Summary
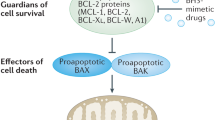
Defective apoptosis is a fundamental hallmark feature of CLL biology and is a major target of cancer therapy development. High levels of Bcl-2 family anti-apoptotic proteins are considered primarily responsible for inhibiting apoptosis in CLL cells. While several approaches were considered to selectively inhibit Bcl-2 family anti-apoptotic proteins, the discovery that gossypol binds and antagonizes anti-apoptotic effect of Bcl-2 family proteins was a major breakthrough in identifying specific Bcl-2 antagonists. The concept of mimicking BH3 domain emphasized the importance of Bcl-2 family-targeted therapy that can modulate the function of anti-apoptotic proteins. Although parent compound gossypol did not sustain in the clinic, its structural modifications led to the development of additional analogues that demonstrated improved efficacy and reduced toxicity in preclinical and clinical investigations. Proof of concept of this hypothesis was demonstrated by structure based BH3 mimetic ABT-737 that has shown greater cytotoxicity towards CLL cells both in pre-clinical models and clinical trials. Its oral compound ABT-263 has demonstrated the substantial susceptibility of chronic lymphocytic leukemia cells through Bcl-2 inhibition. Collectively, results of a Phase I Study of Navitoclax (ABT-263) in patients with relapsed or refractory disease warrants Bcl-2 as a valid therapeutic target in CLL. Importantly, molecules that mimic pro-apoptotic BH3 domains represent a direct approach to overcoming the protective effects of anti-apoptotic proteins such as Mcl-1, Bcl-2 and Bcl-XL.



Similar content being viewed by others
References
Cragg GM, Newman DJ (2005) Plants as a source of anti-cancer agents. J Ethnopharmacol 100:72–79
Rowinsky EK, Onetto N, Canetta RM, Arbuck SG (1992) Taxol: the first of the taxanes, an important new class of antitumor agents. Semin Oncol 19:646–662
Wani MC, Taylor HL, Wall ME, Coggon P, McPhail AT (1971) Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc 93:2325–2327
Stahelin H (1973) Activity of a new glycosidic lignan derivative (VP 16-213) related to podophyllotoxin in experimental tumors. Eur J Cancer 9:215–221
Harvey AL (1999) Medicines from nature: are natural products still relevant to drug discovery? Trends Pharmacol Sci 20:196–198
Powell RG, Weisleder D, Smith CR, Jr., Rohwedder WK (1970) Structures of harringtonine, isoharringtonine, and homoharringtonine. Tetrahedron Lett 815–818
Kantarjian HM, O’Brien S, Anderlini P, Talpaz M (1996) Treatment of myelogenous leukemia: current status and investigational options. Blood 87:3069–3081
Kelland LR (2000) Flavopiridol, the first cyclin-dependent kinase inhibitor to enter the clinic: current status. Expert Opin Investig Drugs 9:2903–2911
Christian MC, Pluda JM, Ho PT, Arbuck SG, Murgo AJ, Sausville EA (1997) Promising new agents under development by the Division of Cancer Treatment, Diagnosis, and Centers of the National Cancer Institute. Semin Oncol 24:219–240
Meijer L, Raymond E (2003) Roscovitine and other purines as kinase inhibitors. From starfish oocytes to clinical trials. Acc Chem Res 36:417–425
Pettit GR, Singh SB, Niven ML, Hamel E, Schmidt JM (1987) Isolation, structure, and synthesis of combretastatins A-1 and B-1, potent new inhibitors of microtubule assembly, derived from Combretum caffrum. J Nat Prod 50:119–131
