Abstract
Difficulties in the regulation of emotion are hypothesized to play a key role in the development and maintenance of posttraumatic stress disorder (PTSD). The current study used functional magnetic resonance imaging (fMRI) to assess neural activity during task preparation and image presentation during different emotion regulation strategies, cognitive reappraisal and expressive suppression, in PTSD. Patients with combat-related PTSD (n = 18) and combat-exposed controls (n = 27) were instructed to feel, reappraise or suppress their emotional response prior to viewing combat-related images during fMRI, while also providing arousal ratings. In the reappraise condition, patients showed lower medial prefrontal neural activity during task preparation and higher prefrontal neural activity during image presentation, compared with controls. No difference in neural activity was observed between the groups during the feel or suppress conditions, although patients rated images as more arousing than controls across all three conditions. By distinguishing between preparation and active regulation, and between reappraisal and suppression, the current findings reveal greater complexity regarding the dynamics of emotion regulation in PTSD and have implications for our understanding of the etiology and treatment of PTSD.
Similar content being viewed by others
Introduction
Posttraumatic stress disorder (PTSD) is a debilitating psychiatric disorder, which develops following exposure to extreme stress or trauma. Trauma-related symptoms, including hyperarousal, avoidance and intrusive memories, are often experienced in the period immediately following trauma exposure (McFarlane 2000). In the majority of cases these symptoms resolve in the first month following exposure, however a subset of individuals will go on to develop the persistent symptoms that characterize PTSD (Kessler 1995). One reason for the failure of symptoms to resolve over time may be the use of emotion regulation strategies that aim to suppress rather than modify an emotional response, when confronted with negative emotions (Cisler and Olatunji 2012; Gross 1998; Moore et al. 2008; Tull et al. 2007).
Gross’s highly influential process model of emotion regulation distinguishes regulation strategies by the point at which they occur during an unfolding emotional response. At the broadest level, strategies can be divided into early (antecedent-focused) and late (response-focused) (Gross 1998, 2002). Early strategies aim to modify an emotional response before it occurs, such as through reinterpreting the meaning or context of a stimulus—situation-focused cognitive reappraisal—or by modifying one’s perspective—self-focused cognitive reappraisal (Ochsner et al. 2004; Willroth and Hilimire 2016). For example, when faced with a challenging situation, a medical professional may adopt a professional distance to perceive it in a detached and objective manner rather than a personal or emotional one (Doulougeri et al. 2016; Ochsner et al. 2004; Shapiro 2013). In contrast, late strategies attempt to inhibit an emotional response once it has already occurred, for example, by suppressing any outward expression of emotion. Late strategies have been shown to be less effective than early strategies in regulating experienced emotion (Gross 1998).
In PTSD populations, difficulties with emotion regulation were found to be significantly associated with PTSD symptom severity and to partially mediate the relationship between PTSD and related comorbidities (Klemanski et al. 2012). PTSD symptoms have been associated with greater spontaneous use of suppression and less use of reappraisal (Shepherd and Wild 2014), and shifting from suppression- (late) to reappraisal-based (early) emotion regulation strategies has been associated with improvements in treatment outcomes (Price et al. 2006). In addition, therapeutic interventions such as cognitive behavior therapy (CBT) aim to promote emotional regulation self-efficacy, training clients to change their emotions by changing their thoughts through cognitive restructuring (Beck 2011).
At the neural level, cognitive reappraisal and expressive suppression have been shown to be both functionally and temporally distinct. Previous work in healthy individuals (Goldin et al. 2008) has demonstrated that during emotion regulation, patterns of neural activity differ between reappraisal and suppression and change over time, with reappraisal showing a pattern of high initial medial prefrontal neural activity which then decreases over time and suppression showing low initial medial prefrontal neural activity which then increases. At a functional level, the medial prefrontal cortex is involved in multiple processes relevant to emotion regulation, including emotional processing and modulation of the fear response (Diekhof et al. 2011; Etkin et al. 2011; Milad et al. 2007a, b; Northoff et al. 2006). At a structural level, regions within the medial prefrontal cortex show strong connections to the limbic system, including the amygdala (Bush et al. 2000; Devinsky et al. 1995). Goldin et al. conclude that the early activation of prefrontal control regions during reappraisal leads to the effective down-regulation of amygdala and insular reactivity, accompanied by successful regulation of the emotional response and a reduced need for continued cognitive control. Conversely, during suppression, later activation of prefrontal control regions seemingly fails to inhibit the emotional response, leading to a need for sustained activity in these regions and no decrease in amygdala and insular activity.
PTSD patients have previously demonstrated less prefrontal neural activity than controls in response to threat-related stimuli, and this is thought to play a key role in the development and maintenance of PTSD symptoms (Rauch et al. 2006; Shin et al. 2006). Lower prefrontal neural activity has also been observed during the down-regulation of negative emotion in PTSD, in populations as diverse as female victims of sexual violence (New et al. 2009), male combat veterans (Rabinak et al. 2014) and male and female motor vehicle accident victims (Xiong et al. 2013). The above studies did not contrast different types of down-regulation, such as expressive suppression and cognitive reappraisal. However, in a study by Shepherd and Wild (Shepherd and Wild 2014), PTSD patients and trauma-exposed controls were instructed to feel, maintain or decrease their emotional response to negative and neutral images. PTSD symptoms were associated with greater spontaneous use of suppression and less use of reappraisal (Shepherd and Wild 2014). In addition, previous neuroimaging work in PTSD has not distinguished between task preparation and image presentation. It is therefore unclear whether and how neural activity alters during an unfolding emotional response in individuals with PTSD, and whether patterns of change differ between groups in during suppression, as both groups can employ suppression when instructed.
