Abstract
Purpose
From the MINDACT trial, Cardoso et al. did not demonstrate a significant efficacy for adjuvant chemotherapy (CT) for women with early-stage breast cancer presenting high clinical and low genomic risks. Our objective was to assess the usefulness of the 70-gene signature in this population by using an alternative endpoint: the number of Quality-Adjusted Life-Years (QALYs), i.e., a synthetic measure of quantity and quality of life.
Methods
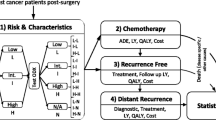
Based on the results of the MINDACT trial, we simulated a randomized clinical trial consisting of 1497 women with early-stage breast cancer presenting high clinical and low genomic risks. The individual preferences for the different health states and corresponding decrements were obtained from the literature.
Results
The gain in terms of 5-year disease-free survival was 2.8% (95% CI from − 0.1 to 5.7%, from 90.4% for women without CT to 93.3% for women with CT). In contrast, due to the associated side effects, CT significantly reduced the number of QALYs by 62 days (95% CI from 55 to 70 days, from 4.13 years for women without CT to 3.96 years for women with CT).
Conclusion
Our results support the conclusions published by Cardoso et al. by providing additional evidence that the 70-gene signature can be used to avoid overtreatment by CT for women with high clinical risk but low genomic risk.
Similar content being viewed by others
Abbreviations
- 95% CI:
-
95% confidence interval
- ABC:
-
Adjuvant breast cancer trial
- CMF:
-
Cyclophosphamide, methotrexate, and fluorouracil
- CT:
-
Chemotherapy
- DFS:
-
Disease-free survival
- DMFS:
-
Distant metastasis-free survival
- EBC:
-
Early-stage breast cancer
- E-CMF:
-
Epirubicin followed by cyclophosphamide, methotrexate, fluorouracil
- EORTC:
-
European organisation for research and treatment of cancer
- FEC60:
-
Fluorouracil, epirubicin, and cyclophosphamide
- FEC-D:
-
FEC60 followed by docetaxel
- HRQoL:
-
Health-related quality of life
- HR:
-
Hazard ratio
- MINDACT:
-
Microarray in node-negative disease may avoid chemotherapy
- NEAT:
-
National epirubicin adjuvant trial
- QALYs:
-
Quality-adjusted life-years
- TACT:
-
Taxotere as adjuvant chemotherapy trial
References
Ravdin PM, Siminoff LA, Davis GJ et al (2001) Computer program to assist in making decisions about adjuvant therapy for women with early breast cancer. J Clin Oncol 19:980–991. https://doi.org/10.1200/JCO.2001.19.4.980
Buyse M, Loi S, van’t Veer L et al (2006) Validation and clinical utility of a 70-gene prognostic signature for women with node-negative breast cancer. J Natl Cancer Inst 98:1183–1192. https://doi.org/10.1093/jnci/djj329
Bueno-de-Mesquita JM, Linn SC, Keijzer R et al (2009) Validation of 70-gene prognosis signature in node-negative breast cancer. Breast Cancer Res Treat 117:483–495. https://doi.org/10.1007/s10549-008-0191-2
Mook S, Schmidt MK, Viale G et al (2009) The 70-gene prognosis-signature predicts disease outcome in breast cancer patients with 1–3 positive lymph nodes in an independent validation study. Breast Cancer Res Treat 116:295–302. https://doi.org/10.1007/s10549-008-0130-2
van’t Veer LJ, Dai H, van de Vijver MJ et al (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 415:530–536. https://doi.org/10.1038/415530a
Harris L, Fritsche H, Mennel R et al (2007) American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 25:5287–5312. https://doi.org/10.1200/JCO.2007.14.2364
Cardoso F, van’t Veer LJ, Bogaerts J et al (2016) 70-Gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med 375:717–729. https://doi.org/10.1056/NEJMoa1602253
Lange S, Scheibler F, Fleer D, Windeler J (2017) Interpretation of the results of the MINDACT Study and consequent recommendations in the updated ASCO clinical practice guideline. JCO 36:429–430. https://doi.org/10.1200/JCO.2017.75.9506
Harris LN, Ismaila N, McShane LM et al (2016) Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol 34:1134–1150. https://doi.org/10.1200/JCO.2015.65.2289
Krop I, Ismaila N, Andre F et al (2017) Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline Focused Update. J Clin Oncol 35:2838–2847. https://doi.org/10.1200/JCO.2017.74.0472
Thewes B, Prins J, Friedlander M (2016) 70-gene signature in early-stage breast cancer. N Engl J Med 375:2199–2200. https://doi.org/10.1056/NEJMc1612048
Basch E (2013) Toward patient-centered drug development in oncology. N Engl J Med 369:397–400. https://doi.org/10.1056/NEJMp1114649
Foucher Y, Lorent M, Tessier P et al (2018) A mini-review of quality of life as an outcome in prostate cancer trials: patient-centered approaches are needed to propose appropriate treatments on behalf of patients. Health Qual Life Outcomes 16:40. https://doi.org/10.1186/s12955-018-0870-6
Sloan JA, Sargent DJ, Novotny PJ et al (2014) Calibration of quality-adjusted life years for oncology clinical trials. J Pain Symptom Manag 47:1091–1099.e3. https://doi.org/10.1016/j.jpainsymman.2013.07.016
