Abstract
Purpose
An antibody–drug conjugate targeting HER2, DS8201, has shown clinical activity against breast cancer with low-level HER2 expression. We aimed to evaluate the prognostic impact of intermediate HER2 expression in estrogen receptor (ER)+ early breast cancer (EBC) and metastatic breast cancer (MBC) cohorts.
Methods
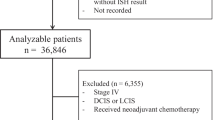
We analyzed prospectively collected data from EBC and MBC cohorts at Yonsei Cancer Center. Patients with HER2 immunohistochemistry (IHC) 0 ~ 1+ were assigned to the HER2-negative group, and patients with IHC 2+ and in situ hybridization (ISH)-negativity were assigned to the HER2-intermediate group. After the exclusion of HER2 IHC 3+ or ISH+ patients, a total of 2657 EBC and 535 MBC patients were analyzed.
Results
In total, 654 (24.6%) EBC and 166 (31.0%) MBC patients were classified in the HER2-intermediate group. The HER2-intermediate patients more frequently tended to have progesterone receptor (PR)-negativity and higher nuclear grade in the EBC cohort, and showed a higher proportion of patients aged ≥ 55 years compared with the HER2-negative group in the MBC cohort. The HER2-intermediate patients showed significantly poorer recurrence-free survival (RFS) compared to the HER2-negative patients in the EBC cohort (p = 0.044). Notably, intermediate HER2 expression predicted poorer RFS in EBC patients aged ≥ 55 years (hazard ratio 1.95; p = 0.042) in multivariate Cox analysis but did not affect RFS in those aged < 55 years. In line with the EBC cohort results, intermediate HER2 expression predicted poorer overall survival (OS) in MBC patients aged ≥ 55 (hazard ratio 1.45; p = 0.044) without affecting OS of those aged < 55 years.
Conclusion
Intermediate HER2 expression is an independent predictor of poor prognosis in both ER+ EBC and MBC patients aged ≥ 55 years. The clinical efficacy of new HER2-targeting antibody–drug conjugates needs to be validated in this high-risk subset of ER+ breast cancer patients.


Similar content being viewed by others
Data availability
The data from this study are available on request to the authors.
References
Perou CM, Sorlie T, Eisen MB et al (2000) Molecular portraits of human breast tumours. Nature 406(6797):747–752. https://doi.org/10.1038/35021093
Borg A, Tandon AK, Sigurdsson H et al (1990) HER-2/neu amplification predicts poor survival in node-positive breast cancer. Cancer Res 50(14):4332–4337
Romond EH, Perez EA, Bryant J et al (2005) Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 353(16):1673–1684. https://doi.org/10.1056/NEJMoa052122
Slamon DJ, Leyland-Jones B, Shak S et al (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344(11):783–792. https://doi.org/10.1056/NEJM200103153441101
Swain SM, Baselga J, Kim SB et al (2015) Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med 372(8):724–734. https://doi.org/10.1056/NEJMoa1413513
Geyer CE, Forster J, Lindquist D et al (2006) Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med 355(26):2733–2743. https://doi.org/10.1056/NEJMoa064320
Fehrenbacher L, Cecchini RS, Geyer CE et al (2018) NSABP B-47 (NRG oncology): phase III randomized trial comparing adjuvant chemotherapy with adriamycin (A) and cyclophosphamide (C) → weekly paclitaxel (WP), or docetaxel (T) and C with or without a year of trastuzumab (H) in women with node-positive or high-risk node-negative invasive breast cancer (IBC) expressing HER2 staining intensity of IHC 1+ or 2+ with negative FISH (HER2-Low IBC). Cancer Res. https://doi.org/10.1158/1538-7445.SABCS17-GS1-02
Arpino G, Wiechmann L, Osborne CK et al (2008) Crosstalk between the estrogen receptor and the HER tyrosine kinase receptor family: molecular mechanism and clinical implications for endocrine therapy resistance. Endocr Rev 29(2):217–233. https://doi.org/10.1210/er.2006-0045