Jd E (1958) Total synthesis of gossypol. J Am Chem Soc 80:3798–3799
Adams RMR, Geissman TA, Butterbaugh DJ, Kirkpatrick EC (1938) Structure of gossypol. An interpretation of its reaction. J Am Chem Soc 60:2193–2203
Wu D (1989) An overview of the clinical pharmacology and therapeutic potential of gossypol as a male contraceptive agent and in gynaecological disease. Drugs 38:333–341
Waites GM, Wang C, Griffin PD (1998) Gossypol: reasons for its failure to be accepted as a safe, reversible male antifertility drug. Int J Androl 21:8–12
Coutinho EM (2002) Gossypol: a contraceptive for men. Contraception 65:259–263
Zhang M, Liu H, Guo R, Ling Y, Wu X, Li B et al (2003) Molecular mechanism of gossypol-induced cell growth inhibition and cell death of HT-29 human colon carcinoma cells. Biochem Pharmacol 66:93–103
Zhang M, Liu H, Tian Z, Griffith BN, Ji M, Li QQ (2007) Gossypol induces apoptosis in human PC-3 prostate cancer cells by modulating caspase-dependent and caspase-independent cell death pathways. Life Sci 80:767–774
Oliver CL, Bauer JA, Wolter KG, Ubell ML, Narayan A, O’Connell KM et al (2004) In vitro effects of the BH3 mimetic, (-)-gossypol, on head and neck squamous cell carcinoma cells. Clin Cancer Res 10:7757–7763
Van Poznak C, Seidman AD, Reidenberg MM, Moasser MM, Sklarin N, Van Zee K et al (2001) Oral gossypol in the treatment of patients with refractory metastatic breast cancer: a phase I/II clinical trial. Breast Cancer Res Treat 66:239–248
Tso WW, Lee CS (1982) Lactate dehydrogenase-X: an isozyme particularly sensitive to gossypol inhibition. Int J Androl 5:205–209
Teng CS (1995) Gossypol-induced apoptotic DNA fragmentation correlates with inhibited protein kinase C activity in spermatocytes. Contraception 52:389–395
Wang Y, Rao PN (1984) Effect of gossypol on DNA synthesis and cell cycle progression of mammalian cells in vitro. Cancer Res 44:35–38
Ligueros M, Jeoung D, Tang B, Hochhauser D, Reidenberg MM, Sonenberg M (1997) Gossypol inhibition of mitosis, cyclin D1 and Rb protein in human mammary cancer cells and cyclin-D1 transfected human fibrosarcoma cells. Br J Cancer 76:21–28
Shidaifat F, Canatan H, Kulp SK, Sugimoto Y, Zhang Y, Brueggemeier RW et al (1997) Gossypol arrests human benign prostatic hyperplastic cell growth at G0/G1 phase of the cell cycle. Anticancer Res 17:1003–1009
Hou DX, Uto T, Tong X, Takeshita T, Tanigawa S, Imamura I et al (2004) Involvement of reactive oxygen species-independent mitochondrial pathway in gossypol-induced apoptosis. Arch Biochem Biophys 428:179–187
Chang JS, Hsu YL, Kuo PL, Chiang LC, Lin CC (2004) Upregulation of Fas/Fas ligand-mediated apoptosis by gossypol in an immortalized human alveolar lung cancer cell line. Clin Exp Pharmacol Physiol 31:716–722
Lei X, Chen Y, Du G, Yu W, Wang X, Qu H et al (2006) Gossypol induces Bax/Bak-independent activation of apoptosis and cytochrome c release via a conformational change in Bcl-2. FASEB J 20:2147–2149
Moon DO, Kim MO, Lee JD, Kim GY (2008) Gossypol suppresses NF-kappaB activity and NF-kappaB-related gene expression in human leukemia U937 cells. Cancer Lett 264:192–200
Lian J, Wu X, He F, Karnak D, Tang W, Meng Y et al (2011) A natural BH3 mimetic induces autophagy in apoptosis-resistant prostate cancer via modulating Bcl-2-Beclin1 interaction at endoplasmic reticulum. Cell Death Differ 18:60–71