In the current study, we investigate neural and behavioral differences between combat-related PTSD patients and combat-exposed controls during an emotion regulation task. In line with Gross’s process model of emotion regulation, we distinguish between cognitive reappraisal and expressive suppression. In addition, previous work has demonstrated that patterns of neural activity not only differ between reappraisal and suppression, but also change over time (Goldin et al. 2008, 2009), and that physiological and behavioral changes are observable during preparation for emotion regulation (Gross 1998). Gross and colleagues propose that when individuals are aware that they will soon be required to manage their emotions, they “appear to steel themselves”, and that physiological and behavioural changes are indicative of an individual’s efforts to prepare themselves (1998). As such, we also distinguish between preparatory neural activity, during the instruction phase, and active emotion regulation, during the image presentation phase, a distinction that has not previously been made in neuroimaging research on PTSD.
We recruited patients with combat-related PTSD and combat-exposed individuals without psychopathology, and employed a mixed design to compare the effects of cognitive reappraisal and expressive suppression on subjective arousal and neural activity in individuals with and without PTSD. Combat-related images were used to generate a powerful trauma-related negative affective state. We hypothesize that during emotional regulation, patients and controls will differ at both a behavioral and neural level. On the basis of previous literature, one may postulate on a number of potential neural patterns that PTSD patients may demonstrate. Based on neuroimaging work in PTSD, one may expect that prefrontal neural activity will be lower in PTSD patients than controls during all stages and for all forms of emotion regulation (New et al. 2009; Rabinak et al. 2014; Rauch et al. 2006; Xiong et al. 2013). Alternatively, if PTSD is particularly associated with the use of suppression (Shepherd and Wild 2014), then one may expect that when instructed to use reappraisal, PTSD patients will rather demonstrate a pattern similar to suppression in healthy controls, of lower initial prefrontal neural activity which then increases (Goldin et al. 2008). In this case, one would not expect to observe a difference between patients and controls. We aim to clarify whether during cognitive reappraisal, PTSD patients show reduced neural activity across both task preparation and stimulus presentation, or if they present a pattern similar to that of expressive suppression in healthy controls, with lower preparatory neural activity followed by sustained activity during stimulus presentation. At the behavioral level, we hypothesize that PTSD patients will be less effective in down-regulating emotion and will report higher subjective arousal ratings than controls during cognitive reappraisal and expressive suppression.
Methods and Materials
Participants
Eighteen soldiers with combat-related PTSD, prior to onset of therapy, and 27 combat-exposed soldiers without mental illness were recruited from the German Armed Forces. Participants were screened for inclusion and exclusion criteria. Inclusion criteria: all participants were male, had been previously deployed overseas to areas of conflict, and had experienced trauma within the last 2 years, as assessed by the Mental Health Advisory Team Combat Experiences Scale. Clinical psychologists interviewed participants, and for the patient group a diagnosis of PTSD was made using ICD 10 criteria. Exclusion criteria: no participants had MRI contraindications or a history of concussion or traumatic brain imagery, none were using psychotropic medication or had current or previous comorbid Axis II psychiatric disorders (American Psychiatric Association 2000). The mean age of the patient group was 28.3 years (SD = 6.4, ranging between 23 and 52 years) and the mean age of the control group was 32.7 years (SD = 5.9, ranging between 23 and 47 years). Because the patient group was significantly younger than the control group [t(43) = 2.387, p < 0.05], in the subsequent reported neuroimaging analyses, age was included as a covariate of no interest, although repeating the neuroimaging analyses without including age did not change the results.
All participants had completed secondary education, and the proportion of patients (12 of 18) and controls (21 of 27) who had completed additional vocational training was not significantly different [X2(1, N = 45) = 2.143, p = 0.143].
The protocol was approved by the ethics committee of Charité University Clinic, Berlin, Germany, and all subjects gave written informed consent in accordance with the Declaration of Helsinki.
Questionnaires
Prior to neuroimaging, all participants completed a number of questionnaires assessing psychological symptoms and experiences during deployment. Participants completed German versions of the following self-report questionnaires: the Post-traumatic Diagnostic Scale (PDS) (Foa et al. 1997), the Posttraumatic Cognitions Inventory (PTCI) (Foa et al. 1999), the Interpretation of PTSD Symptoms Inventory (IPSI) (Clohessy and Ehlers 1999) and a 33-item questionnaire measuring frequency of combat-related events based on the list of the Mental Health Advisory Team Combat Experiences Scale (MHAT-CES) (Hoge et al. 2004; Mental Health Advisory Team 2006). Independent samples t tests were conducted on questionnaire data. Patients scored significantly higher than controls on PTSD questionnaires (PDS, PTCI & IPSI) but not on combat experiences (MHAT-CES) (see Table 1). MHAT-CES data was missing for four patients.
Scanning Procedure
Magnetic resonance images were acquired using a 3 T Magnetom Tim Trio MRI scanner system (Siemens Medical Systems, Erlangen, Germany) and a 12-channel radiofrequency head coil. Structural images were obtained using a three-dimensional T1-weighted magnetization prepared gradient-echo sequence (MPRAGE) based on the ADNI protocol (http://www.adni-info.org; TR = 2500 ms; TE = 4.77 ms; TI = 1100 ms, acquisition matrix = 256 × 256 × 176, flip angle = 7°; 1 × 1 × 1 mm3 voxel size). Whole-brain functional images were acquired using a T2*-weighted echo-planar-imaging (EPI) sequence sensitive to bold contrast (TR = 2000 ms, TE = 30 ms, image matrix = 72 × 72, FOV = 216 mm, flip angle = 80°, slice thickness = 3.0 mm, distance factor = 20%, slice order = interleaved, voxel size 3 × 3 × 3 mm3, 36 axial slices).
