Abstract
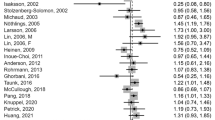
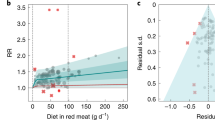
Epidemiological studies regarding the association between red and processed meat intake and the risk of breast cancer have yielded inconsistent results. Therefore, we conducted an updated and comprehensive meta-analysis which included 14 prospective studies to evaluate the association of red and processed meat intake with breast cancer risk. Relevant prospective cohort studies were identified by searching PubMed through October 31, 2014, and by reviewing the reference lists of retrieved articles. Study-specific relative risk (RR) estimates were pooled using a random-effects model. Fourteen prospective studies on red meat (involving 31,552 cases) and 12 prospective studies on processed meat were included in the meta-analysis. The summary RRs (95 % CI) of breast cancer for the highest versus the lowest categories were 1.10 (1.02, 1.19) for red meat, and 1.08 (1.01, 1.15) for processed meat. The estimated summary RRs (95 % CI) were 1.11 (1.05, 1.16) for an increase of 120 g/day of red meat, and 1.09 (1.03, 1.16) for an increase of 50 g/day of processed meat. Our findings indicate that increased intake of red and processed meat is associated with an increased risk of breast cancer. Further research with well-designed cohort or interventional studies is needed to confirm the association.




Similar content being viewed by others
References
Parkin DM, Pisani P, Ferlay J (1999) Global cancer statistics. CA 49:33–64
DeSantis C, Ma JM, Bryan L, Jemal A (2014) Breast cancer statistics, 2013. CA 64:52–62
de Lorgeril M, Salen P (2014) Helping women to good health: breast cancer, omega-3/omega-6 lipids, and related lifestyle factors. BMC Med 12:54
Farvid MS, Cho E, Chen WY, Eliassen AH, Willett WC (2014) Dietary protein sources in early adulthood and breast cancer incidence: prospective cohort study. BMJ 348:g3437
Farvid MS, Cho E, Chen WY, Eliassen AH, Willett WC (2014) Adolescent meat intake and breast cancer risk. Int J Cancer 136:1909–1920
Pouchieu C, Deschasaux M, Hercberg S et al (2014) Prospective association between red and processed meat intakes and breast cancer risk: modulation by an antioxidant supplementation in the SU.VI.MAX randomized controlled trial. Int J Epidemiol 43:1583–1592
Genkinger JM, Makambi KH, Palmer JR, Rosenberg L, Adams-Campbell LL (2013) Consumption of dairy and meat in relation to breast cancer risk in the Black Women’s Health Study. Cancer Causes Control 24:675–684
Lee HJ, Wu K, Cox DG et al (2013) Polymorphisms in xenobiotic metabolizing genes, intakes of heterocyclic amines and red meat, and postmenopausal breast cancer. Nutr Cancer 65:1122–1131
Alexander DD, Morimoto LM, Mink PJ, Cushing CA (2010) A review and meta-analysis of red and processed meat consumption and breast cancer. Nutr Res Rev 23:349–365
Stroup DF, Berlin JA, Morton SC et al (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 283:2008–2012
Byrne C, Ursin G, Ziegler RG (1996) A comparison of food habit and food frequency data as predictors of breast cancer in the NHANES I/NHEFS cohort. J Nutr 126:2757–2764
Missmer SA, Smith-Warner SA, Spiegelman D et al (2002) Meat and dairy food consumption and breast cancer: a pooled analysis of cohort studies. Int J Epidemiol 31:78–85
van der Hel OL, Peeters PH, Hein DW et al (2004) GSTM1 null genotype, red meat consumption and breast cancer risk (The Netherlands). Cancer Causes Control 15:295–303
Shannon J, Ray R, Wu C et al (2005) Food and botanical groupings and risk of breast cancer: a case-control study in Shanghai, China. Cancer Epidemiol Biomark Prev 14:81–90
Cho EY, Chen WY, Hunter DJ et al (2006) Red meat intake and risk of breast cancer among premenopausal women. Arch Intern Med 166:2253–2259
Taylor EF, Burley VJ, Greenwood DC, Cade JE (2007) Meat consumption and risk of breast cancer in the UK Women’s Cohort Study. Br J Cancer 96:1139–1146
Cross AJ, Leitzmann MF, Gail MH et al (2007) A prospective study of red and processed meat intake in relation to cancer risk. PLoS Med 4:1973–1984
Egeberg R, Olsen A, Autrup H et al (2008) Meat consumption, N-acetyl transferase 1 and 2 polymorphism and risk of breast cancer, in Danish postmenopausal women. Eur J Cancer Prev 17:39–47
Pala V, Krogh V, Berrino F et al (2009) Meat, eggs, dairy products, and risk of breast cancer in the European prospective investigation into cancer and nutrition (EPIC) cohort. Am J Clin Nutr 90:602–612
