Abstract
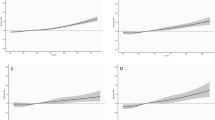
Caffeine is commonly consumed during pregnancy, crosses the placenta, with fetal serum concentrations similar to the mother’s, but studies of birth outcome show conflicting findings. We systematically searched Medline and Embase for relevant publications. We conducted meta-analysis of dose–response curves for associations between caffeine intake and spontaneous abortion, stillbirth, preterm delivery, low birth weight and small for gestational age (SGA) infants. Meta-analyses included 60 unique publications from 53 cohort and case–control studies. An increment of 100 g caffeine was associated with a 14 % (95 % CI 10–19 %) increase in risk of spontaneous abortion, 19 % (5–35 %) stillbirth, 2 % (−2 to 6 %) preterm delivery, 7 % (1–12 %) low birth weight, and 10 % (95 % CI 6–14 %) SGA. There was substantial heterogeneity in all models, partly explained by adjustment for smoking and previous obstetric history, but not by prospective assessment of caffeine intake. There was evidence of small-study effects such as publication bias. Greater caffeine intake is associated with an increase in spontaneous abortion, stillbirth, low birth weight, and SGA, but not preterm delivery. There is no identifiable threshold below which the associations are not apparent, but the size of the associations are generally modest within the range of usual intake and are potentially explained by bias in study design or publication. There is therefore insufficient evidence to support further reductions in the maximum recommended intake of caffeine, but maintenance of current recommendations is a wise precaution.



Similar content being viewed by others
References
Ministry of Agriculture Fisheries and Food. Survey of caffeine and other methylxanthines in energy drinks and other caffeine-containing products (updated). London: Ministry of Agriculture, Fisheries and Food. 1998. Report No.: 144.
Committee on Toxicity of Chemicals in Food Consumer Products and the Environment. Statement on the reproductive effects of caffeine. Food Standards Agency. 2001.
Wilcox A, Weinberg C, Baird D. Caffeinated beverages and decreased fertility. The Lancet. 1988;332:1453–6.
Santos IS, Victora CG, Huttly S, Morris S. Caffeine intake and pregnancy outcomes: a meta-analytic review. Cad Saude Publica. 1998;14:523–30.
Fernandes O, Sabharwal M, Smiley T, Pastuszak A, Koren G, Einarson T. Moderate to heavy caffeine consumption during pregnancy and relationship to spontaneous abortion and abnormal fetal growth: a meta-analysis. Reprod Toxicol. 1998;12:435–44.
Boylan SM, Cade JE, Kirk SFL, Greenwood DC, White KLM, Shires S, et al. Assessing caffeine exposure in pregnant women. Br J Nutr. 2008;100:875–82.
Bech BH, Obel C, Henriksen TB, Olsen J. Effect of reducing caffeine intake on birth weight and length of gestation: randomised controlled trial. BMJ. 2007;334:409.
Committee on Toxicity of Chemicals in Food Consumer Products and the Environment. Statement on the review of caffeine. Department of Health. 1984.
Committee on Toxicity of Chemicals in Food Consumer Products and the Environment. Statement on the reproductive effects of caffeine. Food Standards Agency. 2008.
Organisation of Teratology Information Specialists. Caffeine and pregnancy. 2006. http://www.otispregnancy.org/pdf/caffeine. Accessed 13 June 2014.
NHS Choices. Caffeine and pregnancy. http://www.nhs.uk/news/2008/11November/Pages/Newcaffeineadviceinpregnancy.aspx. Accessed 13 June 2014.
American College of Obstetricians and Gynecologists. Education pamphlet AP001—Nutrition during pregnancy. Washington DC: American College of Obstetricians and Gynecologists; 2008.
Yu WW, Schmid CH, Lichtenstein AH, Lau J, Trikalinos TA. Empirical evaluation of meta-analytic approaches for nutrient and health outcome dose–response data. Res Syn Meth. 2013;4:256–68.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. J Am Med Assoc. 2000;283:2008–12.
NHS Choices. Should I limit caffeine during pregnancy? http://www.nhs.uk/chq/Pages/limit-caffeine-during-pregnancy.aspx. Accessed 13 June 2014.
Wells GA, Shea B, O’Connell D, Peterson J, Losos M, Tugwell P. The Newcastle–Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 13 June 2014.
Greenland S, Longnecker MP. Methods for trend estimation from summarized dose–response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301–9.
Chiaffarino F, Parazzini F, Chatenoud L, Ricci E, Tozzi L, Chiantera V, et al. Coffee drinking and risk of preterm birth. Eur J Clin Nutr. 2006;60:610–3.