Image Stimuli
Sixty combat images were selected from a larger battery of genuine war photographs provided by the German Armed Forces, taken by soldiers during active duty, mainly in Afghanistan. Combat images were selected as we consider these images to be more salient to our participants than negative images from other potential sources, for example the International Affective Picture System (IAPS) (Lang et al. 2008). Affective valence and arousal of the images were assessed by the experimenters, and images with unpleasant valence and medium arousal were selected. Examples included photos of destroyed vehicles, explosions and soldiers under enemy fire. Images showing explicit scenes of death or injury were excluded to reduce the likelihood of images triggering a flashback in the patient population.
Emotional Regulation Instructions
Prior to magnetic resonance imaging (MRI), participants were instructed in cognitive reappraisal and expressive suppression strategies (see Supplemental Material S1 for original instructions in German and Supplemental Material S2 for English translation). In the feel condition, participants were instructed to allow the image to trigger an emotional response and to experience this emotional response. For reappraisal, participants were instructed to think objectively while viewing the images in order to decrease emotional reactivity. They were to adopt the perspective of a professional performing a task requiring high concentration and try to perceive the stimuli objectively, rather than emotionally. In the suppress condition, participants were instructed to suppress any outward signs of emotion, so that an external observer would be unable to detect what the participant was experiencing subjectively. In the current study, we compare PTSD patients prior to onset of therapy to controls, and are interested in assessing neural and behavioral differences in their current ability to employ different emotion regulation strategies, rather than the ability to develop these techniques with training. As such, we provide instructions including a specific example, but not additional training in emotion regulation techniques. The instructions and approach that we employ are similar to those used in previous studies (Goldin et al. 2008, 2009; Gross 1998; Gross and Levenson 1997; McRae et al. 2008; Ochsner et al. 2002).
Experimental Task
Sixty combat-related images were presented using adjustable goggles and Presentation® software (Version 0.70, http://www.neurobs.com). We employed an event-related design, with the twenty trails per condition (feel, reappraise and suppress) randomly interspersed, rather than presented in a continuous block. In addition, condition-image combinations were counterbalanced across participants to ensure that images were not consistently paired with the same emotion regulation condition. A variable jitter interval of 0–1.5 s (varied in steps of 500 ms) was inserted before the instruction phase.
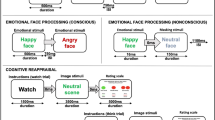
Prior to each image, there was a preparation phase, in which the instruction feel (Fühlen), reappraise (Distanzieren) or suppress (Unterdrücken) was presented for 1 s. Following this there was a presentation phase, in which the target image was then presented for 10 s. Participants were then asked to rate on a scale 1–4, “How much were you affected by this image?” (Wie sehr hat Sie dieses Bild bewegt?), with 1 indicating not affected and 4 indicting strongly affected. Participants responded using a 4-button response box in their right hand, and the scale was randomly presented in the order 1–4 or 4–1 to avoid movement preparation. The duration of the scale presentation was response-dependent. An 8-s fixation cross was then presented (see Fig. 1).
Experimental design for a single trial. The experiment consisted of 60 trials with 60 unique combat-related images. 20 trials of each condition (feel, reappraise and suppress) were randomly interspersed in an event related design. A single trial consisted of a variable jitter interval of 0–1.5 s (varied in steps of 500 ms), a 1 s instruction to either feel, reappraise or suppress, 10 s image presentation, “How much were you moved by this image?” (1–4) was presented until the participant made a response, and an 8-s fixation cross
Functional MRI Statistical Analysis
The fMRI data were analyzed using SPM8 software (Wellcome Department of Cognitive Neurology, London, UK). The first four volumes of all Echo Planar Imaging (EPI) series were excluded from the analysis to allow the magnetization to approach a dynamic equilibrium. Data processing started with slice time correction and realignment of the EPI datasets. A mean image for all EPI volumes was created, to which individual volumes were spatially realigned by means of rigid body transformations. The structural image was co-registered with the mean image of the EPI series. Then the structural image was normalized to the Montreal Neurological Institute (MNI) space using the ICBM152 template (Mazziotta et al. 2001), and the normalization parameters were applied to the EPI images to ensure an anatomically informed normalization. To correct for head movement, participants showing head motion above 3 mm of maximal translation (in any direction of x, y or z) and 3.0° of maximal rotation throughout the course of scanning would have been excluded. No participants showed head movements that exceeded these boundaries. A commonly applied filter of 8 mm full-width at half maximum (FWHM) was used. Low-frequency drifts in the time domain were removed by modeling the time series for each voxel by a set of discrete cosine functions, to which a cut-off of 128 s was applied.