Gao P, Bauvy C, Souquere S, Tonelli G, Liu L, Zhu Y et al (2010) The Bcl-2 homology domain 3 mimetic gossypol induces both Beclin 1-dependent and Beclin 1-independent cytoprotective autophagy in cancer cells. J Biol Chem 285:25570–25581
Flack MR, Pyle RG, Mullen NM, Lorenzo B, Wu YW, Knazek RA et al (1993) Oral gossypol in the treatment of metastatic adrenal cancer. J Clin Endocrinol Metab 76:1019–1024
Stein RC, Joseph AE, Matlin SA, Cunningham DC, Ford HT, Coombes RC (1992) A preliminary clinical study of gossypol in advanced human cancer. Cancer Chemother Pharmacol 30:480–482
Bushunow P, Reidenberg MM, Wasenko J, Winfield J, Lorenzo B, Lemke S et al (1999) Gossypol treatment of recurrent adult malignant gliomas. J Neurooncol 43:79–86
Reed JC, Pellecchia M (2005) Apoptosis-based therapies for hematologic malignancies. Blood 106:408–418
Kitada S, Leone M, Sareth S, Zhai D, Reed JC, Pellecchia M (2003) Discovery, characterization, and structure-activity relationships studies of proapoptotic polyphenols targeting B-cell lymphocyte/leukemia-2 proteins. J Med Chem 46:4259–4264
Meng Y, Li Y, Li J, Li H, Fu J, Liu Y et al (2007) (-)Gossypol and its combination with imatinib induce apoptosis in human chronic myeloid leukemic cells. Leuk Lymphoma 48:2204–2212
Balakrishnan K, Wierda WG, Keating MJ, Gandhi V (2008) Gossypol, a BH3 mimetic, induces apoptosis in chronic lymphocytic leukemia cells. Blood 112:1971–1980
Zhai D, Jin C, Satterthwait AC, Reed JC (2006) Comparison of chemical inhibitors of antiapoptotic Bcl-2-family proteins. Cell Death Differ 13:1419–1421
Oliver CL, Miranda MB, Shangary S, Land S, Wang S, Johnson DE (2005) (-)-Gossypol acts directly on the mitochondria to overcome Bcl-2- and Bcl-X(L)-mediated apoptosis resistance. Mol Cancer Ther 4:23–31
Becattini B, Kitada S, Leone M, Monosov E, Chandler S, Zhai D et al (2004) Rational design and real time, in-cell detection of the proapoptotic activity of a novel compound targeting Bcl-X(L). Chem Biol 11:389–395
Ross AJ, Waymire KG, Moss JE, Parlow AF, Skinner MK, Russell LD et al (1998) Testicular degeneration in Bclw-deficient mice. Nat Genet 18:251–256
Johnsen O, Mas Diaz J, Eliasson R (1982) Gossypol; a potent inhibitor of human sperm acrosomal proteinase. Int J Androl 5:636–640
Qiu J, Levin LR, Buck J, Reidenberg MM (2002) Different pathways of cell killing by gossypol enantiomers. Exp Biol Med (Maywood) 227:398–401
Wang G, Nikolovska-Coleska Z, Yang CY, Wang R, Tang G, Guo J et al (2006) Structure-based design of potent small-molecule inhibitors of anti-apoptotic Bcl-2 proteins. J Med Chem 49:6139–6142
Balakrishnan K, Burger JA, Wierda WG, Gandhi V (2009) AT-101 induces apoptosis in CLL B cells and overcomes stromal cell-mediated Mcl-1 induction and drug resistance. Blood 113:149–153
Vogler M, Butterworth M, Majid A, Walewska RJ, Sun XM, Dyer MJ et al (2009) Concurrent up-regulation of BCL-XL and BCL2A1 induces approximately 1000-fold resistance to ABT-737 in chronic lymphocytic leukemia. Blood 113:4403–4413
Burger JA, Tsukada N, Burger M, Zvaifler NJ, Dell’Aquila M, Kipps TJ (2000) Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood 96:2655–2663
Pedersen IM, Kitada S, Leoni LM, Zapata JM, Karras JG, Tsukada N et al (2002) Protection of CLL B cells by a follicular dendritic cell line is dependent on induction of Mcl-1. Blood 100:1795–1801
