Abstract
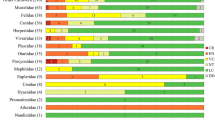
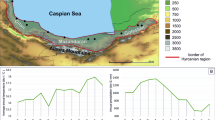
The vast majority of empirical studies on regional mammal defaunation across the Atlantic Forest biome of South America are concentrated in Southeastern and Northeastern regions. Thus, the lack of empirical information on the medium- to large-bodied mammal population declines across the subtropical region of this tropical biome and its major causes are paramount to conservation efforts. We investigate the influence of land use on mammal defaunation across the subtropical Atlantic Forest by sampling 91 points using 65 camera-traps installed for five consecutive years—totaling an effort of 30,189 camera-trap-days. We observed that the average defaunation across the Devonian Kniferidges Environmental Protection Area meta-region (spanning over subtropical Atlantic Forest plateaus) was 42% (D = 0.42; ± 0.17) when compared to presumed assemblages of historical times going back to the Pre-Columbian era (ca. 500 years ago). The main landscape predictors of regional defaunation were silviculture and agriculture, once the highest defaunation indexes were concentered in sites intensely human-modified by these features. Protected areas had significantly lower defaunation indexes in comparison with the unprotected areas. Although our results of defaunation were 29.2% lesser than the average for the entire biome, the negative consequences of regional defaunation on ecosystem services are already occurring, once two in each five local species presumably are locally extinct or present low abundances (i.e. functionally extinct). Given that habitat conversion is the primary cause of global biodiversity decline, our results reinforce that biodiversity conservation is still strongly dependent on natural protected areas networks.




Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Almeida CG, Moro RS (2007) Análise da cobertura florestal no Parque Nacional dos Campos Gerais, Paraná, como subsídio ao seu plano de manejo. Terra Plur 1:115–122
Altrichter M, Taber A, Beck H et al (2012) Range-wide declines of a key Neotropical ecosystem architect, the near threatened white-lipped peccary Tayassu pecari. Oryx 46:87–98. https://doi.org/10.1017/S0030605311000421
Arroyo-Rodríguez V, Fahring L, Watling JI et al (2020) Designing optimal human-modified landscapes for forest biodiversity conservation. Ecol Lett 23:1404–1420. https://doi.org/10.1111/ele.13535
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Beck H (2006) A review of Peccary–Palm interactions and their ecological ramifications across the Neotropics. J Mammal 87:519–530. https://doi.org/10.1644/05-mamm-a-174r1.1
Beisiegel B, Sana D, Moranes E (2012) The jaguar in the Atlantic Forest. CAT News 14–18
Bello C, Galetti M, Pizo MA et al (2015) Defaunation affects carbon storage in tropical forests. Sci Adv 1:1DUMMY. https://doi.org/10.1126/sciadv.1501105
Bogoni JA, Graipel ME, de Castilho PV et al (2016) Contributions of the mammal community, habitat structure, and spatial distance to dung beetle community structure. Biodivers Conserv 25:1661–1675. https://doi.org/10.1007/s10531-016-1147-1
Bogoni JA, Peres CA, Ferraz KMPMB (2021) Medium- to large-bodied mammal surveys across the Neotropics are heavily biased against the most faunally intact baseline assemblages. Mammal Review. In press
Bogoni JA, Pires JSR, Graipel ME et al (2018) Wish you were here: How defaunated is the Atlantic Forest biome of its medium- to largebodied mammal fauna? PLoS One 13:1–23. https://doi.org/10.1371/journal.pone.0204515
Bogoni JA, Silva PG da, Peres CA (2019) Co-declining mammal–dung beetle faunas throughout the Atlantic Forest biome of South America. Ecography 42:1803–1818. https://doi.org/10.1111/ecog.04670
Bogoni JA, Peres CA, Ferraz KMPMB (2020) Extent, intensity and drivers of mammal defaunation: a continental scale analysis across the Neotropics. Sci Rep. https://doi.org/10.1038/s41598-020-72010-w
