 199, 453–456.
Stansley, W., & Roscoe, D. (1996). The uptake and effects of lead in small mammals and frogs at a trap and skeet range. Archives of Environmental Contamination and Toxicology, 30(2), 220–226. https://doi.org/10.1007/BF00215801.
Tessier, N., Noël, S., & Lapointe, F.-J. (2004). A new method to discriminate the deer mouse (Peromyscus maniculatus) from the white-footed mouse (Peromyscus leucopus) using species-specific primers in multiplex PCR. Canadian Journal of Zoology, 82(11), 1832–1835. https://doi.org/10.1139/z04-173.
Wardrop, J., & Weiss, D. (2010). Schalm’s veterinary hematology (6th ed.). Ames: Blackwell Publishing Ltd..
Whitaker, J. O. (1966). Food of Peromyscus maniculatus bairdi and Peromyscus leucopus in Vigo County, Indiana. Journal of Mammalogy, 47(3), 473–486. https://doi.org/10.2307/1377688.
Acknowledgements
We thank Catherine Maddox for assistance with the hepatic biomarker analyses. David Hoffman of the Patuxent Wildlife Research Center and 2 anonymous reviewers provided helpful suggestions on the manuscript. This work was partly funded by the Missouri Department of Natural Resources through the U.S. Department of the Interior’s Natural Resource Damage and Restoration Program. Use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. government. Supplemental data are available at the US Geological Survey ScienceBase-Catalog website at https://doi.org/10.5066/F7JD4V85.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Beyer, W., Casteel, S.W., Friedrichs, K.R. et al. Biomarker responses of Peromyscus leucopus exposed to lead and cadmium in the Southeast Missouri Lead Mining District. Environ Monit Assess 190, 104 (2018). https://doi.org/10.1007/s10661-017-6442-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-6442-0