Abstract
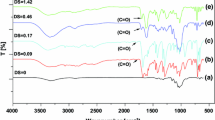
The ionic liquid 1-allyl-3-methylimidazolium chloride (AmimCl) as a reaction medium was studied for the synthesis of cellulose benzoates by homogeneous acylation of dissolved cellulose with benzoyl chlorides in the absence of any catalysts. Cellulose benzoates with a degree of substitution (DS) in the range from about 1 to 3.0 were accessible under mild conditions. The DS of cellulose derivatives increased with the increase of the molar ratio of benzoyl chloride/anhydroglucose unit (AGU) in cellulose, reaction time, and reaction temperature. Benzoylation of cellulose with some 4-substituted benzoyl chlorides including 4-toluoyl chloride, 4-chlorobenzoyl chloride and 4-nitrobenzoyl chloride was also readily carried out under mild conditions. Furthermore, regioselectively substituted mixed cellulose esters were synthesized in this work. All products were characterized by means of FT-IR, 1H-NMR, and 13C-NMR spectroscopy. In addition, at the end of benzoylation of cellulose, the ionic liquid AmimCl was easily recycled. When the recycled AmimCl was used as the reaction media, the cellulose benzoate with a similar DS was obtained under comparable reaction conditions.







Similar content being viewed by others
References
Acemoglu M, Kusters E, Baumann J, Hernandez I, Mak CP (1998) Synthesis of regioselectively substituted cellulose derivatives and applications in chiral chromatography. Chirality 10:294–306. doi:10.1002/(SICI)1520-636X(1998)10:4<294::AID-CHIR3>3.0.CO;2-8
Barthel S, Heinze T (2006) Acylation and carbanilation of cellulose in ionic liquids. Green Chem 8:301–306. doi:10.1039/b513157j
Braun D, Bahlig KH (1994) Herstellung und eigenschaften von cellulosebenzoat. Angew Makromol Chem 220:199–207. doi:10.1002/apmc.1994.052200117
Cao Y, Wu J, Meng T, Zhang J, He JS, Li HQ et al (2007) Acetone-soluble cellulose acetates prepared by one-step homogeneous acetylation of cornhusk cellulose in an ionic liquid 1-allyl-3-methylimidazolium chloride (AmimCl). Carbohydr Polym 69:665–672. doi:10.1016/j.carbpol.2007.02.001
Chen XM, Zou HF, Ni JY, Feng S (2003) Synthesis and characteristics of composite chiral stationary phases based on cellulose derivatives. J Sep Sci 26:29–36. doi:10.1002/jssc.200390010
El Seoud OA, Koschella A, Fidale LC, Dorn S, Heinze T (2007) Applications of ionic liquids in carbohydrate chemistry: a window of opportunities. Biomacromolecules 8:2629–2647. doi:10.1021/bm070062i
Felix G (2001) Regioselectively modified polysaccharide derivatives as chiral stationary phases in high-performance liquid chromatography. J Chromatogr A 906:171–184. doi:10.1016/S0021-9673(00)00943-2
Franco P, Senso A, Oliveros L, Minguillón C (2001) Covalently bonded polysaccharide derivatives as chiral stationary phases in high-performance liquid chromatography. J Chromatogr A 906:155–170. doi:10.1016/S0021-9673(00)00531-8
Francotte E, Wolf RM (1992) Chromatographic resolution on methylbenzoylcellulose beads: modulation of the chiral recognition by variation of the position of the methyl group on the aromatic ring. J Chromatogr A 595:63–75. doi:10.1016/0021-9673(92)85147-L
Goodlett VW, Dougherty JT, Patton HW (1971) Characterization of cellulose acetates by nuclear magnetic resonance. J Polym Sci Part Polym Chem 9:155–161. doi:10.1002/pol.1971.150090114
Holbrey JD, Seddon KR (1999) Ionic liquids. Clean Prod Process 1:223–236
