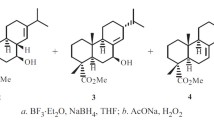
Ozonolysis of 3β,28-di-O-acetyl-29-norlupan-20-one-O-methylketoxime in the presence of ketones was used as an example to show that secondary ozonides at the C20 position of betulin are unstable and decompose to form the “anomalous product” methyl-3β,28-di-O-acetyl-29,30-bisnorlupan-20-oate, the structure of which was proved by an x-ray crystal structure analysis.


Similar content being viewed by others
References
K. Griesbaum, Trends Org. Chem., 6, 145 (1997).
K. Griesbaum, X. Liu, A. Kassiaris, and M. Scherer, Liebigs Ann., 1381 (1997).
G.-J. Ten Brink, I. W. C. E. Arends, and R. A. Sheldon, Chem. Rev., 104, 4105 (2004).
R. T. Aplin, R. P. K. Chan, and T. G. Halsall, J. Chem. Soc. C, 2322 (1969).
P. S. Bailey, Chem. Rev., 58, 925 (1958).
F. W. Semmler, Ber. Dtsch. Chem. Ges., 42, 246 (1909).
O. B. Flekhter, G. V. Giniyatullina, F. Z. Galin, N. Zh. Baschenko, N. S. Makara, F. S. Zarudii, E. I. Boreko, O. V. Savinova, N. I. Pavlova, Z. A. Starikova, and G. A. Tolstikov, Khim. Prir. Soedin., 582 (2005).
O. B. Kazakova, N. I. Medvedeva, and K. Yu. Suponitskii, Khim. Prir. Soedin., 365 (2011).
APEX2 and SAINT, Bruker AXS Inc., Madison, Wisconsin, USA, 2005.
G. M. Sheldrick, Acta Crystallogr. Sect. A: Found. Crystallogr., 64, 112 (2008).
Acknowledgment
The work was supported financially by the RFBR (Project No. 09-03-00831) and a grant of the RF President for State Support of Young Russian Scientists (MD-3852.2009.3.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2011, pp. 648–650.
Rights and permissions
About this article
Cite this article
Kazakova, O.B., Yamansarov, E.Y., Kukovinets, O.S. et al. Anomalous ozonolysis product of 3β,28-di-O-acetyl-29-norlupan-20-one-O-methyloxime. Chem Nat Compd 47, 738–740 (2011). https://doi.org/10.1007/s10600-011-0047-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-011-0047-6