Paoluzzi L, Gonen M, Gardner JR, Mastrella J, Yang D, Holmlund J et al (2008) Targeting Bcl-2 family members with the BH3 mimetic AT-101 markedly enhances the therapeutic effects of chemotherapeutic agents in in vitro and in vivo models of B-cell lymphoma. Blood 111:5350–5358
Hu W, Wang F, Tang J, Liu X, Yuan Z, Nie C et al (2012) Proapoptotic protein Smac mediates apoptosis in cisplatin-resistant ovarian cancer cells when treated with the anti-tumor agent AT101. J Biol Chem 287:68–80
Baggstrom MQ, Qi Y, Koczywas M, Argiris A, Johnson EA, Millward MJ et al (2011) A phase II study of AT-101 (Gossypol) in chemotherapy-sensitive recurrent extensive-stage small cell lung cancer. J Thorac Oncol 6:1757–1760
Ready N, Karaseva NA, Orlov SV, Luft AV, Popovych O, Holmlund JT et al (2011) Double-blind, placebo-controlled, randomized phase 2 study of the proapoptotic agent AT-101 plus docetaxel, in second-line non-small cell lung cancer. J Thorac Oncol 6:781–785
Wei J, Rega MF, Kitada S, Yuan H, Zhai D, Risbood P et al (2009) Synthesis and evaluation of Apogossypol atropisomers as potential Bcl-xL antagonists. Cancer Lett 273:107–113
Kitada S, Kress CL, Krajewska M, Jia L, Pellecchia M, Reed JC (2008) Bcl-2 antagonist apogossypol (NSC736630) displays single-agent activity in Bcl-2-transgenic mice and has superior efficacy with less toxicity compared with gossypol (NSC19048). Blood 111:3211–3219
Jia L, Coward LC, Kerstner-Wood CD, Cork RL, Gorman GS, Noker PE et al (2008) Comparison of pharmacokinetic and metabolic profiling among gossypol, apogossypol and apogossypol hexaacetate. Cancer Chemother Pharmacol 61:63–73
Wei J, Kitada S, Rega MF, Emdadi A, Yuan H, Cellitti J et al (2009) Apogossypol derivatives as antagonists of antiapoptotic Bcl-2 family proteins. Mol Cancer Ther 8:904–913
Wei J, Kitada S, Rega MF, Stebbins JL, Zhai D, Cellitti J et al (2009) Apogossypol derivatives as pan-active inhibitors of antiapoptotic B-cell lymphoma/leukemia-2 (Bcl-2) family proteins. J Med Chem 52:4511–4523
Dash R, Azab B, Quinn BA, Shen X, Wang XY, Das SK et al (2011) Apogossypol derivative BI-97C1 (Sabutoclax) targeting Mcl-1 sensitizes prostate cancer cells to mda-7/IL-24-mediated toxicity. Proc Natl Acad Sci U S A 108:8785–8790
Azab B, Dash R, Das SK, Bhutia SK, Shen XN, Quinn BA et al (2012) Enhanced delivery of mda-7/IL-24 using a serotype chimeric adenovirus (Ad.5/3) in combination with the Apogossypol derivative BI-97C1 (Sabutoclax) improves therapeutic efficacy in low CAR colorectal cancer cells. J Cell Physiol 227:2145–2153
Gilbert NE, O’Reilly JE, Chang CJ, Lin YC, Brueggemeier RW (1995) Antiproliferative activity of gossypol and gossypolone on human breast cancer cells. Life Sci 57:61–67
Dao VT, Dowd MK, Martin MT, Gaspard C, Mayer M, Michelot RJ (2004) Cytotoxicity of enantiomers of gossypol Schiff’s bases and optical stability of gossypolone. Eur J Med Chem 39:619–624
Zhan Y, Jia G, Wu D, Xu Y, Xu L (2009) Design and synthesis of a gossypol derivative with improved antitumor activities. Arch Pharm (Weinheim) 342:223–229
Mohammad RM YD, Chen B, Aboukameel A, Chen J, Nikolovska-Coleska Z, Al-Katib A, Wang S (2006) ApoG2, a potent, non-toxic small-molecule inhibitor of Bcl-2 family: a preclinical trial in lymphoma Proc Amer Assoc Cancer Res;Abstract #1335
