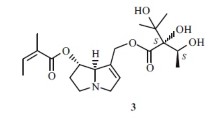
A new aporphine alkaloid, 6-formyl-1,2,9,10-tetramethoxy-6a,7-dehydroaporphine (1), was isolated from the aerial parts of Aconitum carmichaelii along with two known isoquinoline alkaloids, glaucine (2) and norglaucine hydrochloride (3). The structure of 1 was determined by extensive spectroscopic methods, especially 2D NMR analyses.
Similar content being viewed by others
References
Editorial Committee of Flora of China, Flora of China, Vol. 27, Science Press, Beijing, 1979, p. 113.
D. C. Hao, X. J. Gu, P. G. Xiao, L. J. Xu, and Y. Peng, J. Chin. Pharm. Sci., 22, 209 (2013).
S. J. Qu, C. H. Tan, Z. L. Liu, S. H. Jiang, L. Yu, and D. Y. Zhu, Phytochem. Lett., 4, 144 (2011).
Q. F. Tang, W. C. Ye, J. H. Liu, and C. H. Yang, Phytochem. Lett., 5, 397 (2012).
S. Vitalini, A. Braca, and G. Fico, Phytochem. Lett., 5, 476 (2012).
Y. J. Wang, J. Zhang, C. J. Zeng, Z. Yao, and Y. Zhang, Phytochem. Lett., 4, 166 (2011).
S. Yang, X. D. Yang, J. F. Zhao, H. B. Zhang, and L. Li, Helv. Chim. Acta, 90, 1160 (2007).
S. Yang, X. D. Yang, J. F. Zhao, J. Yang, H. B. Zhang, and L. Li, Chem. Nat. Compd., 44, 334 (2008).
X. D. Yang, S. Yang, J. Yang, J. F. Zhao, H. B. Zhang, and L. Li, Helv. Chim. Acta, 91, 569 (2008).
J. J. Chen, H. C. Hung, P. J. Sung, I. S. Chen, and W. L. Kuo, Phytochemistry, 72, 523 (2011).
A. Z. Turmukhambetov, G. K. Mukusheva, R. B. Seidakhmetova, E. E. Shults, M. M. Shakirov, I. Yu. Bagryanskaya, Yu. V. Gatilov, and S. M. Adekenov, Pharm. Chem. J., 43, 255 (2009).
M. P. Cava, I. Noquchi, and K. T. Buck, J. Org. Chem., 38, 2394 (1973).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 3, May–June, 2017, pp. 426–427.
Rights and permissions
About this article
Cite this article
Qin, Xd., Yang, S., Zhao, Y. et al. A New Aporphine Alkaloid from Aconitum carmichaelii . Chem Nat Compd 53, 501–503 (2017). https://doi.org/10.1007/s10600-017-2031-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-017-2031-2