Williams MA, Mittendorf R, Stubblefield PG, Lieberman E, Schoenbaum SC, Monson RR. Cigarettes, coffee, and preterm premature rupture of the membranes. Am J Epidemiol. 1992;135:895–903.
Hamling J, Lee P, Weitkunat R, Ambuhl M. Facilitating meta-analyses by deriving relative effect and precision estimates for alternative comparisons from a set of estimates presented by exposure level or disease category. Stat Med. 2008;27:954–70.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Harrell FE Jr, Lee KL, Pollock BG. Regression models in clinical studies: determining relationships between predictors and response. J Natl Cancer Inst. 1988;80:1198–202.
White IR. Multivariate random-effects meta-analysis. Stata J. 2009;9:40–56.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58.
Boylan SM, Greenwood DC, Alwan N, Cooke MS, Dolby VA, Hay AW, et al. Does nausea and vomiting of pregnancy play a role in the association found between maternal caffeine intake and fetal growth restriction? Matern Child Health J. 2013;17:601–8.
StataCorp. Stata statistical software: Release 13.1. College Station, TX: Stata Corporation; 2013.
Infante-Rivard C, Fernandez A, Gauthier R, David M, Rivard GE. Fetal loss associated with caffeine intake before and during pregnancy. JAMA. 1993;270:2940–3.
Caan BJ, Goldhaber MK. Caffeinated beverages and low birthweight: a case–control study. Am J Public Health. 1989;79:1299–300.
Boylan S, Cade JE, Dolby VA, Greenwood DC, Hay AWM, Kirk SFL, et al. Maternal caffeine intake during pregnancy and risk of fetal growth restriction: a large prospective observational study. BMJ. 2008;337:a2332.
Khoury JC, Miodovnik M, Buncher CR, Kalkwarf H, McElvy S, Khoury PR, et al. Consequences of smoking and caffeine consumption during pregnancy in women with type 1 diabetes. J Matern Fetal Neonatal Med. 2004;15:44–50.
Sengpiel V, Elind E, Bacelis J, Nilsson S, Grove J, Myhre R, et al. Maternal caffeine intake during pregnancy is associated with birth weight but not with gestational length: results from a large prospective observational cohort study. BMC Med. 2013;11:42.
Spinillo A, Capuzzo E, Nicola SE, Colonna L, Egbe TO, Zara C. Factors potentiating the smoking-related risk of fetal growth retardation. Br J Obstet Gynaecol. 1994;101:954–8.
Al-Ansary LA, Babay ZA. Risk factors for spontaneous abortion: a preliminary study on Saudi women. J R Soc Health. 1994;114:188–93.
Axelsson G, Rylander R, Molin I. Outcome of pregnancy in relation to irregular and inconvenient work schedules. Br J Ind Med. 1989;46:393–8.
Furuhashi N, Sata S, Suzuki M, Hiruta M, Tanaka M, Takahashi T. Effects of caffeine ingestion during pregnancy. Gynecol Obstet Invest. 1985;19:187–91.
Parazzini F, Bocciolone L, Fedele L, Negri E, La VC, Acaia B. Risk factors for spontaneous abortion. Int J Epidemiol. 1991;20:157–61.
Windham GC, Von BJ, Waller K, Fenster L. Exposure to environmental and mainstream tobacco smoke and risk of spontaneous abortion. Am J Epidemiol. 1999;149:243–7.
Pollack AZ, Buck Louis GM, Sundaram R, Lum KJ. Caffeine consumption and miscarriage: a prospective cohort study. Fertil Steril. 2010;93:304–6.
Dominguez-Rojas V, De Juanes-Pardo JR, Astasio-Arbiza P, Ortega-Molina P, Gordillo-Florencio E. Spontaneous abortion in a hospital population: are tobacco and coffee intake risk factors? Eur J Epidemiol. 1994;10:665–8.
Eskenazi B, Stapleton AL, Kharrazi M, Chee WY. Associations between maternal decaffeinated and caffeinated coffee consumption and fetal growth and gestational duration. Epidemiology. 1999;10:242–9.
Tough SC, Newburn-Cook CV, White DE, Fraser-Lee NJ, Faber AJ, Frick C, et al. Do maternal characteristics and past pregnancy experiences predict preterm delivery among women aged 20 to 34? J Obstet Gynaecol Can. 2003;25:656–66.
Kesmodel U, Olsen SF, Secher NJ. Does alcohol increase the risk of preterm delivery? Epidemiology. 2000;11:512–8.
