Abstract
Introduction
Clostridium difficile is the most commonly isolated stool pathogen in inflammatory bowel disease (IBD). Traditional risk factors for C. difficile may not exist in patients with IBD, and no prior studies have assessed the risk factors for the isolation of C. difficile in both symptomatic and asymptomatic IBD outpatients.
Methods
We prospectively recruited consecutive IBD patients presenting to our outpatient clinic between April 2015 and February 2016. We excluded patients with a diverting ostomy or ileoanal pouch. Demographics, healthcare exposures, medical therapies and disease activity were recorded from medical charts or surveys. A rectal swab was performed from which toxigenic culture and PCR analysis for the presence of toxin and fluorescent PCR ribotyping were performed. The primary outcome of interest was isolation of toxigenic C. difficile.
Results
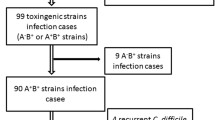
A total of 190 patients were enrolled in this prospective study including 137 (72%) with Crohn’s disease and 53 (28%) with ulcerative colitis. At the time of enrollment, 69 (36%) had clinically active disease. Sixteen (8.4%) patients had toxigenic C. difficile isolated on rectal swab at enrollment and four (2.1%) patients had non-toxigenic C. difficile cultured. Mixed infection with more than one toxigenic isolate was present in 5/16 (33.3%) individuals. Patients with CD with a toxin positive isolate were more likely to have a history of CDI in the past 12 months (40 vs. 11.02%, p = 0.027) and an emergency department visit in the past 12 weeks (50 vs. 20.63%, p = 0.048). In UC, individuals with isolation of C. difficile were more likely to be hospitalized within the past 12 months (66.6 vs. 8.51%, p = 0.003). C. difficile isolation at the time of presentation was not associated with a subsequent disease relapse over a 6-month period in CD (p = 0.557) or UC (p = 0.131).
Conclusion
Healthcare exposures remain a significant risk factor for C. difficile isolation in the IBD population; however, this was not associated with relapse of disease. Further studies assessing the clinical significance of C. difficile isolation is warranted in IBD.

Similar content being viewed by others
Change history
22 August 2018
The original version of the article unfortunately contained an error in a percentage value in Results section of Abstract.
Abbreviations
- CD:
-
Crohn’s disease
- UC:
-
Ulcerative colitis
- IBD:
-
Inflammatory bowel disease
References
Ananthakrishnan AN, McGinley EL, Saeian K, Binion DG. Temporal trends in disease outcomes related to Clostridium difficile infection in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2011;17:976–983.
Bartlett JG. Clostridium difficile and inflammatory bowel disease. Gastroenterology. 1981;80:863–865.
Greenfield C, Aguilar Ramirez JR, Pounder RE, et al. Clostridium difficile and inflammatory bowel disease. Gut. 1983;24:713–717.
Gryboski JD. Clostridium difficile in inflammatory bowel disease relapse. J Pediatr Gastroenterol Nutr. 1991;13:39–41.
Meyer AM, Ramzan NN, Loftus EV Jr., Heigh RI, Leighton JA. The diagnostic yield of stool pathogen studies during relapses of inflammatory bowel disease. J Clin Gastroenterol. 2004;38:772–775.
Meyers S, Mayer L, Bottone E, Desmond E, Janowitz HD. Occurrence of Clostridium difficile toxin during the course of inflammatory bowel disease. Gastroenterology. 1981;80:697–700.
Mylonaki M, Langmead L, Pantes A, Johnson F, Rampton DS. Enteric infection in relapse of inflammatory bowel disease: importance of microbiological examination of stool. Eur J Gastroenterol Hepatol. 2004;16:775–778.
Rolny P, Jarnerot G, Mollby R. Occurrence of Clostridium difficile toxin in inflammatory bowel disease. Scand J Gastroenterol. 1983;18:61–64.
Trnka YM, LaMont JT. Association of Clostridium difficile toxin with symptomatic relapse of chronic inflammatory bowel disease. Gastroenterology. 1981;80:693–696.
Bauer MP, Notermans DW, van Benthem BH, et al. Clostridium difficile infection in Europe: a hospital-based survey. Lancet (London, England). 2011;377:63–73.
Issa M, Vijayapal A, Graham MB, et al. Impact of Clostridium difficile on inflammatory bowel disease. Clin Gastroenterol Hepatol. 2007;5:345–351.
Rodemann JF, Dubberke ER, Reske KA, da Seo H, Stone CD. Incidence of Clostridium difficile infection in inflammatory bowel disease. Clin Gastroenterol Hepatol. 2007;5:339–344.
Nguyen GC, Kaplan GG, Harris ML, Brant SR. A national survey of the prevalence and impact of Clostridium difficile infection among hospitalized inflammatory bowel disease patients. Am J Gastroenterol. 2008;103:1443–1450.
