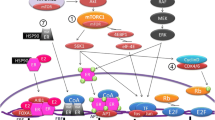
Abstract
Estrogen receptor (ER) is the major driver of most metastatic breast cancers (mBCs). Endocrine therapy (ET) is the most effective treatment for ER + mBC, but its effectiveness is limited by high rates of de novo and acquired resistance. A growing understanding of the biological characteristics and complexity of the ER pathway and the mechanisms of ET resistance has led to the development of a new generation of targeted therapies. One such mechanism is the cell cycle signaling pathways, which lead to the development of cyclin-dependent kinase 4/6 inhibitors (CDK4/6is) that have, in turn, transformed the management of such tumors. Another important mechanism is the alteration of the phosphatidylinositol 3′-kinase/AKT/mammalian target of rapamycin pathway. Drugs targeting each component of these pathways are currently used in clinical practice, and several more are in development. As a result, a myriad of new targeted therapies are consistently being added to the clinical oncologist armamentarium. Navigating the evolving and highly complex treatment landscape of HR + /HER2− mBC remains both an art and a challenge. In this review, we discuss the biological features of HR + /HER2− mBC and the different mechanisms of resistance to ET. We also discuss the management of mBC as the disease changes from endocrine-sensitive to endocrine-resistant.


Similar content being viewed by others
References
Osborne CK, Schiff R (2011) Mechanisms of endocrine resistance in breast cancer. Annu Rev Med 62:233–247. https://doi.org/10.1146/annurev-med-070909-182917
Nardone A, De Angelis C, Trivedi M V, Osborne CK, Schiff R (2015) The changing role of ER in endocrine resistance. Breast. 24Suppl 2(02):S60–S66. https://doi.org/10.1016/j.breast.2015.07.015
American Cancer Society. Breast Cancer Facts & Figures 2019–2020. Am Cancer Soc Inc. 2019
Buzdar AU (2004) Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the international letrozole breast cancer group. J Clin Oncol Off J Am Soc Clin Oncol 22(15):3199
Ballinger TJ, Meier JB, Jansen VM (2018) Current landscape of targeted therapies for hormone-receptor positive, HER2 negative metastatic breast cancer. Front Oncol 8:308
Finn RS, Aleshin A, Slamon DJ (2016) Targeting the cyclin-dependent kinases (CDK) 4/6 in estrogen receptor-positive breast cancers. Breast Cancer Res 18(1):17. https://doi.org/10.1186/s13058-015-0661-5
Giuliano M, Schettini F, Rognoni C et al (2019) Endocrine treatment versus chemotherapy in postmenopausal women with hormone receptor-positive, HER2−negative, metastatic breast cancer: a systematic review and network meta-analysis. Lancet Oncol 20(10):1360–1369. https://doi.org/10.1016/S1470-2045(19)30420-6
Ma CX, Reinert T, Chmielewska I, Ellis MJ (2015) Mechanisms of aromatase inhibitor resistance. Nat Rev Cancer 15(5):261–275. https://doi.org/10.1038/nrc3920
Razavi P, Chang MT, Xu G et al (2018) The genomic landscape of endocrine-resistant advanced breast cancers. Cancer Cell 34(3):427-438.e6. https://doi.org/10.1016/j.ccell.2018.08.008
Dustin D, Gu G, Fuqua SAW (2019) ESR1 mutations in breast cancer. Cancer 125(21):3714–3728. https://doi.org/10.1002/cncr.32345
Fu X, De Angelis C, Veeraraghavan J, Osborne CK, Schiff R (2019) Molecular Mechanisms of Endocrine Resistance BT - Estrogen Receptor and Breast Cancer: Celebrating the 60th Anniversary of the Discovery of ER. In: Zhang X, (ed) Cham: Springer International Publishing; 265–307. https://doi.org/10.1007/978-3-319-99350-8_11