Arnold AA, Aboukameel A, Chen J, Yang D, Wang S, Al-Katib A et al (2008) Preclinical studies of Apogossypolone: a new nonpeptidic pan small-molecule inhibitor of Bcl-2, Bcl-XL and Mcl-1 proteins in Follicular Small Cleaved Cell Lymphoma model. Mol Cancer 7:20
Sun J, Li ZM, Hu ZY, Lin XB, Zhou NN, Xian LJ et al (2008) ApoG2 inhibits antiapoptotic Bcl-2 family proteins and induces mitochondria-dependent apoptosis in human lymphoma U937 cells. Anticancer Drugs 19:967–974
Balakrishnan K, Aggarwal S, Wierda W, Gandhi V (2013) Bax and Bak are required for apogossypolone, a BH3-mimetic, induced apoptosis in chronic lymphocytic leukemia cells. Leuk Lymphoma 54:1097–1100
Sun Y, Wu J, Aboukameel A, Banerjee S, Arnold AA, Chen J et al (2008) Apogossypolone, a nonpeptidic small molecule inhibitor targeting Bcl-2 family proteins, effectively inhibits growth of diffuse large cell lymphoma cells in vitro and in vivo. Cancer Biol Ther 7:1418–1426
Hu ZY, Sun J, Zhu XF, Yang D, Zeng YX (2009) ApoG2 induces cell cycle arrest of nasopharyngeal carcinoma cells by suppressing the c-Myc signaling pathway. J Transl Med 7:74
Wei J, Kitada S, Stebbins JL, Placzek W, Zhai D, Wu B et al (2010) Synthesis and biological evaluation of Apogossypolone derivatives as pan-active inhibitors of antiapoptotic B-cell lymphoma/leukemia-2 (Bcl-2) family proteins. J Med Chem 53:8000–8011
Nguyen M, Marcellus RC, Roulston A, Watson M, Serfass L, Murthy Madiraju SR et al (2007) Small molecule obatoclax (GX15-070) antagonizes MCL-1 and overcomes MCL-1-mediated resistance to apoptosis. Proc Natl Acad Sci U S A 104:19512–19517
Trudel S, Li ZH, Rauw J, Tiedemann RE, Wen XY, Stewart AK (2007) Preclinical studies of the pan-Bcl inhibitor obatoclax (GX015-070) in multiple myeloma. Blood 109:5430–5438
Konopleva M, Watt J, Contractor R, Tsao T, Harris D, Estrov Z et al (2008) Mechanisms of antileukemic activity of the novel Bcl-2 homology domain-3 mimetic GX15-070 (obatoclax). Cancer Res 68:3413–3420
Campas C, Cosialls AM, Barragan M, Iglesias-Serret D, Santidrian AF, Coll-Mulet L et al (2006) Bcl-2 inhibitors induce apoptosis in chronic lymphocytic leukemia cells. Exp Hematol 34:1663–1669
Perez-Galan P, Roue G, Lopez-Guerra M, Nguyen M, Villamor N, Montserrat E et al (2008) BCL-2 phosphorylation modulates sensitivity to the BH3 mimetic GX15-070 (Obatoclax) and reduces its synergistic interaction with bortezomib in chronic lymphocytic leukemia cells. Leukemia 22:1712–1720
Perez-Galan P, Roue G, Villamor N, Campo E, Colomer D (2007) The BH3-mimetic GX15-070 synergizes with bortezomib in mantle cell lymphoma by enhancing Noxa-mediated activation of Bak. Blood 109:4441–4449
Mitchell C, Yacoub A, Hossein H, Martin AP, Bareford MD, Eulitt P et al (2010) Inhibition of MCL-1 in breast cancer cells promotes cell death in vitro and in vivo. Cancer Biol Ther 10:903–917
Huang S, Okumura K, Sinicrope FA (2009) BH3 mimetic obatoclax enhances TRAIL-mediated apoptosis in human pancreatic cancer cells. Clin Cancer Res 15:150–159
Wei Y, Kadia T, Tong W, Zhang M, Jia Y, Yang H et al (2010) The combination of a histone deacetylase inhibitor with the BH3-mimetic GX15-070 has synergistic antileukemia activity by activating both apoptosis and autophagy. Autophagy 6:976–978