The statistical analyses were performed using the general linear model (GLM). We modeled the instruction as an event to capture brain activity related to the preparatory phase, and modeled the target as a block (duration 10 s) to capture actual emotion regulation during image presentation. These vectors were convolved with a canonical hemodynamic response function (HRF) and its temporal derivatives to form regressors in a design matrix. The parameters of the ensuing GLM were estimated using the SPM8 standard specifications and used to form whole brain contrasts, comparing each condition with baseline (fixation cross) to test for main effects between conditions resulting in six separate linear contrast images (preparatory phase: feel-baseline, reappraise-baseline, suppress-baseline; image presentation: feel-baseline, reappraise-baseline, suppress-baseline). Separately for both groups, statistical information from all contrasts can be found in the supplemental material (see Supplemental Material S3). Following this, each emotion regulation condition was contrasted with the feel condition, resulting in four separate contrasts (preparatory phase: reappraise-feel, suppress-feel, image presentation: reappraise-feel, suppress-feel). The resulting contrast images were then entered into a series of two sample t tests at the second (between-subject) level, where we compared the two groups. A significant effect was reported when the results met a peak-level threshold of p < 0.001 and a cluster-level threshold of p < 0.05 in SPM, and when the size of the cluster was greater than the Monte Carlo simulation determined minimum cluster size above which the probability of type I error was < 0.05, using the 3dClustSim method, implemented in AFNI version 16.3.07 (α < 0.05, p < 0.001, FWHM 13.17 × 12.93 × 12.72, two-tailed) k = 56 voxels (Cox 1996; Cox et al. 2017). Monte Carlo simulation is a commonly used and widely accepted cluster correction method for SPM analyses (Cox et al. 2017; Ward 2000). The FWHM smoothness estimate was calculated directly from the data, as the average of the individual smoothness estimates. Due to the novel nature of this study, we also tested a more lenient threshold of (α < 0.10, p < 0.001, FWHM 13.17 × 12.93 × 12.72, two-tailed) k = 45 voxels, and report the single cluster that met this threshold, although due to the more lenient threshold this cluster should be interpreted with caution. For display purposes the resulting SPMs were thresholded at p < 0.001 (uncorrected). In addition, for information purposes we also report exploratory analysis at the lenient voxel level threshold of p < 0.005, and cluster corrected at k = 56 voxels (see Supplemental Material S4).
Rather than adopting a time-series analysis approach such as the one used by Goldin et al. (2008), we analyzed the image presentation phase as a single block. In the study by Goldin, images were presented for 15 s and the task was completed in three 9-min runs, while in the current study images were presented for 10 s, and the task was completed in 2 to 10-min runs. We chose a shorter image presentation duration, as we had a higher number of images to present, and we wanted to keep the total task duration down, as we are cognisant that PTSD patients can find MRI scanning unpleasant, due to the close confinement and loud noises. However, given the shorted image presentation, modelling the data using a time series approach is unlikely to produce interpretable results.
Results
Arousal Ratings
A MANOVA was conducted to compare arousal ratings for images for patients and controls. There was a significant group-by-condition interaction F(1,42) = 5.065, p = 0.004; Wilks’s Λ = 0.734, η2 = 0.266. Post-hoc independent and paired-sample t tests were then conducted. Patients found images significantly more arousing than controls for the feel [t(43) = 2.669, p < 0.01, one-tailed], reappraise [t(43) = 3.384, p < 0.01, one-tailed] and suppress [t(43) = 3.683, p < 0.001, one-tailed] conditions (see Table 2). Controls had significantly lower arousal ratings in the reappraisal condition than the feel condition [t(26) = 1.948, p < 0.05, one-tailed], while patients did not [t(17) = 0.456, p = 0.327, one-tailed]. Comparing the suppression and feel conditions, neither controls [t(26) = 1.519, p = 0.071, one-tailed) nor patients [t(17) = 1.373, p = 0.099, one-tailed] showed a significant difference. In addition, comparing the reappraisal and suppression conditions, neither controls [t(26) = 0.801, p = 0.215, one-tailed] nor patients [t(17) = 1.527, p = 0.073, one-tailed] showed a significant difference.
Functional Neuroimaging
Negative Emotion Induction Manipulation Check
To assess if the combat images elicited a neural response we contrasted the feel condition against baseline (fixation cross) for all participants. This contrast revealed higher activity during image presentation in the visual cortex, amygdala/hippocampus and medial orbitofrontal cortex (mOFC) (see Table 3 and Supplemental Material S4 for exploratory analysis). Independent samples t tests were also conducted in SPM and revealed no significant difference between patients and controls in either the task preparation or image presentation phases of the feel condition against baseline.
Preparatory Phase
Next, we turn to the key aim of the study, to investigate neural differences between PTSD patients and controls during the task preparation and image presentation phases of an emotion regulation task. During the preparatory phase, patients showed less activation in the vmPFC and left parietal lobe than controls during reappraisal (α < 0.05, p < 0.001, k = 77) (see Table 4; Fig. 2a, and Supplemental Material S4 for exploratory analysis). No difference was observed in the suppress and feel conditions during the preparatory phase.
Brain regions showing a significant group (PTSD patients and controls) difference in the reappraise minus feel condition in blood oxygen level dependent bold signal during a preparation and b image presentation. Warm colour indicates higher activation, and cold colour indicates lower activation in patients compared with controls during cognitive reappraisal. All clusters significant at α < 0.05, p < 0.001, k = 56, with the exception of the dACC which was significant at α < 0.10, p < 0.001, k = 45. dACC dorsal anterior cingulate cortex, rACC rostral anterior cingulate cortex, vmPFC ventromedial prefrontal cortex
Image Presentation
During the image presentation phase, comparing the reappraise and feel conditions, patients showed greater activation in the visual cortex (α < 0.05, p < 0.001, k = 73) and dACC (α < 0.10, p < 0.001, k = 45) (see Table 4; Fig. 2b, and Supplemental Material S4 for exploratory analysis) than controls during reappraisal. No difference was observed in the contrast between suppress and feel conditions during the image presentation phase.
Discussion
The goal of this study was to assess differences between combat-PTSD patients and combat-exposed healthy controls during an emotion regulation task, both at a behavioral and neural level. Two emotion regulation strategies, cognitive reappraisal and expressive suppression, were assessed during preparation and image presentation. In the reappraisal condition, controls showed higher prefrontal neural activity than patients during the preparatory phase, while patients showed higher prefrontal neural activity than controls during image presentation. No difference between patients and controls was observed in the suppression condition, either during the preparatory phase or during image presentation.