Powles TJ, Hickish T, Kanis JA, Tidy A, Ashley S (1996) Effect of tamoxifen on bone mineral density measured by dual-energy x-ray absorptiometry in healthy premenopausal and postmenopausal women. J Clin Oncol 14(1):78–84. https://doi.org/10.1200/JCO.1996.14.1.78
Fisher B, Costantino JP, Wickerham DL et al (1998) Tamoxifen for prevention of breast cancer: report of the national surgical adjuvant breast and bowel project P-1 study. JNCI J Natl Cancer Inst 90(18):1371–1388. https://doi.org/10.1093/jnci/90.18.1371
(1998). Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet. 351(9114):1451-1467. https://doi.org/10.1016/S0140-6736(97)11423-44
Crump M, Sawka CA, DeBoer G et al (1997) An individual patient-based meta-analysis of tamoxifen versus ovarian ablation as first line endocrine therapy for premenopausal women with metastatic breast cancer. Breast Cancer Res Treat 44(3):201–210
Yan S, Li K, Jiao X, Zou H (2015) Tamoxifen with ovarian function suppression versus tamoxifen alone as an adjuvant treatment for premenopausal breast cancer: a meta-analysis of published randomized controlled trials. Onco Targets Ther 8:1433
Smith IE, Dowsett M (2003) Aromatase inhibitors in breast cancer. N Engl J Med 348(24):2431–2442. https://doi.org/10.1056/NEJMra023246
Mouridsen H, Gershanovich M, Sun Y et al (2001) Superior efficacy of letrozole versus tamoxifen as first-line therapy for postmenopausal women with advanced breast cancer: results of a phase III study of the International Letrozole Breast Cancer Group. J Clin Oncol 19(10):2596–2606
Nabholtz JM, Buzdar A, Pollak M et al (2000) Anastrozole is superior to tamoxifen as first-line therapy for advanced breast cancer in postmenopausal women: results of a North American multicenter randomized trial. J Clin Oncol 18(22):3758–3767
Paridaens RJ, Dirix LY, Beex LV et al (2008) Phase III study comparing exemestane with tamoxifen as first-line hormonal treatment of metastatic breast cancer in postmenopausal women: the European Organisation for Research and Treatment of Cancer Breast Cancer Cooperative Group. J Clin Oncol 26(30):4883–4890. https://doi.org/10.1200/JCO.2007.14.4659
Mauri D, Pavlidis N, Polyzos NP, Ioannidis JPA (2006) Survival with aromatase inhibitors and inactivators versus standard hormonal therapy in advanced breast cancer: meta-analysis. J Natl Cancer Inst 98(18):1285–1291. https://doi.org/10.1093/jnci/djj357
The A (2002) Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet 359(9324):2131–2139
Di Leo A, Jerusalem G, Petruzelka L et al (2014) Final overall survival: fulvestrant 500 mg vs 250 mg in the randomized CONFIRM trial. J Natl Cancer Inst. 106(1):djt337. https://doi.org/10.1093/jnci/djt337
Robertson JFR, Lindemann JPO, Llombart-Cussac A et al (2012) Fulvestrant 500 mg versus anastrozole 1 mg for the first-line treatment of advanced breast cancer: follow-up analysis from the randomized “FIRST” study. Breast Cancer Res Treat 136(2):503–511. https://doi.org/10.1007/s10549-012-2192-4
Ellis MJ, Llombart-Cussac A, Feltl D et al (2015) Fulvestrant 500 mg versus anastrozole 1 mg for the first-line treatment of advanced breast cancer: overall survival analysis from the phase II FIRST study. J Clin Oncol Off J Am Soc Clin Oncol 33(32):3781–3787. https://doi.org/10.1200/JCO.2015.61.5831
Robertson JFR, Bondarenko IM, Trishkina E et al (2016) Fulvestrant 500 mg versus anastrozole 1 mg for hormone receptor-positive advanced breast cancer (FALCON): an international, randomised, double-blind, phase 3 trial. Lancet 388(10063):2997–3005. https://doi.org/10.1016/S0140-6736(16)32389-3
Mehta RS, Barlow WE, Albain KS et al (2012) Combination anastrozole and fulvestrant in metastatic breast cancer. N Engl J Med 367(5):435–444. https://doi.org/10.1056/NEJMoa1201622