Shou J, Massarweh S, Osborne CK et al (2004) Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J Natl Cancer Inst 96(12):926–935. https://doi.org/10.1093/jnci/djh166
Gilcrease MZ, Woodward WA, Nicolas MM et al (2009) Even low-level HER2 expression may be associated with worse outcome in node-positive breast cancer. Am J Surg Pathol 33(5):759–767. https://doi.org/10.1097/PAS.0b013e31819437f9
Ignatov T, Eggemann H, Burger E et al (2015) Moderate level of HER2 expression and its prognostic significance in breast cancer with intermediate grade. Breast Cancer Res Treat 151(2):357–364. https://doi.org/10.1007/s10549-015-3407-2
Eggemann H, Ignatov T, Burger E et al (2015) Moderate HER2 expression as a prognostic factor in hormone receptor positive breast cancer. Endocr Relat Cancer 22(5):725–733. https://doi.org/10.1530/ERC-15-0335
Paik S, Shak S, Tang G et al (2004) A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med 351(27):2817–2826. https://doi.org/10.1056/NEJMoa041588
Schalper KA, Kumar S, Hui P et al (2014) A retrospective population-based comparison of HER2 immunohistochemistry and fluorescence in situ hybridization in breast carcinomas: impact of 2007 American Society of Clinical Oncology/College of American Pathologists criteria. Arch Pathol Lab Med 138(2):213–219. https://doi.org/10.5858/arpa.2012-0617-OA
Ogitani Y, Aida T, Hagihara K et al (2016) DS-8201a, A novel HER2-targeting ADC with a novel DNA topoisomerase I inhibitor, demonstrates a promising antitumor efficacy with differentiation from T-DM1. Clin Cancer Res 22(20):5097–5108. https://doi.org/10.1158/1078-0432.CCR-15-2822
Iwata H, Tamura K, Doi T et al (2018) Trastuzumab deruxtecan (DS-8201a) in subjects with HER2-expressing solid tumors: long-term results of a large phase 1 study with multiple expansion cohorts. J Clin Oncol 36(15_suppl):2501. https://doi.org/10.1200/JCO.2018.36.15_suppl.2501
Hausman DF, Hamilton EP, Beeram M et al. (2017) Phase 1 study of ZW25, a bispecific anti-HER2 antibody, in patients with advanced HER2-expressing cancers. J Clin Oncol. https://doi.org/10.1200/JCO.2017.35.4_suppl.TPS215
Carlomagno C, Perrone F, Gallo C et al (1996) c-erb B2 overexpression decreases the benefit of adjuvant tamoxifen in early-stage breast cancer without axillary lymph node metastases. J Clin Oncol 14(10):2702–2708. https://doi.org/10.1200/JCO.1996.14.10.2702
De Laurentiis M, Arpino G, Massarelli E et al (2005) A meta-analysis on the interaction between HER-2 expression and response to endocrine treatment in advanced breast cancer. Clin Cancer Res 11(13):4741–4748. https://doi.org/10.1158/1078-0432.CCR-04-2569
Wolff AC, Hammond MEH, Allison KH et al (2018) Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. J Clin Oncol 36(20):2105–2122. https://doi.org/10.1200/JCO.2018.77.8738
Leong SP, Shen ZZ, Liu TJ et al (2010) Is breast cancer the same disease in Asian and Western countries? World J Surg 34(10):2308–2324. https://doi.org/10.1007/s00268-010-0683-1
Giuliano M, Trivedi MV, Schiff R (2013) Bidirectional crosstalk between the estrogen receptor and human epidermal growth factor receptor 2 signaling pathways in breast cancer: molecular basis and clinical implications. Breast Care (Basel) 8(4):256–262. https://doi.org/10.1159/000354253
Miller TW, Hennessy BT, Gonzalez-Angulo AM et al (2010) Hyperactivation of phosphatidylinositol-3 kinase promotes escape from hormone dependence in estrogen receptor-positive human breast cancer. J Clin Invest 120(7):2406–2413. https://doi.org/10.1172/JCI41680
Guo S, Sonenshein GE (2004) Forkhead box transcription factor FOXO3a regulates estrogen receptor alpha expression and is repressed by the Her-2/neu/phosphatidylinositol 3-kinase/Akt signaling pathway. Mol Cell Biol 24(19):8681–8690. https://doi.org/10.1128/MCB.24.19.8681-8690.2004
Creighton CJ, Hilger AM, Murthy S et al (2006) Activation of mitogen-activated protein kinase in estrogen receptor alpha-positive breast cancer cells in vitro induces an in vivo molecular phenotype of estrogen receptor alpha-negative human breast tumors. Cancer Res 66(7):3903–3911. https://doi.org/10.1158/0008-5472.CAN-05-4363