In the preparatory phase, controls showed higher activation than patients in the vmPFC and rACC, regions that have been previously implicated in affective and cognitive processing, including the regulation of fear expression, memory and emotional processing (Carter et al. 1999; Diekhof et al. 2011; Etkin et al. 2011; Milad et al. 2007a, b), as well as in the neurocircuitry of PTSD (Rauch et al. 2006). The rACC is known to regulate emotional responses and assess the salience of emotional stimuli, and has strong connections to limbic and paralimbic regions including the amygdala (Bush et al. 2000; Devinsky et al. 1995). The vmPFC is known to play a key role in self-referential processing and the extinction of conditioned fear (Milad et al. 2007a; Northoff et al. 2006). Previous work on cognitive reappraisal in healthy controls has demonstrated that early enhanced bold responses in the medial prefrontal cortex correlates with reduced late activation of the amygdala (Goldin et al. 2008), and a decrease in self-reported negative experiences. As such, higher activation of these regions during the preparatory phase should lead to more effective down regulation of negative emotion during the image presentation phase. This is reflected in the current results, as controls also reported significantly less arousal than patients.
During image presentation, patients showed higher activity than controls in the dorsal ACC (dACC) and in the visual cortex. The dACC shows strong connections with regions including the dorsolateral prefrontal cortex and supplementary motor areas, and has been implicated in conflict monitoring and response selection (Devinsky et al. 1995). The region observed in this study also corresponds to the anterior mid-cingulate cortex (aMCC) (Rotge et al. 2015), a region shown to be involved in negative affect, (social) pain, and cognitive control (Rotge et al. 2015; Shackman et al. 2011). In addition, the aMCC includes the rostral cingulate zone (RCZ), a region involved in conflict monitoring and facial movement (Picard and Strick 1996, 2001), with links to motor centers responsible for expressing affect and executing goal-directed behavior (Shackman et al. 2011). Greater activation in the visual cortex during image presentation in patients may suggest that controls disengage from the stimuli more than patients. Although not instructed, controls may have spontaneously engaged in attentional deployment, another form of early emotion regulation (Gross 2008). Previous eye-tracking studies have shown that PTSD is related to attentional bias for trauma-related stimuli, accompanied by greater autonomic arousal, compared with trauma-exposed controls (Felmingham et al. 2011; Kimble et al. 2010).
Therefore, patients and controls differ not only on a temporal dimension, but also on a regional, and therewith functional level, suggesting that during the reappraisal condition, patients and controls actually engaged in different strategies at different times. During the reappraisal condition, higher activation in controls in the rACC during the preparation phase may suggest that they implement preparatory affective regulation strategies. Higher activation in patients in the dACC during image presentation may suggest that patients implement more suppression-based strategies, such as control or suppression of motoric responses, later and less effectively, as indexed by higher subjective arousal ratings. In a similar neuroimaging study with healthy controls, higher early activation of prefrontal regions including the mPFC was observed during cognitive reappraisal, while higher late activation in regions including the vmPFC and dACC was observed during expressive suppression (Goldin et al. 2008). We did not observe a difference in neural activity between patients and controls in the suppression condition, either during the preparatory phase or during image presentation. This may indicate that PTSD patients and controls use similar brain regions at similar times when suppressing negative emotion.
We did not observe a difference in amygdala activity between patients and controls in any condition. This may be due to the highly relevant nature of the stimuli, as both groups had been exposed to combat. This is in line with previous neuroimaging studies that have also failed to demonstrate increased amygdala activity in PTSD patients during emotion regulation (New et al. 2009; Rabinak et al. 2014; Xiong et al. 2013), as well as image presentation (Phan et al. 2006) and traumatic reminders (Britton et al. 2005). In future studies, the addition of a non-trauma exposed control group may prove useful in teasing apart differences between trauma exposure per se and PTSD in amygdala reactivity.
One potential limitation of this study is that we did not include a measure of how effectively participants felt they had implemented each strategy. Previous work in healthy controls that has included a post-task measure to verify that the correct emotion regulation strategy was implemented has shown a compliance rate of 96% (Ochsner et al. 2002). Nevertheless, future work could usefully incorporate a measure of perceived success of strategy implementation to assess how PTSD patients perceive their abilities to down-regulate negative emotion regulation. In addition, the short preparation phase may have resulted in low power and future studies could provide longer preparation times to further disentangle preparation from target presentation.
Another potential limitation is that the patient group was significantly younger than the control group. Previous neuroimaging studies comparing age groups have found age-related differences, however these appear to be more pronounced at the structural level rather than the functional level (Rajah and D’Esposito 2005). In addition, although precise cutoffs vary, studies of age-related differences usually compare younger adults, for example between 18 and 35 years of age, to older adults, 60 years of age and above (Persson and Reuter-Lorenz 2008). However, in the current study, all individuals were in young to middle adulthood, with no individuals in old age. Nevertheless, age was included as a covariate in the neuroimaging analysis, although repeating the analyses without including age did not change the results.
One should also note that the generalizability of the current findings may be limited by both the specific nature and size of the participant groups. Given the relatively small sample size and the unequal subgroup sizes, we may lack sufficient power to detect smaller effects. Future work is needed to replicate the current findings and to explore how these results may extend to non-military and female samples.
In the current study, we analyzed the image presentation phase as a single block, as due to the duration of image presentation modelling the data using a time series approach was unlikely to produce interpretable results. Future studies may consider using a longer image presentation duration, similar to one used by Goldin et al. (2008), to also allow a time-series analysis approach.
The patients in the current study were assessed prior to treatment. Psychotherapies, such as CBT, train individuals to effectively employ emotion regulation techniques (Beck 2011), and one may expect that behavioral and neural correlates of emotion regulation will change following successful therapeutic intervention. Going forward, studies employing a longitudinal design would be useful to assess whether neural correlates of emotion regulation are useful in predicting responsiveness to therapy, and if these patterns change following training and the resolution of symptoms.