Mehta RS, Barlow WE, Albain KS et al (2019) Overall survival with fulvestrant plus anastrozole in metastatic breast cancer. N Engl J Med 380(13):1226–1234. https://doi.org/10.1056/NEJMoa1811714
Finn RS, Crown JP, Lang I et al (2015) The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2−negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol 16(1):25–35
Finn RS, Martin M, Rugo HS et al (2016) Palbociclib and letrozole in advanced breast cancer. N Engl J Med 375(20):1925–1936. https://doi.org/10.1056/NEJMoa1607303
Hortobagyi GN, Stemmer SM, Burris HA et al (2016) Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med 375(18):1738–1748. https://doi.org/10.1056/NEJMoa1609709
Slamon DJ, Neven P, Chia S et al (2019) Overall survival with ribociclib plus fulvestrant in advanced breast cancer. N Engl J Med 382(6):514–524. https://doi.org/10.1056/NEJMoa1911149
Im S-A, Lu Y-S, Bardia A et al (2019) Overall survival with ribociclib plus endocrine therapy in breast cancer. N Engl J Med 381(4):307–316. https://doi.org/10.1056/NEJMoa1903765
Goetz MP, Toi M, Campone M et al (2017) MONARCH 3: abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol 35(32):3638–3646. https://doi.org/10.1200/JCO.2017.75.6155
van Ommen-Nijhof A, Konings IR, van Zeijl CJJ et al (2018) Selecting the optimal position of CDK4/6 inhibitors in hormone receptor-positive advanced breast cancer - the SONIA study: study protocol for a randomized controlled trial. BMC Cancer 18(1):1146. https://doi.org/10.1186/s12885-018-4978-1
Martín M, Zielinski C, Ruíz-Borrego M, et al (2020) Abstract GS2–07: Results from PEARL study (GEICAM/2013–02_CECOG/BC.1.3.006): A phase 3 trial of Palbociclib (PAL) in combination with endocrine therapy (ET) versus Capecitabine (CAPE) in hormonal receptor (HR)-positive/human epidermal growth factor recept. Cancer Res. 80(4 Supplement):GS2–07 LP-GS2–07. https://doi.org/10.1158/1538-7445.SABCS19-GS2-07
Park YH, Kim T-Y, Kim GM et al (2019) Palbociclib plus exemestane with gonadotropin-releasing hormone agonist versus capecitabine in premenopausal women with hormone receptor-positive, HER2−negative metastatic breast cancer (KCSG-BR15-10): a multicentre, open-label, randomised, phase 2 trial. Lancet Oncol 20(12):1750–1759. https://doi.org/10.1016/s1470-2045(19)30565-0
Shah M, Nunes MR, Stearns V (2018) CDK4/6 inhibitors: game changers in the management of hormone receptor-positive advanced breast cancer? Oncology 32(5):216–222
Tripathy D, Im S-A, Colleoni M et al (2018) Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): a randomised phase 3 trial. Lancet Oncol 19(7):904–915. https://doi.org/10.1016/S1470-2045(18)30292-4
Rugo HS, Finn RS, Diéras V et al (2019) Palbociclib plus letrozole as first-line therapy in estrogen receptor-positive/human epidermal growth factor receptor 2-negative advanced breast cancer with extended follow-up. Breast Cancer Res Treat 174(3):719–729. https://doi.org/10.1007/s10549-018-05125-4
Johnston S, Martin M, Di Leo A et al (2019) MONARCH 3 final PFS: a randomized study of abemaciclib as initial therapy for advanced breast cancer. NPJ Breast Cancer 5:5. https://doi.org/10.1038/s41523-018-0097-z
Llombart-Cussac A, Pérez-García JM, Bellet M et al (2020) PARSIFAL: A randomized, multicenter, open-label, phase II trial to evaluate palbociclib in combination with fulvestrant or letrozole in endocrine-sensitive patients with estrogen receptor (ER)[+]/HER2[-] metastatic breast cancer. J Clin Oncol. 38(15_suppl):1007. https://doi.org/10.1200/JCO.2020.38.15_suppl.1007