Dantan E, Foucher Y, Lorent M et al (2016) Optimal threshold estimator of a prognostic marker by maximizing a time-dependent expected utility function for a patient-centered stratified medicine. Stat Methods Med Res 096228021667116. https://doi.org/10.1177/0962280216671161
Health related quality of life by age, gender and history of cardiovascular disease: results from the Health Survey for England—HEDS_DP_09_12.pdf. http://eprints.whiterose.ac.uk/10880/1/HEDS_DP_09-12.pdf. Accessed 20 Nov 2017
Campbell HE, Epstein D, Bloomfield D et al (2011) The cost-effectiveness of adjuvant chemotherapy for early breast cancer: A comparison of no chemotherapy and first, second, and third generation regimens for patients with differing prognoses. Eur J Cancer 47:2517–2530. https://doi.org/10.1016/j.ejca.2011.06.019
Dolan P, Gudex C, Kind P, Williams A (1995) A social tariff for EuroQol: results from a UK general population survey. Centre for Health Economics, University of York
Weinstein MC, Torrance G, McGuire A (2009) QALYs: the basics. Value Health 12(Suppl 1):S5–S9. https://doi.org/10.1111/j.1524-4733.2009.00515.x
Poisot T (2011) The digitize package: extracting numerical data from scatterplots. R J 3:25–26
R Development Core Team (2010) R: a language and environment for statistical computing. Vienna, Austria
Flores M, Glusman G, Brogaard K et al (2013) P4 medicine: how systems medicine will transform the healthcare sector and society. Per Med 10:565–576. https://doi.org/10.2217/PME.13.57
Duffy MJ, Harbeck N, Nap M et al (2017) Clinical use of biomarkers in breast cancer: Updated guidelines from the European Group on Tumor Markers (EGTM). Eur J Cancer 75:284–298. https://doi.org/10.1016/j.ejca.2017.01.017
Sparano JA, Gray RJ, Makower DF et al (2018) Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med 379:111–121. https://doi.org/10.1056/NEJMoa1804710
Collinson FJ, Gregory WM, McCabe C et al (2012) The STAR trial protocol: a randomised multi-stage phase II/III study of Sunitinib comparing temporary cessation with allowing continuation, at the time of maximal radiological response, in the first-line treatment of locally advanced/metastatic Renal Cancer. BMC Cancer. https://doi.org/10.1186/1471-2407-12-598
Royce TJ, Feldman AS, Mossanen M et al (2018) Comparative effectiveness of bladder-preserving tri-modality therapy versus radical cystectomy for muscle-invasive bladder cancer. Clin Genitourin Cancer. https://doi.org/10.1016/j.clgc.2018.09.023
Ferguson ND, Scales DC, Pinto R et al (2013) Integrating Mortality and Morbidity Outcomes. Am J Respir Crit Care Med 187:256–261. https://doi.org/10.1164/rccm.201206-1057OC
Glasziou PP, Simes RJ, Gelber RD (1990) Quality adjusted survival analysis. Stat Med 9:1259–1276. https://doi.org/10.1002/sim.4780091106
Cole BF, Gelber RD, Goldhirsch A (1993) Cox regression models for quality adjusted survival analysis. Stat Med 12:975–987
Aaronson NK, Ahmedzai S, Bergman B et al (1993) The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85:365–376. https://doi.org/10.1093/jnci/85.5.365
Towse A (2010) Net clinical benefit: the art and science of jointly estimating benefits and risks of medical treatment. Value Health 13(Suppl 1):S30–S32. https://doi.org/10.1111/j.1524-4733.2010.00753.x
Kind P, Lafata JE, Matuszewski K, Raisch D (2009) The use of QALYs in clinical and patient decision-making: issues and prospects. Value Health 12(Suppl 1):S27–S30. https://doi.org/10.1111/j.1524-4733.2009.00519.x
Ellis P, Barrett-Lee P, Johnson L et al (2009) Sequential docetaxel as adjuvant chemotherapy for early breast cancer (TACT): an open-label, phase III, randomised controlled trial. Lancet 373:1681–1692. https://doi.org/10.1016/S0140-6736(09)60740-6
Poole CJ, Earl HM, Hiller L et al (2006) Epirubicin and Cyclophosphamide, methotrexate, and fluorouracil as adjuvant therapy for early breast cancer. N Engl J Med 355:1851–1862. https://doi.org/10.1056/NEJMoa052084
Adjuvant Breast Cancer Trials Collaborative Group (2007) Polychemotherapy for early breast cancer: results from the international adjuvant breast cancer chemotherapy randomized trial. J Natl Cancer Inst 99:506–515. https://doi.org/10.1093/jnci/djk108
Acknowledgements
This work was supported by the Cancer National Institute (INCa, MAP-MARKER, No. 2013-137).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
This article does not contain any studies with animals performed by any of the authors.
Informed consent
For this type of study formal consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Caruana, E., Foucher, Y., Tessier, P. et al. Patient-centered simulations to assess the usefulness of the 70-gene signature for adjuvant chemotherapy administration in early-stage breast cancer. Breast Cancer Res Treat 174, 537–542 (2019). https://doi.org/10.1007/s10549-018-05107-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-05107-6