Martin AP, Park MA, Mitchell C, Walker T, Rahmani M, Thorburn A et al (2009) BCL-2 family inhibitors enhance histone deacetylase inhibitor and sorafenib lethality via autophagy and overcome blockade of the extrinsic pathway to facilitate killing. Mol Pharmacol 76:327–341
Rahmani M, Aust MM, Attkisson E, Williams DC, Jr (2012) Ferreira-Gonzalez A, Grant S. Inhibition of Bcl-2 anti-apoptotic members by obatoclax potently enhances sorafenib-induced apoptosis in human myeloid leukemia cells through a Bim-dependent process. Blood
Martinez-Paniagua MA, Baritaki S, Huerta-Yepez S, Ortiz-Navarrete VF, Gonzalez-Bonilla C, Bonavida B et al (2011) Mcl-1 and YY1 inhibition and induction of DR5 by the BH3-mimetic Obatoclax (GX15-070) contribute in the sensitization of B-NHL cells to TRAIL apoptosis. Cell Cycle 10:2792–2805
Schimmer AD, O’Brien S, Kantarjian H, Brandwein J, Cheson BD, Minden MD et al (2008) A phase I study of the pan bcl-2 family inhibitor obatoclax mesylate in patients with advanced hematologic malignancies. Clin Cancer Res 14:8295–8301
O’Brien SM, Claxton DF, Crump M, Faderl S, Kipps T, Keating MJ et al (2009) Phase I study of obatoclax mesylate (GX15-070), a small molecule pan-Bcl-2 family antagonist, in patients with advanced chronic lymphocytic leukemia. Blood 113:299–305
Paik PK, Rudin CM, Pietanza MC, Brown A, Rizvi NA, Takebe N et al (2011) A phase II study of obatoclax mesylate, a Bcl-2 antagonist, plus topotecan in relapsed small cell lung cancer. Lung Cancer 74:481–485
Oki Y, Copeland A, Hagemeister F, Fayad LE, Fanale M, Romaguera J et al (2012) Experience with obatoclax mesylate (GX15-070), a small molecule pan-Bcl-2 family antagonist in patients with relapsed or refractory classical Hodgkin lymphoma. Blood 119:2171–2172
Oltersdorf T, Elmore SW, Shoemaker AR, Armstrong RC, Augeri DJ, Belli BA et al (2005) An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature 435:677–681
Vogler M, Furdas SD, Jung M, Kuwana T, Dyer MJ, Cohen GM (2010) Diminished sensitivity of chronic lymphocytic leukemia cells to ABT-737 and ABT-263 due to albumin binding in blood. Clin Cancer Res 16:4217–4225
Vogler M, Weber K, Dinsdale D, Schmitz I, Schulze-Osthoff K, Dyer MJ et al (2009) Different forms of cell death induced by putative BCL2 inhibitors. Cell Death Differ 16:1030–1039
Touzeau C, Dousset C, Bodet L, Gomez-Bougie P, Bonnaud S, Moreau A et al (2011) ABT-737 induces apoptosis in mantle cell lymphoma cells with a Bcl-2high/Mcl-1low profile and synergizes with other antineoplastic agents. Clin Cancer Res 17:5973–5981
Kuroda J, Puthalakath H, Cragg MS, Kelly PN, Bouillet P, Huang DC et al (2006) Bim and Bad mediate imatinib-induced killing of Bcr/Abl + leukemic cells, and resistance due to their loss is overcome by a BH3 mimetic. Proc Natl Acad Sci U S A 103:14907–14912
Konopleva M, Contractor R, Tsao T, Samudio I, Ruvolo PP, Kitada S et al (2006) Mechanisms of apoptosis sensitivity and resistance to the BH3 mimetic ABT-737 in acute myeloid leukemia. Cancer Cell 10:375–388
Del Gaizo Moore V, Brown JR, Certo M, Love TM, Novina CD, Letai A (2007) Chronic lymphocytic leukemia requires BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2 antagonist ABT-737. J Clin Invest 117:112–121
Vogler M, Dinsdale D, Sun XM, Young KW, Butterworth M, Nicotera P et al (2008) A novel paradigm for rapid ABT-737-induced apoptosis involving outer mitochondrial membrane rupture in primary leukemia and lymphoma cells. Cell Death Differ 15:820–830