Lei JT, Gou X, Seker S, Ellis MJ (2019) ESR1 alterations and metastasis in estrogen receptor positive breast cancer. J Cancer Metastas Treat. https://doi.org/10.20517/2394-4722.2019.12
Johnston SR, Kilburn LS, Ellis P et al (2013) Fulvestrant plus anastrozole or placebo versus exemestane alone after progression on non-steroidal aromatase inhibitors in postmenopausal patients with hormone-receptor-positive locally advanced or metastatic breast cancer (SoFEA): a composite, multicent. Lancet Oncol 14(10):989–998. https://doi.org/10.1016/S1470-2045(13)70322-X
Spoerke JM, Gendreau S, Walter K et al (2016) Heterogeneity and clinical significance of ESR1 mutations in ER-positive metastatic breast cancer patients receiving fulvestrant. Nat Commun 7:11579. https://doi.org/10.1038/ncomms11579
Fribbens C, O’Leary B, Kilburn L et al (2016) Plasma ESR1 mutations and the treatment of estrogen receptor-positive advanced breast cancer. J Clin Oncol Off J Am Soc Clin Oncol 34(25):2961–2968. https://doi.org/10.1200/JCO.2016.67.3061
Sammons S, Shastry M, Dent S, Anders C, Hamilton E (2020) Practical treatment strategies and future directions after progression while receiving CDK4/6 inhibition and endocrine therapy in advanced HR(+)/HER2(-) breast cancer. Clin Breast Cancer 20(1):1–11. https://doi.org/10.1016/j.clbc.2019.06.017
Laine M, Fanning SW, Greene M et al (2019) Lasofoxifene as a potential treatment for ER+ metastatic breast cancer. J Clin Oncol. 37(15):1056. https://doi.org/10.1200/JCO.2019.37.15_suppl.1056
Yuan Y, Frankel P, Synold T, et al (2018) Abstract OT1-05-02: A phase II clinical trial of the combination of pembrolizumab and selective androgen receptor modulator GTx-024 in patients with advanced androgen receptor positive triple negative breast cancer. Cancer Res 78(4 Supplement):OT1-05-02 LP-OT1-05-02. https://doi.org/10.1158/1538-7445.SABCS17-OT1-05-02
Palmieri C, Linden HM, Birrell S et al (2021) Efficacy of enobosarm, a selective androgen receptor (AR) targeting agent, correlates with the degree of AR positivity in advanced AR+/estrogen receptor (ER)+ breast cancer in an international phase 2 clinical study. J Clin Oncol. 39(15_suppl):1020. https://doi.org/10.1200/JCO.2021.39.15_suppl.1020
Turner NC, Slamon DJ, Ro J et al (2018) Overall survival with palbociclib and fulvestrant in advanced breast cancer. N Engl J Med 379(20):1926–1936. https://doi.org/10.1056/NEJMoa1810527
Sledge GW Jr, Toi M, Neven P et al (2020) The effect of abemaciclib plus fulvestrant on overall survival in hormone receptor-positive, ERBB2-negative breast cancer that progressed on endocrine therapy—MONARCH 2: a randomized clinical trial. JAMA Oncol 6(1):116–124. https://doi.org/10.1001/jamaoncol.2019.4782
Wander SA, Zangardi M, Niemierko A et al (2019) A multicenter analysis of abemaciclib after progression on palbociclib in patients (pts) with hormone receptor-positive (HR+)/HER2- metastatic breast cancer (MBC). J Clin Oncol. 37(15_suppl):1057. https://doi.org/10.1200/JCO.2019.37.15_suppl.1057
André F, Ciruelos E, Rubovszky G et al (2019) Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. N Engl J Med 380(20):1929–1940. https://doi.org/10.1056/NEJMoa1813904
André F, Ciruelos EM, Juric D et al (2020) Alpelisib plus fulvestrant for PIK3CA-mutated, hormone receptor-positive, human epidermal growth factor receptor-2–negative advanced breast cancer: final overall survival results from SOLAR-1. Ann Oncol. https://doi.org/10.1016/j.annonc.2020.11.011
Rugo HS, Lerebours F, Ciruelos E et al (2020) Alpelisib (ALP) + fulvestrant (FUL) in patients (pts) with PIK3CA-mutated (mut) hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2–) advanced breast cancer (ABC) previously treated with cyclin-dependent kinase 4/6 inh. J Clin Oncol. 38(15_suppl):1006. https://doi.org/10.1200/JCO.2020.38.15_suppl.1006