Ichida A, Shibata T, Okamoto I, Yuki Y, Namikoshi H, Toga Y (1984) Resolution of enantiomers by HPLC on cellulose derivatives. Chromatographia 19:280–284. doi:10.1007/BF02687754
Köhler S, Heinze T (2007) Efficient synthesis of cellulose furoates in 1-N-butyl-3-methylimidazolium chloride. Cellulose 14:489–495. doi:10.1007/s10570-007-9138-8
Liu CF, Sun RC, Zhang AP, Qin MH, Ren JL, Wang XA (2007) Preparation and characterization of phthalated cellulose derivatives in room-temperature ionic liquid without catalysts. J Agric Food Chem 55:2399–2406. doi:10.1021/jf062876g
Okamoto Y, Yashima E (1998) Polysaccharide derivatives for chromatographic separation of enantiomers. Angew Chem Int Ed 37:1020–1043. doi:10.1002/(SICI)1521-3773(19980504)37:8<1020::AID-ANIE1020>3.0.CO;2-5
Okamoto Y, Kawashima M, Yamamoto K, Hatada K (1984) Useful chiral packing materials for high-performance liquid chromatographic resolution. Cellulose triacetate and tribenzoate coated on macroporous silical gel. Chem Lett 149:739–742. doi:10.1246/cl.1984.739
Okamoto Y, Aburatani R, Hatada K (1987) Chromatographic chiral resolution XIV Cellulose tribenzoate derivatives as chiral stationary phases for high-performance liquid chromatography. J Chromatogr A 389:95–102. doi:10.1016/S0021-9673(01)94414-0
Ren Q, Wu J, Zhang J, He JS, Guo ML (2003) Synthesis of 1-allyl, 3-methylimidazolium-based roomtemperature ionic liquid and preliminary study of its dissolving cellulose. Acta Polym Sin 3:448–451
Rimböck KH, Kastner F, Mannschreck A (1986) Microcrystalline tribenzoylcellulose: a high-performance liquid chromatographic sorbent for the separation of enantiomer. J Chromatogr A 351:346–350. doi:10.1016/S0021-9673(01)83506-8
Rogers RD, Seddon KR (2002) Ionic Liquids: Industrial Applications for Green Chemistry. American Chemical Society, Washington DC
Sheldon RA (2005) Green solvents for sustainable organic synthesis: state of the art. Green Chem 7:267–278. doi:10.1039/b418069k
Sun JX, Xu F, Geng ZC, Sun XF, Sun RC (2005) Comparative study of cellulose isolated by totally chlorine-free method from wood and cereal straw. J Appl Polym Sci 97:322–335. doi:10.1002/app.21728
Swatloski RP, Spear SK, Holbrey JD, Rogers RD (2002) Dissolution of cellose with ionic liquids. J Am Chem Soc 124:4974–4975. doi:10.1021/ja025790m
Wasserscheid P, Welton T (2003) Ionic Liquids in Synthesis. Wiley-VCH, Weinheim
Welton T (1999) Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem Rev 99:2071–2083. doi:10.1021/cr980032t
Wu J, Zhang J, Zhang H, He JS, Ren Q, Guo ML (2004) Homogeneous acetylation of cellulose in a new ionic liquid. Biomacromolecules 5:266–268. doi:10.1021/bm034398d
Yashima E (2001) Polysaccharide-based chiral stationary phases for high-performance liquid chromatographic enantioseparation. J Chromatogr A 906:105–125. doi:10.1016/S0021-9673(00)00501-X
Zhang H, Wu J, Zhang J, He JS (2005) 1-Ally-3-methylimidazolium chloride room temperature ionic liquid: a new and powerful nonderivatizing solvent for cellulose. Macromolecules 38:8272–8277. doi:10.1021/ma0505676
Acknowledgments
The authors thank the National Basic Research Program of China (No. 2004CB719701) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Wu, J., Cao, Y. et al. Synthesis of cellulose benzoates under homogeneous conditions in an ionic liquid. Cellulose 16, 299–308 (2009). https://doi.org/10.1007/s10570-008-9260-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-008-9260-2