Kline MP, Rajkumar SV, Timm MM, Kimlinger TK, Haug JL, Lust JA et al (2007) ABT-737, an inhibitor of Bcl-2 family proteins, is a potent inducer of apoptosis in multiple myeloma cells. Leukemia 21:1549–1560
Bodet L, Gomez-Bougie P, Touzeau C, Dousset C, Descamps G, Maiga S et al (2011) ABT-737 is highly effective against molecular subgroups of multiple myeloma. Blood 118:3901–3910
Kang MH, Kang YH, Szymanska B, Wilczynska-Kalak U, Sheard MA, Harned TM et al (2007) Activity of vincristine, L-ASP, and dexamethasone against acute lymphoblastic leukemia is enhanced by the BH3-mimetic ABT-737 in vitro and in vivo. Blood 110:2057–2066
Del Gaizo Moore V, Schlis KD, Sallan SE, Armstrong SA, Letai A (2008) BCL-2 dependence and ABT-737 sensitivity in acute lymphoblastic leukemia. Blood 111:2300–2309
Chen S, Dai Y, Pei XY, Grant S (2009) Bim up-regulation by histone deacetylase inhibitors mediates interactions with the Bcl-2 antagonist ABT-737: evidence for distinct roles for Bcl-2, Bcl-xL and Mcl-1. Mol Cell Biol
van Delft MF, Wei AH, Mason KD, Vandenberg CJ, Chen L, Czabotar PE et al (2006) The BH3 mimetic ABT-737 targets selective Bcl-2 proteins and efficiently induces apoptosis via Bak/Bax if Mcl-1 is neutralized. Cancer cell 10:389–399
Lin X, Morgan-Lappe S, Huang X, Li L, Zakula DM, Vernetti LA et al (2007) ‘Seed’ analysis of off-target siRNAs reveals an essential role of Mcl-1 in resistance to the small-molecule Bcl-2/Bcl-XL inhibitor ABT-737. Oncogene 26:3972–3979
Inuzuka H, Shaik S, Onoyama I, Gao D, Tseng A, Maser RS et al (2011) SCF(FBW7) regulates cellular apoptosis by targeting MCL1 for ubiquitylation and destruction. Nature 471:104–109
Schwickart M, Huang X, Lill JR, Liu J, Ferrando R, French DM et al (2010) Deubiquitinase USP9X stabilizes MCL1 and promotes tumour cell survival. Nature 463:103–107
Paoluzzi L, Gonen M, Bhagat G, Furman RR, Gardner JR, Scotto L et al (2008) The BH3-only mimetic ABT-737 synergizes the antineoplastic activity of proteasome inhibitors in lymphoid malignancies. Blood 112:2906–2916
Chen S, Dai Y, Pei XY, Grant S (2009) Bim upregulation by histone deacetylase inhibitors mediates interactions with the Bcl-2 antagonist ABT-737: evidence for distinct roles for Bcl-2, Bcl-xL, and Mcl-1. Mol Cell Biol 29:6149–6169
Mason KD, Khaw SL, Rayeroux KC, Chew E, Lee EF, Fairlie WD et al (2009) The BH3 mimetic compound, ABT-737, synergizes with a range of cytotoxic chemotherapy agents in chronic lymphocytic leukemia. Leukemia 23:2034–2041
Kuroda J, Kimura S, Andreeff M, Ashihara E, Kamitsuji Y, Yokota A et al (2008) ABT-737 is a useful component of combinatory chemotherapies for chronic myeloid leukaemias with diverse drug-resistance mechanisms. Br J Haematol 140:181–190
Chen S, Dai Y, Harada H, Dent P, Grant S (2007) Mcl-1 down-regulation potentiates ABT-737 lethality by cooperatively inducing Bak activation and Bax translocation. Cancer Res 67:782–791
Kojima K, Duvvuri S, Ruvolo V, Samaniego F, Younes A, Andreeff M (2012) Decreased sensitivity of 17p-deleted chronic lymphocytic leukemia cells to a small molecule BCL-2 antagonist ABT-737. Cancer 118:1023–1031
Tromp JM, Geest CR, Breij EC, Elias JA, van Laar J, Luijks DM et al (2012) Tipping the Noxa/Mcl-1 balance overcomes ABT-737 resistance in chronic lymphocytic leukemia. Clin Cancer Res 18:487–498