Baselga J, Campone M, Piccart M et al (2011) Everolimus in postmenopausal hormone-receptor–positive advanced breast cancer. N Engl J Med 366(6):520–529. https://doi.org/10.1056/NEJMoa1109653
Cook M, Al Rabadi L, Mitri ZI (2019) Everolimus and exemestane for the treatment of metastatic hormone receptor-positive breast cancer patients previously treated with CDK4/6 inhibitor based therapies. J Clin Oncol. 37(15_suppl):1058. https://doi.org/10.1200/JCO.2019.37.15_suppl.1058
Giridhar K V, Choong G, Leon-Ferre RA, et al (2019) Abstract P6–18–09: clinical management of metastatic breast cancer (MBC) after CDK 4/6 inhibitors: a retrospective single-institution study. Cancer Res 79
Hortobagyi GN, Chen D, Piccart M et al (2016) Correlative analysis of genetic alterations and everolimus benefit in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: results from BOLERO-2. J Clin Oncol Off J Am Soc Clin Oncol 34(5):419–426. https://doi.org/10.1200/JCO.2014.60.1971
Spring L, Bardia A (2017) SWISH-ing steroids: new standard of care to prevent everolimus-induced oral mucositis? Lancet Oncol 18(5):564–565. https://doi.org/10.1016/S1470-2045(17)30106-7
Ellis MJ, Gao F, Dehdashti F et al (2009) Lower-dose vs high-dose oral estradiol therapy of hormone receptor-positive, aromatase inhibitor-resistant advanced breast cancer: a phase 2 randomized study. JAMA 302(7):774–780. https://doi.org/10.1001/jama.2009.1204
Ingle JN, Ahmann DL, Green SJ et al (1981) Randomized clinical trial of diethylstilbestrol versus tamoxifen in postmenopausal women with advanced breast cancer. N Engl J Med 304(1):16–21. https://doi.org/10.1056/NEJM198101013040104
Iwase H, Yamamoto Y, Yamamoto-Ibusuki M et al (2013) Ethinylestradiol is beneficial for postmenopausal patients with heavily pre-treated metastatic breast cancer after prior aromatase inhibitor treatment: a prospective study. Br J Cancer 109(6):1537–1542. https://doi.org/10.1038/bjc.2013.520
Lønning PE, Taylor PD, Anker G et al (2001) High-dose estrogen treatment in postmenopausal breast cancer patients heavily exposed to endocrine therapy. Breast Cancer Res Treat 67(2):111–116. https://doi.org/10.1023/A:1010619225209
Ha G, Freeman SS, Choudhury AD, et al (2017) Scalable whole-exome sequencing of cell-free DNA reveals high concordance with metastatic tumors
Turner NC, O’Leary B, Cutts R et al (2018) Genetic landscape of resistance to CDK4/6 inhibition in circulating tumor DNA (ctDNA) analysis of the PALOMA3 trial of palbociclib and fulvestrant versus placebo and fulvestrant. J Clin Oncol 36(15_suppl):1001. https://doi.org/10.1200/JCO.2018.36.15_suppl.1001
Meng X, Song S, Jiang Z et al (2016) Receptor conversion in metastatic breast cancer: a prognosticator of survival. Oncotarget 7(44):71887–71903. https://doi.org/10.18632/oncotarget.12114
Gutierrez MC, Detre S, Johnston S et al (2005) Molecular changes in tamoxifen-resistant breast cancer: relationship between estrogen receptor, HER-2, and p38 mitogen-activated protein kinase. J Clin Oncol Off J Am Soc Clin Oncol 23(11):2469–2476. https://doi.org/10.1200/JCO.2005.01.172
Yang C, Li Z, Bhatt T et al (2017) Acquired CDK6 amplification promotes breast cancer resistance to CDK4/6 inhibitors and loss of ER signaling and dependence. Oncogene 36(16):2255–2264. https://doi.org/10.1038/onc.2016.379
Ma, Cynthia X; Sparano JA. Treatment approach to metastatic hormone receptor-positive, HER2-negative breast cancer: endocrine therapy and targeted agents. uptodate.com.