Lee AV, Cui X, Oesterreich S (2001) Cross-talk among estrogen receptor, epidermal growth factor, and insulin-like growth factor signaling in breast cancer. Clin Cancer Res 7(12 Suppl):4429s–4435s (Discussion 4411s-4412s)
Pietras RJ, Marquez-Garban DC (2007) Membrane-associated estrogen receptor signaling pathways in human cancers. Clin Cancer Res 13(16):4672–4676. https://doi.org/10.1158/1078-0432.CCR-07-1373
Baselga J, Campone M, Piccart M et al (2012) Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N Engl J Med 366(6):520–529. https://doi.org/10.1056/NEJMoa1109653
Andre F, Ciruelos E, Rubovszky G et al (2019) Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. N Engl J Med 380(20):1929–1940. https://doi.org/10.1056/NEJMoa1813904
Johnston S, Pippen J Jr, Pivot X et al (2009) Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for postmenopausal hormone receptor-positive metastatic breast cancer. J Clin Oncol 27(33):5538–5546. https://doi.org/10.1200/JCO.2009.23.3734
Kaufman B, Mackey JR, Clemens MR et al (2009) Trastuzumab plus anastrozole versus anastrozole alone for the treatment of postmenopausal women with human epidermal growth factor receptor 2-positive, hormone receptor-positive metastatic breast cancer: results from the randomized phase III TAnDEM study. J Clin Oncol 27(33):5529–5537. https://doi.org/10.1200/JCO.2008.20.6847
Martin M, Holmes FA, Ejlertsen B et al (2017) Neratinib after trastuzumab-based adjuvant therapy in HER2-positive breast cancer (ExteNET): 5-year analysis of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 18(12):1688–1700. https://doi.org/10.1016/S1470-2045(17)30717-9
Anders CK, Johnson R, Litton J et al (2009) Breast cancer before age 40 years. Semin Oncol 36(3):237–249. https://doi.org/10.1053/j.seminoncol.2009.03.001
Azim HA Jr, Michiels S, Bedard PL et al (2012) Elucidating prognosis and biology of breast cancer arising in young women using gene expression profiling. Clin Cancer Res 18(5):1341–1351. https://doi.org/10.1158/1078-0432.CCR-11-2599
Sparano JA, Gray RJ, Makower DF et al (2018) Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med 379(2):111–121. https://doi.org/10.1056/NEJMoa1804710
Diab SG, Elledge RM, Clark GM (2000) Tumor characteristics and clinical outcome of elderly women with breast cancer. J Natl Cancer Inst 92(7):550–556. https://doi.org/10.1093/jnci/92.7.550
Funding
This study was supported by a Grant from the National R&D Program for Cancer Control, Ministry of Health and Welfare, Republic of Korea (Grant No. HA17C0055).
Author information
Authors and Affiliations
Contributions
MHK and JS conceived and designed the study. MHK and JS performed analysis and interpretation of the data and wrote the manuscript. MHK, GMK, JHK, and JS contributed to metastatic breast cancer database administration, and JYK, HSP SP, YUC, BWP, and SIK contributed to early breast cancer database administration. All authors were involved in clinical care of the enrolled patients, provided study data, and commented on the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional review board of the Severance Hospital Yonsei University Health System (IRB 4–2016-0574) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants for clinical data collection and investigational use at the time of breast cancer diagnosis in the EBC cohort and at stage IV disease diagnosis in the MBC cohort.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, M.H., Kim, G.M., Kim, J.H. et al. Intermediate HER2 expression is associated with poor prognosis in estrogen receptor-positive breast cancer patients aged 55 years and older. Breast Cancer Res Treat 179, 687–697 (2020). https://doi.org/10.1007/s10549-019-05505-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05505-4