Conclusions
In the current study, we distinguish between task preparation and image presentation during two emotion regulation strategies, cognitive reappraisal and expressive suppression. Previous studies have not distinguished between these phases or strategies and show rather a general pattern of reduced neural activity in PTSD patients during emotion regulation. We found however, that patients showed lower neural activity during task preparation, and higher activity during image presentation in the cognitive reappraisal condition, a pattern of activity similar to that previously observed in expressive suppression in healthy controls. In the suppression condition, no difference between groups was observed. The current findings provide greater granularity of detail and reveal greater complexity regarding the neural dynamics of emotion regulation in PTSD than previously described.
References
American Psychiatric Association. (2000). DSM-IV. Diagnostic and Statistical Manual of Mental Disorders 4th ed. Washington, DC: American Psychiatric Association. https://doi.org/10.1016/B978-1-4377-2242-0.00016-X.
Beck, J. S. (2011). Cognitive behavior therapy: Basics and beyond. New York: Guilford Press.
Britton, J. C., Phan, K. L., Taylor, S. F., Fig, L. M., & Liberzon, I. (2005). Corticolimbic blood flow in posttraumatic stress disorder during script-driven imagery. Biological Psychiatry, 57(8), 832–840. https://doi.org/10.1016/j.biopsych.2004.12.025.
Bush, G., Luu, P., & Posner, M. I. (2000). Cognitive and emotional influences in anterior cingulate cortex. Trends in Cognitive Sciences, 4(6), 215–222. https://doi.org/10.1016/S1364-6613(00)01483-2.
Carter, C. S., Botvinick, M. M., & Cohen, J. D. (1999). The contribution of the anterior cingulate cortex to executive processes in cognition. Reviews in the Neurosciences, 10(1), 49–57. https://doi.org/10.1515/REVNEURO.1999.10.1.49.
Cisler, J. M., & Olatunji, B. O. (2012). Emotion regulation and anxiety disorders. Current Psychiatry Reports, 14(3), 182–187. https://doi.org/10.1007/s11920-012-0262-2.
Clohessy, S., & Ehlers, A. (1999). PTSD symptoms, response to intrusive memories and coping in ambulance service workers. The British Journal of Clinical Psychology: The British Psychological Society, 38(Pt 3), 251–265. https://doi.org/10.1348/014466599162836.
Cox, R. W. (1996). AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Computers and Biomedical Research, 29(3), 162–173. https://doi.org/10.1006/cbmr.1996.0014.
Cox, R. W., Chen, G., Glen, D. R., Reynolds, R. C., & Taylor, P. A. (2017). FMRI clustering in AFNI: False-positive rates redux. Brain Connectivity, 7(3), 152–171. https://doi.org/10.1089/brain.2016.0475.
Devinsky, O., Morrell, M. J., & Vogt, B. A. (1995). Contributions of anterior cingulate cortex to behaviour. Brain, 118(1), 279–306. https://doi.org/10.1093/brain/118.1.279.
Diekhof, E. K., Geier, K., Falkai, P., & Gruber, O. (2011). Fear is only as deep as the mind allows. A coordinate-based meta-analysis of neuroimaging studies on the regulation of negative affect. NeuroImage, 58(1), 275–285. https://doi.org/10.1016/j.neuroimage.2011.05.073.
Doulougeri, K., Panagopoulou, E., & Montgomery, A. (2016). How) do medical students regulate their emotions? BMC Medical Education, 16(1), 312. https://doi.org/10.1186/s12909-016-0832-9.
Etkin, A., Egner, T., & Kalisch, R. (2011). Emotional processing in anterior cingulate and medial prefrontal cortex. Trends in Cognitive Sciences. https://doi.org/10.1016/j.tics.2010.11.004.
Felmingham, K. L., Rennie, C., Manor, B., & Bryant, R. A. (2011). Eye tracking and physiological reactivity to threatening stimuli in posttraumatic stress disorder. Journal of Anxiety Disorders, 25(5), 668–673. https://doi.org/10.1016/j.janxdis.2011.02.010.
Foa, E. B., Cashman, L., Jaycox, L., & Perry, K. (1997). The validation of a self-report measure of posttraumatic stress disorder: The Posttraumatic Diagnostic Scale. Psychological Assessment. https://doi.org/10.1037/1040-3590.9.4.445.
Foa, E. B., Ehlers, A., Clark, D. M., Tolin, D. F., & Orsillo, S. M. (1999). The posttraumatic cognitions inventory (PTCI): Development and validation. Psychological Assessment. https://doi.org/10.1037/1040-3590.11.3.303.
Goldin, P. R., Manber-Ball, T., Werner, K., Heimberg, R., & Gross, J. J. (2009). Neural mechanisms of cognitive reappraisal of negative self-beliefs in social anxiety disorder. Biological Psychiatry, 66(12), 1091–1099. https://doi.org/10.1016/j.biopsych.2009.07.014.
Goldin, P. R., McRae, K., Ramel, W., & Gross, J. J. (2008). The neural bases of emotion regulation: Reappraisal and suppression of negative emotion. Biological Psychiatry, 63(6), 577–586. https://doi.org/10.1016/j.biopsych.2007.05.031.
Gross, J. J. (1998). Antecedent- and response-focused emotion regulation: Divergent consequences for experience, expression, and physiology. Journal of Personality and Social Psychology, 74(1), 224–237. https://doi.org/10.1037/0022-3514.74.1.224.
Gross, J. J. (2002). Emotion regulation: Affective, cognitive, and social consequences. Psychophysiology, 39(3), 281–291.
Gross, J. J. (2008). Emotion regulation. Handbook of emotions, 3, 497–513.