Winter C, Nilsson MP, Olsson E et al (2016) Targeted sequencing of BRCA1 and BRCA2 across a large unselected breast cancer cohort suggests that one-third of mutations are somatic. Ann Oncol Off J Eur Soc Med Oncol 27(8):1532–1538. https://doi.org/10.1093/annonc/mdw209
Robson M, Im SA, Senkus E et al (2017) Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Engl J Med 377(6):523–533. https://doi.org/10.1056/NEJMoa1706450
Litton JK, Rugo HS, Ettl J et al (2018) Talazoparib in patients with advanced breast cancer and a germline BRCA mutation. N Engl J Med 379(8):753–763. https://doi.org/10.1056/NEJMoa1802905
Dear RF, McGeechan K, Jenkins MC, Barratt A, Tattersall MHN, Wilcken N (2013) Combination versus sequential single agent chemotherapy for metastatic breast cancer. Cochrane Database Syst Rev 12:CD008792. https://doi.org/10.1002/14651858.CD008792.pub2
Kalinsky K, Diamond JR, Vahdat LT et al (2020) Sacituzumab govitecan in previously treated hormone receptor-positive/HER2-negative metastatic breast cancer: final results from a phase I/II, single-arm, basket trial. Ann Oncol 31(12):1709–1718
Rugo HS, Bardia A, Tolaney SM et al (2020) TROPiCS-02: A Phase III study investigating sacituzumab govitecan in the treatment of HR+/HER2-metastatic breast cancer. Futur Oncol 16(12):705–715
Modi S, Park H, Murthy RK et al (2020) Antitumor activity and safety of trastuzumab deruxtecan in patients with HER2-low-expressing advanced breast cancer: results from a phase Ib study. J Clin Oncol Off J Am Soc Clin Oncol 38(17):1887–1896. https://doi.org/10.1200/JCO.19.02318
Wolff AC, Hammond MEH, Allison KH et al (2018) Human epidermal growth factor receptor 2 testing in breast cancer: american society of clinical oncology/college of american pathologists clinical practice guideline focused update. Arch Pathol Lab Med 142(11):1364–1382. https://doi.org/10.5858/arpa.2018-0902-SA
Tarantino P, Hamilton E, Tolaney SM et al (2020) HER2-low breast cancer: pathological and clinical landscape. J Clin Oncol 38(17):1951–1962. https://doi.org/10.1200/JCO.19.02488
Nayar U, Cohen O, Kapstad C et al (2019) Acquired HER2 mutations in ER(+) metastatic breast cancer confer resistance to estrogen receptor-directed therapies. Nat Genet 51(2):207–216. https://doi.org/10.1038/s41588-018-0287-5
Yamashita H, Ishida N, Hatanaka Y et al (2020) HER2 gene amplification in ER-positive HER2 immunohistochemistry 0 or 1+ breast cancer with early recurrence. Anticancer Res 40(2):645–652. https://doi.org/10.21873/anticanres.13994
Mao P, Cohen O, Kowalski KJ et al (2020) Acquired FGFR and FGF alterations confer resistance to estrogen receptor (ER) targeted therapy in ER(+) metastatic breast cancer. Clin cancer Res an Off J Am Assoc Cancer Res 26(22):5974–5989. https://doi.org/10.1158/1078-0432.CCR-19-3958
Jones RH, Casbard A, Carucci M et al (2020) Fulvestrant plus capivasertib versus placebo after relapse or progression on an aromatase inhibitor in metastatic, oestrogen receptor-positive breast cancer (FAKTION): a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol 21(3):345–357
Veeraraghavan J, Tan Y, Cao X-X et al (2014) Recurrent ESR1-CCDC170 rearrangements in an aggressive subset of oestrogen receptor-positive breast cancers. Nat Commun 5:4577. https://doi.org/10.1038/ncomms5577
Rheinbay E, Parasuraman P, Grimsby J et al (2017) Recurrent and functional regulatory mutations in breast cancer. Nature 547(7661):55–60. https://doi.org/10.1038/nature22992
Berns K, Caumanns JJ, Hijmans EM, et al (2017) Abstract 3380: synthetic lethal interaction between <em>ARID1A</em> mutation and BET bromodomain inhibition in ovarian clear cell carcinoma. Cancer Res 77(13 Supplement):3380 LP–3380. https://doi.org/10.1158/1538-7445.AM2017-3380
Pearson A, Proszek P, Pascual J, et al (2020) Inactivating <em>NF1</em> mutations are enriched in advanced breast cancer and contribute to endocrine therapy resistance. Clin Cancer Res 26(3):608 LP–622. https://doi.org/10.1158/1078-0432.CCR-18-4044