Mikkelsen TB, Osterdal ML, Knudsen VK, Haugen M, Meltzer HM, Bakketeig L, et al. Association between a Mediterranean-type diet and risk of preterm birth among Danish women: a prospective cohort study. Acta Obstet Gynecol Scand. 2008;87:325–30.
Jarosz M, Wierzejska R, Siuba M. Maternal caffeine intake and its effect on pregnancy outcomes. Eur J Obstet Gynecol Reprod Biol. 2012;160:156–60.
Stusser R, Paz G, Ortega M, Pineda S, Infante O, Martin P, et al. Risk of low birth weight in the Plaza de la Habana region. Bol Oficina Sanit Panam. 1993;114:229–41.
Jadsri S, Jadsri C. Parental smoking habits and infant birth weight. Asia Pac J Public Health. 1995;8:134–7.
Alonso J, Sosa C, Verde ME, Balsamo A, Moraes M, Zolessi M, et al. Risk factors for term small for gestational age. A case-control study in an uruguayan population. Int J Gynecol Obstet. 2012;119:S276.
Infante-Rivard C. Caffeine intake and small-for-gestational-age birth: modifying effects of xenobiotic-metabolising genes and smoking. Paediatr Perinat Epidemiol. 2007;21:300–9.
Leonardi-Bee J, Britton J, Venn A. Secondhand smoke and adverse fetal outcomes in nonsmoking pregnant women: a meta-analysis. Pediatrics. 2011;127:734–41.
Barker DJ. Childhood causes of adult diseases. Arch Dis Child. 1988;63:867–9.
Nathanielsz PW. Fetal programming: how the quality of fetal life alters biology for a lifetime. NeoReviews. 2000;1:e126–31.
Eriksson M, Wallander MA, Krakau I, Wedel H, Svardsudd K. The impact of birth weight on coronary heart disease morbidity and mortality in a birth cohort followed up for 85 years: a population-based study of men born in 1913. J Intern Med. 2004;256:472–81.
Ong KK, Dunger DB. Birth weight, infant growth and insulin resistance. Eur J Endocrinol. 2004;151(Suppl 3):U131–9.
Levy-Marchal C, Jaquet D. Long-term metabolic consequences of being born small for gestational age. Pediatr Diabetes. 2004;5:147–53.
Grosso LM, Triche EW, Belanger K, Benowitz NL, Holford TR, Bracken MB. Caffeine metabolites in umbilical cord blood, cytochrome P-450 1A2 activity, and intrauterine growth restriction. Am J Epidemiol. 2006;163:1035–41.
Cook DG, Peacock JL, Feyerabend C, Carey IM, Jarvis MJ, Anderson HR, et al. Relation of caffeine intake and blood caffeine concentrations during pregnancy to fetal growth: prospective population based study. BMJ. 1996;313:1358–62.
Maslova E, Bhattacharya S, Lin SW, Michels KB. Caffeine consumption during pregnancy and risk of preterm birth: a meta-analysis. Am J Clin Nutr. 2010;92:1120–32.
Leviton A, Cowan L. A review of the literature relating caffeine consumption by women to their risk of reproductive hazards. Food Chem Toxicol. 2002;40:1271–310.
Peck JD, Leviton A, Cowan LD. A review of the epidemiologic evidence concerning the reproductive health effects of caffeine consumption: a 2000–2009 update. Food Chem Toxicol. 2010;48:2549–76.
Landi MT, Sinha R, Lang NP, Kadlubar FF. Human cytochrome P4501A2. IARC scientific publications 1999. Issue 148.
Klebanoff MA, Levine RJ, DerSimonian R, Clemens JD, Wilkins DG. Maternal serum paraxanthine, a caffeine metabolite, and the risk of spontaneous abortion. N Engl J Med. 1999;341:1639–44.
Klebanoff MA, Levine RJ, Clemens JD, Wilkins DG. Maternal serum caffeine metabolites and small-for-gestational age birth. Am J Epidemiol. 2002;155:32–7.
Acknowledgments
This review was funded by the Food Standards Agency (Contract T01033). We would like to acknowledge the contribution of Alastair Hay, Kay White and Nigel Simpson from the University of Leeds for comments on preliminary analyses and Gary Welsh from the Food Standards Agency Information Services and the University of Leeds Health Sciences Library for assistance with the literature searches.
Conflict of interests
The authors have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Greenwood, D.C., Thatcher, N.J., Ye, J. et al. Caffeine intake during pregnancy and adverse birth outcomes: a systematic review and dose–response meta-analysis. Eur J Epidemiol 29, 725–734 (2014). https://doi.org/10.1007/s10654-014-9944-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-014-9944-x