Ferrucci LM, Cross AJ, Graubard BI et al (2009) Intake of meat, meat mutagens, and iron and the risk of breast cancer in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Br J Cancer 101:178–184
Larsson SC, Bergkvist L, Wolk A (2009) Long-term meat intake and risk of breast cancer by oestrogen and progesterone receptor status in a cohort of Swedish women. Eur J Cancer 45:3042–3046
Stang A (2010) Critical evaluation of the Newcastle–Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Greenland S, Longnecker MP (1992) Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol 135:1301–1309
Orsini N, Bellocco R, Greenland S (2006) Generalized least squares for trend estimation of summarized dose–response data. Stata J 6:40–57
Norat T, Lukanova A, Ferrari P, Riboli E (2002) Meat consumption and colorectal cancer risk: dose-response meta-analysis of epidemiological studies. Int J Cancer 98:241–256
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Begg CB, Mazumdar M (1994) Operating characteristics of a rank correlation test for publication bias. Biometrics 50:1088–1101
Fonseca-Nunes A, Jakszyn P, Agudo A (2014) Iron and cancer risk-a systematic review and meta-analysis of the epidemiological evidence. Cancer Epidemiol Biomark 23:12–31
Kazerouni N, Sinha R, Hsu CH, Greenberg A, Rothman N (2001) Analysis of 200 food items for benzo[a] pyrene and estimation of its intake in an epidemiologic study. Food Chem Toxicol 39:423–436
Knize MG, Salmon CP, Pais P, Felton JS (1999) Food heating and the formation of heterocyclic aromatic amine and polycyclic aromatic hydrocarbon mutagens/carcinogens. Adv Exp Med Biol 459:179–193
De Stefani E, Ronco A, Mendilaharsu M, Guidobono M, Deneo-Pellegrini H (1997) Meat intake, heterocyclic amines, and risk of breast cancer: a case-control study in Uruguay. Cancer Epidemiol Biomark 6:573–581
Rundle A, Tang DL, Hibshoosh H et al (2000) The relationship between genetic damage from polycyclic aromatic hydrocarbons in breast tissue and breast cancer. Carcinogenesis 21:1281–1289
Sinha R, Gustafson DR, Kulldorff M et al (2000) 2-amino-1-methyl-6-phenylimidazo [4,5 b] pyridine, a arcinogen in high-temperature -cooked meat, and breast cancer risk. J Natl Cancer Inst 92:1352–1354
Gammon MD, Santella RM, Neugut AI et al (2002) Environmental toxins and breast cancer on long Island. I. Polycyclic aromatic hydrocarbon DNA adducts. Cancer Epidemiol Biomark 11:677–685
Bonner MR, Han D, Nie J et al (2005) Breast cancer risk and exposure in early life to polycyclic aromatic hydrocarbons using total suspended particulates as a proxy measure. Cancer Epidemiol Biomark 14:53–60
Zheng W, Lee SA (2009) Well-done meat intake, heterocyclic amine exposure, and cancer risk. Nutr Cancer 61:437–446
Andersson AM, Skakkebaek NE (1999) Exposure to exogenous estrogens in food: possible impact on human development and health. Eur J Endocrinol 140:477–485
Stephany RW (2001) Hormones in meat: different approaches in the EU and in the USA. Apmis 109:S357–S363
Samraj AN, Pearce OM, Laubli H et al (2015) A red meat-derived glycan promotes inflammation and cancer progression. Proc Natl Acad Sci USA 112:542–547
Brody JG, Rudel RA (2003) Environmental pollutants and breast cancer. Environ Health Perspect 111:1007–1019
Brody JG, Moysich KB, Humblet O, Attfield KR, Beehler GP, Rudel RA (2007) Environmental pollutants and breast cancer: epidemiologic studies. Cancer 109:2667–2711
Girschik J, Heyworth J, Fritschi L (2013) Self-reported sleep duration, sleep quality, and breast cancer risk in a population-based case-control study. Am J Epidemiol 177:316–327
Acknowledgments
The study was supported by grants from the Ministry of Science and Technology of China (2011CB510104 and 2012CB945004), the National Natural Science Foundation of China (31090362), the National Natural Science Foundation of China (81071684), the Hundred Talents Program (2010OHTP11), Shanghai PuJiang Talent program (11PJ1411200), and the Ministry of Science and Technology of China (2012BAK01B00).
Conflict of interest
The authors of this study have no conflict of interest or any financial disclosures to make.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, J., Wei, W. & Zhan, L. Red and processed meat intake and risk of breast cancer: a meta-analysis of prospective studies. Breast Cancer Res Treat 151, 191–198 (2015). https://doi.org/10.1007/s10549-015-3380-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-015-3380-9