Klesse LJ, Jordan JT, Radtke HB et al (2020) The use of MEK inhibitors in neurofibromatosis type 1-associated tumors and management of toxicities. Oncologist 25(7):e1109–e1116. https://doi.org/10.1634/theoncologist.2020-0069
Perez EA, Vogel CL, Irwin DH, Kirshner JJ, Patel R (2001) Multicenter phase II trial of weekly paclitaxel in women with metastatic breast cancer. J Clin Oncol 19(22):4216–4223. https://doi.org/10.1200/JCO.2001.19.22.4216
Jones SE, Erban J, Overmoyer B et al (2005) Randomized phase III study of docetaxel compared with paclitaxel in metastatic breast cancer. J Clin Oncol 23(24):5542–5551. https://doi.org/10.1200/JCO.2005.02.027
Gradishar WJ, Krasnojon D, Cheporov S et al (2009) Significantly longer progression-free survival with nab-paclitaxel compared with docetaxel as first-line therapy for metastatic breast cancer. J Clin Oncol 27(22):3611–3619. https://doi.org/10.1200/JCO.2008.18.5397
O’Brien MER, Wigler N, Inbar M et al (2004) Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl (CAELYX/Doxil) versus conventional doxorubicin for first-line treatment of metastatic breast cancer. Ann Oncol Off J Eur Soc Med Oncol 15(3):440–449. https://doi.org/10.1093/annonc/mdh097
Keller AM, Mennel RG, Georgoulias VA et al (2004) Randomized phase III trial of pegylated liposomal doxorubicin versus vinorelbine or mitomycin C plus vinblastine in women with taxane-refractory advanced breast cancer. J Clin Oncol 22(19):3893–3901. https://doi.org/10.1200/JCO.2004.08.157
Oshaughnessy JA, Blum J, Moiseyenko V et al (2001) Randomized, open-label, phase II trial of oral capecitabine (Xeloda) vs. a reference arm of intravenous CMF (cyclophosphamide, methotrexate and 5-fluorouracil) as first-line therapy for advanced/metastatic breast cancer. Ann Oncol Off J Eur Soc Med Oncol. 12(9):1247–1254. https://doi.org/10.1023/a:1012281104865
Fumoleau P, Largillier R, Clippe C et al (2004) Multicentre, phase II study evaluating capecitabine monotherapy in patients with anthracycline- and taxane-pretreated metastatic breast cancer. Eur J Cancer 40(4):536–542. https://doi.org/10.1016/j.ejca.2003.11.007
Cazzaniga ME, Pinotti G, Montagna E et al (2019) Metronomic chemotherapy for advanced breast cancer patients in the real world practice: Final results of the VICTOR-6 study. Breast 48:7–16
Cortes J, O’Shaughnessy J, Loesch D et al (2011) Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): a phase 3 open-label randomised study. Lancet 377(9769):914–923
Zelek L, Barthier S, Riofrio M et al (2001) Weekly vinorelbine is an effective palliative regimen after failure with anthracyclines and taxanes in metastatic breast carcinoma. Cancer 92(9):2267–2272. https://doi.org/10.1002/1097-0142(20011101)92:9%3c2267::AID-CNCR1572%3e3.0.CO;2-Q
Perez EA, Lerzo G, Pivot X et al (2007) Efficacy and safety of ixabepilone (BMS-247550) in a phase II study of patients with advanced breast cancer resistant to an anthracycline, a taxane, and capecitabine. J Clin Oncol Off J Am Soc Clin Oncol 25(23):3407–3414. https://doi.org/10.1200/JCO.2006.09.3849
Acknowledgements
The authors received no funding or financial support for the authorship or publication of this article. This article has been reviewed and proofread by the Editing Services, Research Medical Library at the University of Texas MD Anderson Cancer Center.
Author information
Authors and Affiliations
Contributions
JM wrote this review under the direct input and guidance of CKO, RS, and MR.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mouabbi, J.A., Osborne, C.K., Schiff, R. et al. Management of hormone receptor–positive, human epidermal growth factor 2–negative metastatic breast cancer. Breast Cancer Res Treat 190, 189–201 (2021). https://doi.org/10.1007/s10549-021-06383-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-021-06383-5