Gross, J. J., & Levenson, R. W. (1997). Hiding feelings: The acute effects of inhibiting negative and positive emotion. Journal of Abnormal Psychology, 106(1), 95–103. https://doi.org/10.1037/0021-843X.106.1.95.
Hoge, C. W., Castro, C. A., Messer, S. C., McGurk, D., Cotting, D. I., & Koffman, R. L. (2004). Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. New England Journal of Medicine, 351(1), 13–22. https://doi.org/10.1056/NEJMoa040603.
Kessler, R. C. (1995). Posttraumatic stress disorder in the national comorbidity survey. Archives of General Psychiatry, 52(12), 1048. https://doi.org/10.1001/archpsyc.1995.03950240066012.
Kimble, M. O., Fleming, K., Bandy, C., Kim, J., & Zambetti, A. (2010). Eye tracking and visual attention to threating stimuli in veterans of the Iraq war. Journal of Anxiety Disorders, 24(3), 293–299. https://doi.org/10.1016/j.janxdis.2009.12.006.
Klemanski, D. H., Mennin, D. S., Borelli, J. L., Morrissey, P. M., & Aikins, D. E. (2012). Emotion-related regulatory difficulties contribute to negative psychological outcomes in active-duty iraq war soldiers with and without posttraumatic stress disorder. Depression and Anxiety, 29(7), 621–628. https://doi.org/10.1002/da.21914.
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (2008). International affective picture system (IAPS): Instruction manual and affective ratings. Technical Report A. https://doi.org/10.1016/j.epsr.2006.03.016.
Mazziotta, J., Toga, A., Evans, A., Fox, P., Lancaster, J., Zilles, K., … Mazoyer, B. (2001). A probabilistic atlas and reference system for the human brain: International Consortium for Brain Mapping (ICBM). Philosophical Transactions of the Royal Society of London Series B: Biological Sciences, 356(1412), 1293–1322. https://doi.org/10.1098/rstb.2001.0915.
McFarlane, A. C. (2000). Posttraumatic stress disorder: A model of the longitudinal course and the role of risk factors. Journal of Clinical Psychiatry, 61(Suppl 5), 13–15.
McRae, K., Ochsner, K. N., Mauss, I., & Gabrieli, J. (2008). Gender differences in emotion regulation: An fMRI study of cognitive reappraisal. Group Processes & Intergroup Relations, 11(2), 143–162. https://doi.org/10.1177/1368430207088035.
Mental Health Advisory Team. (2006). Mental Health Advisory Team (MHAT) IV Operation Iraqi Freedom 05–07. Final Report.
Milad, M. R., Quirk, G. J., Pitman, R. K., Orr, S. P., Fischl, B., & Rauch, S. L. (2007a). A role for the human dorsal anterior cingulate cortex in fear expression. Biological Psychiatry, 62(10), 1191–1194. https://doi.org/10.1016/j.biopsych.2007.04.032.
Milad, M. R., Wright, C. I., Orr, S. P., Pitman, R. K., Quirk, G. J., & Rauch, S. L. (2007b). Recall of fear extinction in humans activates the ventromedial prefrontal cortex and hippocampus in concert. Biological Psychiatry, 62(5), 446–454. https://doi.org/10.1016/j.biopsych.2006.10.011.
Moore, S. A., Zoellner, L. A., & Mollenholt, N. (2008). Are expressive suppression and cognitive reappraisal associated with stress-related symptoms? Behaviour Research and Therapy, 46(9), 993–1000. https://doi.org/10.1016/j.brat.2008.05.001.
New, A. S., Fan, J., Murrough, J. W., Liu, X., Liebman, R. E., Guise, K. G., … Charney, D. S. (2009). A functional magnetic resonance imaging study of deliberate emotion regulation in resilience and posttraumatic stress disorder. Biological Psychiatry, 66(7), 656–664. https://doi.org/10.1016/j.biopsych.2009.05.020.
Northoff, G., Heinzel, A., de Greck, M., Bermpohl, F., Dobrowolny, H., & Panksepp, J. (2006). Self-referential processing in our brain-A meta-analysis of imaging studies on the self. NeuroImage, 31(1), 440–457. https://doi.org/10.1016/j.neuroimage.2005.12.002.
Ochsner, K. N., Bunge, S., Gross, J. J., & Gabrieli, J. D. E. (2002). Rethinking feelings: An FMRI study of the cognitive regulation of emotion. Journal of Cognitive Neuroscience, 14(8), 1215–1229. https://doi.org/10.1162/089892902760807212.
Ochsner, K. N., Ray, R. D., Cooper, J. C., Robertson, E. R., Chopra, S., Gabrieli, J. D. E., & Gross, J. J. (2004). For better or for worse: Neural systems supporting the cognitive down- and up-regulation of negative emotion. NeuroImage, 23(2), 483–499. https://doi.org/10.1016/j.neuroimage.2004.06.030.
Persson, J., & Reuter-Lorenz, P. A. (2008). Cognition and aging: Typical development. In C. A. Nelson & M. Luciana (Eds.), Handbook of developmental cognitive neuroscience (pp. 591–605). Cambridge, MA: MIT Press.
Phan, K. L., Britton, J. C., Taylor, S. F., Fig, L. M., & Liberzon, I. (2006). Corticolimbic blood flow during nontraumatic emotional processing in posttraumatic stress disorder. Archives of General Psychiatry, 63(2), 184–192. https://doi.org/10.1001/archpsyc.63.2.184.
Picard, N., & Strick, P. L. (1996). Motor areas of the medial wall: A review of their location and functional activation. Cerebral Cortex. https://doi.org/10.1093/cercor/6.3.342.
