Abstract
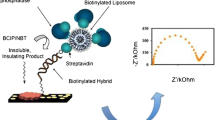
Low-cost detection of pathogens and biomolecules at the point-of-care promises to revolutionize medicine through more individualized monitoring and increased accessibility to diagnostics in remote and resource-limited areas. While many approaches to biosensing are still limited by expensive components or inadequate portability, we present here an ELISA-inspired lab-on-a-chip strategy for biological detection based on liposome tagging and ion-release impedance spectroscopy. Ion-encapsulating dipalmitoylphosphatidylcholine (DPPC) liposomes can be functionalized with antibodies and are stable in deionized water yet permeabilized for ion release upon heating, making them ideal reporters for electrical biosensing of surface-immobilized antigens. We demonstrate the quantification of these liposomes by real-time impedance measurements, as well as the qualitative detection of viruses as a proof-of-concept toward a portable platform for viral load determination which can be applied broadly to the detection of pathogens and other biomolecules.






Similar content being viewed by others
References
T. Chen, D. McIntosh, Y. He, J. Kim, D.A. Tirrell, P. Scherrer, D.B. Fenske, A.P. Sandhu, P.R. Cullis, Mol. Membr. Biol. 21, 385 (2004)
G. Chen, C.J. Alberts, W. Rodriguez, M. Toner, Anal. Chem. 82, 723 (2010)
X. Cheng, Y. Liu, D. Irimia, U. Demirci, L. Yang, L. Zamir, W.R. Rodríguez, M. Toner, R. Bashir, Lab. Chip 7, 746 (2007)
J.T. Connelly, S. Kondapalli, M. Skoupi, J.S.L. Parker, B.J. Kirby, A.J. Baeumner, Anal. Bioanal. Chem. 402, 315 (2012)
G.L. Damhorst, N.N. Watkins, R. Bashir, IEEE Trans. Bio-med. Eng. 60, 715 (2013)
A. deMello, D. van Swaay, Lab. Chip. 13, 752 (2012)
K. A. Edwards, O. R. Bolduc, and A. J. Baeumner, Curr. Opin. Chem. Biol. 16, 1 (2012)
D.H. Ekstrand, R.J. Awad, C.F. Källander, J.S. Gronowitz, Biotechnol. Appl. Biochem. 23(Pt 2), 95 (1996)
R. Gómez, R. Bashir, A. Bhunia, Sens. Actuators B: Chem. 86, 198 (2002)
R. Gómez-sjöberg, D.T. Morisette, R. Bashir, S. Member, J. Microelectromech. Syst. 14, 829 (2005)
D.D. Ho, Science 272, 1124 (1996)
M. Hwang, R. Prud’homme, J. Kohn, J. Thomas, Langmuir 17 (2001)
S.A. Kim, J.S. Peacock, J. Immunol. Methods 158, 57 (1993)
E. Kim, J. Stanton, B. Korber, Nanomedicine 3 (2008)
Y.-G. Kim, S. Moon, D.R. Kuritzkes, U. Demirci, Biosens. Bioelectron. 25, 253 (2009)
S. Kwakye, V.N. Goral, A.J. Baeumner, Biosens. Bioelectron. 21, 2217 (2006)
K.-B. Lee, E.-Y. Kim, C.A. Mirkin, S.M. Wolinsky, Nano Lett 4, 1869 (2004)
S.H. Lee, S.-W. Kim, J.Y. Kang, C.H. Ahn, Lab. Chip 8, 2121 (2008)
H.H. Lee, M.A. Dineva, Y.L. Chua, A.V. Ritchie, I. Ushiro-Lumb, C.A. Wisniewski, J. Infect. Dis. 201(Suppl), S65 (2010)
F. Lisdat, D. Schäfer, Anal. Bioanal. Chem. 391, 1555 (2008)
Q. Liu, B. Boyd, Analyst. 138, 391 (2013)
S.R. Nugen, P.J. Asiello, J.T. Connelly, A.J. Baeumner, Biosens. Bioelectron. 24, 2428 (2009)
Omega Engineering, Omega Engineering, Inc. 11 (2013)
Z.A. Parpia, R. Elghanian, A. Nabatiyan, D.R. Hardie, D.M. Kelso, JAIDS J. Acquir. Immune Defic. Syndr. 55, 413 (2010)
H. Shafiee, M. Jahangir, F. Inci, S. Wang, R. B. M. Willenbrecht, F. F. Giguel, A. M. N. Tsibris, D. R. Kuritzkes, and U. Demirci, Small 1 (2013). doi:10.1002/smll.201202195
F. Shen, B. Sun, J.E. Kreutz, E.K. Davydova, W. Du, P.L. Reddy, L.J. Joseph, R.F. Ismagilov, J. Am. Chem. Soc. 133, 17705 (2011)
S. Tang, I. Hewlett, J. Infect. Dis. 201(Suppl), S59 (2010)
S. Tang, J. Zhao, J. Storhoff, JAIDS J. Acquir. Immune Defic. Syndr. 46, 231 (2007)
W. Tang, W.H.A. Chow, Y. Li, H. Kong, Y.-W. Tang, B. Lemieux, J. Infect. Dis. 201(Suppl), S46 (2010)
S. Tanriverdi, L. Chen, S. Chen, J. Infect. Dis. 201(Suppl), S52 (2010)
S.-Y. Teh, R. Khnouf, H. Fan, A.P. Lee, Biomicrofluidics 5, 44113 (2011)
S. Wang, A. Ip, F. Xu, F. F. Giguel, S. Moon, A. Akay, D. R. Kuritzkes, and U. Demirci, sensors, and command, control, communications, and intelligence (C3I) technologies for homeland security and homeland defense IX 7666, 76661H (2010)
S. Wang, M. Esfahani, U.A. Gurkan, F. Inci, D.R. Kuritzkes, U. Demirci, Lab. Chip 12, 1508 (2012)
Woongjin Chemical Co Ltd, CSM Technical Manual (Seoul, Republic of Korea, 2010), p. 137
N.V. Zaytseva, V.N. Goral, R.A. Montagna, A.J. Baeumner, Lab. Chip 5, 805 (2005a)
N.V. Zaytseva, R.A. Montagna, A.J. Baeumner, Anal. Chem. 77, 7520 (2005b)
Acknowledgments
We would like to thank Mehmet Toner at Massachusetts General Hospital, William Rodriguez and Marta Fernandez Suarez at Daktari Diagnostics, Inc., and Joshua Wood and Brian Dorvel at the University of Illinois for helpful discussions. Partial support was provided by the Illinois Distinguished Fellowship (to GLD) at the University of Illinois at Urbana-Champaign. This work was also supported by the National Institutes of Health (1R01 HL109192 to H.J.K. and Chemistry-Biology Interface Training Program 5T32GM070421 to C.E.S.). The devices were fabricated and tested at the Micro and Nanotechnology Laboratory at the University of Illinois at Urbana-Champaign (www.mntl.illinois.edu).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Damhorst, G.L., Smith, C.E., Salm, E.M. et al. A liposome-based ion release impedance sensor for biological detection. Biomed Microdevices 15, 895–905 (2013). https://doi.org/10.1007/s10544-013-9778-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-013-9778-4