Merino D, Khaw SL, Glaser SP, Anderson DJ, Belmont LD, Wong C, et al (2012) Bcl-2, Bcl-xL and Bcl-w are not equivalent targets of ABT-737 and Navitoclax (ABT-263) in lymphoid and leukemic cells. Blood
Tse C, Shoemaker AR, Adickes J, Anderson MG, Chen J, Jin S et al (2008) ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res 68:3421–3428
Shoemaker AR, Mitten MJ, Adickes J, Ackler S, Refici M, Ferguson D et al (2008) Activity of the Bcl-2 family inhibitor ABT-263 in a panel of small cell lung cancer xenograft models. Clin Cancer Res 14:3268–3277
Ackler S, Xiao Y, Mitten MJ, Foster K, Oleksijew A, Refici M et al (2008) ABT-263 and rapamycin act cooperatively to kill lymphoma cells in vitro and in vivo. Mol Cancer Ther 7:3265–3274
Gandhi L, Camidge DR, Ribeiro de Oliveira M, Bonomi P, Gandara D, Khaira D et al (2011) Phase I study of Navitoclax (ABT-263), a novel Bcl-2 family inhibitor, in patients with small-cell lung cancer and other solid tumors. J Clin Oncol 29:909–916
Wilson WH, O’Connor OA, Czuczman MS, LaCasce AS, Gerecitano JF, Leonard JP et al (2010) Navitoclax, a targeted high-affinity inhibitor of BCL-2, in lymphoid malignancies: a phase 1 dose-escalation study of safety, pharmacokinetics, pharmacodynamics, and antitumour activity. Lancet Oncol 11:1149–1159
Rudin CM, Hann CL, Garon EB, Ribeiro de Oliveira M, Bonomi PD, Camidge DR, et al (2012) Phase II study of single-agent navitoclax (ABT-263) and biomarker correlates in patients with relapsed small cell lung cancer. Clin Cancer Res
Roberts AW, Seymour JF, Brown JR, Wierda WG, Kipps TJ, Khaw SL et al (2012) Substantial susceptibility of chronic lymphocytic leukemia to BCL2 inhibition: results of a phase I study of navitoclax in patients with relapsed or refractory disease. J Clin Oncol 30:488–496
Souers AJ, Leverson JD, Boghaert ER, Ackler SL, Catron ND, Chen J et al (2013) ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med 19:202–208
Doi K, Li R, Sung SS, Wu H, Liu Y, Manieri W et al (2012) Discovery of marinopyrrole A (maritoclax) as a selective Mcl-1 antagonist that overcomes ABT-737 resistance by binding to and targeting Mcl-1 for proteasomal degradation. J Biol Chem 287:10224–10235
Wei J, Stebbins JL, Kitada S et al (2010) BI-97C1, an optically pure Apogossypol derivative as pan-active inhibitor of antiapoptotic B-cell lymphoma/leukemia-2 (Bcl-2) family proteins. J Med Chem 53(10):4166–4176
Rudin CM, Hann CL, Garon EB et al (2012) Phase II Study of Single-Agent Navitoclax (ABT-263) and Biomarker Correlates in Patients with Relapsed Small Cell Lung Cancer. Clin Cancer Res 18(11):3163–3169
Stewart ML, Fire E, Keating AE, Walensky LD (2010) The MCL-1 BH3 helix is an exclusive MCL-1 inhibitor and apoptosis sensitizer. Nat Chem Biol 6(8):595–601
Lee EF, Czabotar PE, van Delft MF, et al (2008) A novel BH3 ligand that selectively targets Mcl-1 reveals that apoptosis can proceed without Mcl-1 degradation. J Cell Biol 180(2):341–355
Acknowledgments
This work is supported in part by a CLL Consortium grant CA81534 from NCI and a CLL Global Research Foundation grant.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balakrishnan, K., Gandhi, V. Bcl-2 antagonists: a proof of concept for CLL therapy. Invest New Drugs 31, 1384–1394 (2013). https://doi.org/10.1007/s10637-013-0002-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-013-0002-4