Picard, N., & Strick, P. L. (2001). Imaging the premotor areas. Current Opinion in Neurobiology. https://doi.org/10.1016/S0959-4388(01)00266-5.
Price, J. L., Monson, C. M., Callahan, K., & Rodriguez, B. F. (2006). The role of emotional functioning in military-related PTSD and its treatment. Journal of Anxiety Disorders, 20(5), 661–674. https://doi.org/10.1016/j.janxdis.2005.04.004.
Rabinak, C. A., MacNamara, A., Kennedy, A. E., Angstadt, M., Stein, M. B., Liberzon, I., & Phan, K. L. (2014). Focal and aberrant prefrontal engagement during emotion regulation in veterans with posttraumatic stress disorder. Depression and Anxiety, 31(10), 851–861. https://doi.org/10.1002/da.22243.
Rajah, M. N., & D’Esposito, M. (2005). Region-specific changes in prefrontal function with age: A review of PET and fMRI studies on working and episodic memory. Brain, 128(9), 1964–1983. https://doi.org/10.1093/brain/awh608.
Rauch, S. L., Shin, L. M., & Phelps, E. A. (2006). Neurocircuitry models of posttraumatic stress disorder and extinction: Human neuroimaging research-past, present, and future. Biological Psychiatry. https://doi.org/10.1016/j.biopsych.2006.06.004.
Rotge, J.-Y., Lemogne, C., Hinfray, S., Huguet, P., Grynszpan, O., Tartour, E., … Fossati, P. (2015). A meta-analysis of the anterior cingulate contribution to social pain. Social Cognitive and Affective Neuroscience, 10(1), 19–27. https://doi.org/10.1093/scan/nsu110.
Shackman, A. J., Salomons, T. V., Slagter, H. A., Fox, A. S., Winter, J. J., & Davidson, R. J. (2011). The integration of negative affect, pain and cognitive control in the cingulate cortex. Nature Reviews. Neuroscience, 12(3), 154–167. https://doi.org/10.1038/nrn2994.
Shapiro, J. (2013). The feeling physician: Educating the emotions in medical training. European Journal for Person Centered Healthcare, 1(2), 310. https://doi.org/10.5750/ejpch.v1i2.664.
Shepherd, L., & Wild, J. (2014). Emotion regulation, physiological arousal and PTSD symptoms in trauma-exposed individuals. Journal of Behavior Therapy and Experimental Psychiatry, 45(3), 360–367. https://doi.org/10.1016/j.jbtep.2014.03.002.
Shin, L. M., Rauch, S. L., & Pitman, R. K. (2006). Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Annals of the New York Academy of Sciences, 1071, 67–79. https://doi.org/10.1196/annals.1364.007.
Tull, M. T., Barrett, H. M., McMillan, E. S., & Roemer, L. (2007). A preliminary investigation of the relationship between emotion regulation difficulties and posttraumatic stress symptoms. Behavior Therapy, 38(3), 303–313. https://doi.org/10.1016/j.beth.2006.10.001.
Ward, B. D. (2000). AlphaSim documentation. In: Simultaneous inference for fMRI data. Milwaukee: Medical College of Wisconsin.
Willroth, E. C., & Hilimire, M. R. (2016). Differential effects of self- and situation-focused reappraisal. Emotion, 16(4), 468–474. https://doi.org/10.1037/emo0000139.
Xiong, K., Zhang, Y., Qiu, M., Zhang, J., Sang, L., Wang, L., … Li, M. (2013). Negative emotion regulation in patients with posttraumatic stress disorder. PLoS ONE, 8(12), e81957. https://doi.org/10.1371/journal.pone.0081957.
Acknowledgements
Open access funding provided by Max Planck Society. The authors would like to thank Elisabeth Labuhn for her helpful contribution.
Data Availability
The datasets generated and analyzed during the current study are not publicly available as this was not approved by the ethics committee and the participants did not agree to share their data publicly. However, the datasets are available from the corresponding author on reasonable request.
Funding
The study was funded by the Military Medical Academy of German Armed Forces, German Ministry of Defense. Oisin Butler received a PhD student stipend from the International Max Planck Research School on the Life Course (LIFE). Jürgen Gallinat has received research funding from the German Federal Ministry of Education and Research, German Science Foundation, AstraZeneca, and speaker fees from Lundbeck, Janssen-Cilag, Lilly and Otsuka. Simone Kühn has been funded by two grants from the German Science Foundation (DFG KU 3322/1-1, SFB 936/C7), the European Union (ERC-2016-StG-Self-Control-677804) and a Fellowship from the Jacobs Foundation (JRF 2016–2018).
Author information
Authors and Affiliations
Contributions
OB and GW contributed equally to this work. OB, GW, TG, PZ, UL, JG and SK contributed to the study design, acquisition and analysis of data and drafting of the manuscript. All co-authors have seen and approve with the contents of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Gerd Willmund and Peter Zimmermann are employed by the German Armed Forces. Their employment had no influence on the study design. All authors declare that they have no conflict of interest.
Ethical Approval
The protocol was approved by the ethics committee of Charité University Clinic, Berlin, Germany.
Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (national and institutional). Informed consent was obtained from all individual subjects participating in the study. All subjects gave written informed consent in accordance with the Declaration of Helsinki.
Research Involving with Human and Animal Participants
No animal studies were carried out by the authors for this article.
Additional information
Oisin Butler and Gerd Willmund have contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Butler, O., Willmund, G., Gleich, T. et al. Cognitive Reappraisal and Expressive Suppression of Negative Emotion in Combat-Related Posttraumatic Stress Disorder: A Functional MRI Study. Cogn Ther Res 43, 236–246 (2019). https://doi.org/10.1007/s10608-018-9905-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10608-018-9905-x