Ananthakrishnan AN, McGinley EL, Binion DG. Excess hospitalisation burden associated with Clostridium difficile in patients with inflammatory bowel disease. Gut. 2008;57:205–210.
Harvey RF, Bradshaw JM. A simple index of Crohn’s-disease activity. Lancet. 1980;315:514.
Walmsley RS, Ayres RCS, Pounder RE, Allan RN. A simple clinical colitis activity index. Gut. 1998;43:29–32.
Sorg JA, Dineen SS. Laboratory maintenance of Clostridium difficile. Curr Protoc Microbiol. 2009;Chapter 9:Unit9A-1.
Walk ST, Micic D, Jain R, et al. Clostridium difficile ribotype does not predict severe infection. Clin Infect Dis. 2012;55:1661–1668.
Alasmari F, Seiler SM, Hink T, Burnham CA, Dubberke ER. Prevalence and risk factors for asymptomatic Clostridium difficile carriage. Clin Infect Dis. 2014;59:216–222.
Miller MA, Louie T, Mullane K, et al. Derivation and validation of a simple clinical bedside score (ATLAS) for Clostridium difficile infection which predicts response to therapy. BMC Infect Dis. 2013;13:148.
Shivashankar R, Khanna S, Kammer PP, et al. Clinical factors associated with development of severe-complicated Clostridium difficile infection. Clin Gastroenterol Hepatol. 2013;11:1466–1471.
Rao K, Micic D, Chenoweth E, et al. Poor functional status as a risk factor for severe Clostridium difficile infection in hospitalized older adults. J Am Geriatr Soc. 2013;61:1738–1742.
Axelrad JE, Joelson A, Nobel YR, et al. Enteric infection in relapse of inflammatory bowel disease: the utility of stool microbial PCR testing. Inflamm Bowel Dis. 2017;23:1034–1039.
Das R, Feuerstadt P, Brandt LJ. Glucocorticoids are associated with increased risk of short-term mortality in hospitalized patients with Clostridium difficile-associated disease. Am J Gastroenterol. 2010;105:2040–2049.
Bossuyt P, Verhaegen J, Van Assche G, Rutgeerts P, Vermeire S. Increasing incidence of Clostridium difficile-associated diarrhea in inflammatory bowel disease. J Crohn’s Colitis. 2009;3:4–7.
Clayton EM, Rea MC, Shanahan F, et al. The vexed relationship between Clostridium difficile and inflammatory bowel disease: an assessment of carriage in an outpatient setting among patients in remission. Am J Gastroenterol. 2009;104:1162–1169.
Dorman SA, Liggoria E, Winn WC Jr., Beeken WL. Isolation of Clostridium difficile from patients with inactive Crohn’s disease. Gastroenterology. 1982;82:1348–1351.
Singh H, Nugent Z, Yu BN, Lix LM, Targownik LE, Bernstein CN. Higher incidence of Clostridium difficile infection among individuals with inflammatory bowel disease. Gastroenterology. 2017;153:430–438.
Behroozian AA, Chludzinski JP, Lo ES, et al. Detection of mixed populations of Clostridium difficile from symptomatic patients using capillary-based polymerase chain reaction ribotyping. Infect Control Hosp Epidemiol. 2013;34:961–966.
Crowther GS, Chilton CH, Todhunter SL, Nicholson S, Freeman J, Wilcox MH. Recurrence of dual-strain Clostridium difficile infection in an in vitro human gut model. J Antimicrob Chemother. 2015;70:2316–2321.
Gracie DJ, Williams CJ, Sood R, et al. Poor correlation between clinical disease activity and mucosal inflammation, and the role of psychological comorbidity, in inflammatory bowel disease. Am J Gastroenterol. 2016;111:541–551.
Rao K, Higgins PD. Epidemiology, diagnosis, and management of Clostridium difficile infection in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2016;22:1744–1754.
Shakir FA, Thompson D, Marlar R, Ali T. A novel use of rectal swab to test for Clostridium difficile infection by real-time PCR. Am J Gastroenterol. 2012;107:1444.
Hughes M, Qazi T, Berg A, et al. Host immune response to Clostridium difficile infection in inflammatory bowel disease patients. Inflamm Bowel Dis. 2016;22:853–861.
Acknowledgments
This work was supported by grants from the National Institutes of Health [Grant Nos. T32DK007074, K08DK090152 (JP), P30DK42086]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No expressed conflicts of interest with respect to the submitted work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Micic, D., Yarur, A., Gonsalves, A. et al. Risk Factors for Clostridium difficile Isolation in Inflammatory Bowel Disease: A Prospective Study. Dig Dis Sci 63, 1016–1024 (2018). https://doi.org/10.1007/s10620-018-4941-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-4941-7