Bogoni JA, Peres CA, Ferraz KMPM (2020b) Effects of mammal defaunation on natural ecosystem services and human well being throughout the entire Neotropical realm. Ecosyst Serv 45:101173. https://doi.org/10.1016/j.ecoser.2020.101173
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 85:1771–1789. https://doi.org/10.1890/03-9000
Canale GR, Peres CA, Guidorizzi CE et al (2012) Pervasive defaunation of forest remnants in a tropical biodiversity hotspot. PLoS One 7(8):e41671. https://doi.org/10.1371/journal.pone.0041671
Cardillo M, Mace GM, Jones KE et al (2005) Evolution: multiple causes of high extinction risk in large mammal species. Science 309:1239–1241. https://doi.org/10.1126/science.1116030
Chao A, Josi L (2015) Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533–2547. https://doi.org/10.2307/41739612
Chao A, Gotelli NJ, Hsieh TC et al (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67. https://doi.org/10.1890/13-0133.1
Chiarello AG (2000) Density and population size of mammals in remnants of Brazilian Atlantic Forest. Conserv Biol 14:1649–1657. https://doi.org/10.1111/j.1523-1739.2000.99071.x
Chiarello AG (2000b) Conservation value of a native forest fragment in a region of extensive agriculture. Rev Bras Biol 60:237–247. https://doi.org/10.1590/s0034-71082000000200007
Culot L, Bovy E, Vaz-de-Mello FZ, Guevara R, Galetti M (2013) Selective defaunation affects dung beetle communities in continuous Atlantic rainforest. Biol Conserv 163:79–89. https://doi.org/10.1016/j.biocon.2013.04.004
Dean W (1996) A ferro e fogo: a história e a devastação da mata atlântica brasileira. Companhia das Letras, São Paulo
Devictor V, Julliard R, Jiguet F (2008) Distribution of specialist and generalist species along spatial gradients of habitat disturbance and fragmentation. Oikos 117:507–514. https://doi.org/10.1111/j.2008.0030-1299.16215.x
Dirzo R, Miranda A (1990) Contemporary neotropical defaunation and forest to structure, John and Diversity Sequel Terborgh. Conserv Biol 4(4):444–447
Dirzo R, Young HS, Galetti M et al (2014) Defaunation in the Anthropocene. Science 345(6195):401–406. https://doi.org/10.1126/science.1251817
Dobbins M, Sollman R, Menke S, Zambrano AA, Broadbent E (2020) An integrated approach to measure hunting intensity and assess its impacts on mammal populations. J Appl Ecol 57:2100–2111. https://doi.org/10.1111/1365-2664.13750
Elschot K, Bakker JP, Temmerman S et al (2015) Ecosystem engineering by large grazers enhances carbon stocks in a tidal salt marsh. Mar Ecol Prog Ser 537:9–21. https://doi.org/10.3354/meps11447
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Galetti M, Dirzo R (2013) Ecological and evolutionary consequences of living in a defaunated world. Biol Conserv 163:1–6. https://doi.org/10.1016/j.biocon.2013.04.020
Galetti M, Donatti CI, Pires AS et al (2006) Seed survival and dispersal of an endemic Atlantic forest palm: The combined effects of defaunation and forest fragmentation. Bot J Linn Soc 151:141–149. https://doi.org/10.1111/j.1095-8339.2006.00529.x
Galetti M, Giacomini HC, Bueno RS et al (2009) Priority areas for the conservation of Atlantic forest large mammals. Biol Conserv 142:1229–1241. https://doi.org/10.1016/j.biocon.2009.01.023
Galetti M, Bovendorp RS, Guevara R (2015a) Defaunation of large mammals leads to an increase in seed predation in the Atlantic forests. Glob Ecol Conserv 3:824–830. https://doi.org/10.1016/j.gecco.2015.04.008
Galetti M, Guevara R, Neves CL et al (2015b) Defaunation affect population and diet of rodents in Neotropical rainforests. Biol Conserv 190:2–7. https://doi.org/10.1016/j.biocon.2015.04.032
Galetti M, Brocardo CR, Begotti RA et al (2017) Defaunation and biomass collapse of mammals in the largest Atlantic forest remnant. Anim Conserv 20:270–281. https://doi.org/10.1111/acv.12311
Gardner TA, Barlow J, Chazdon R et al (2009) Prospects for tropical forest biodiversity in a human-modified world. Ecol Lett 12:561–582. https://doi.org/10.1111/j.1461-0248.2009.01294.x
Gayot M, Henry O, Dubost G, Sabatier D (2004) Comparative diet of the two forest cervids of the genus Mazama in French Guiana. J Trop Ecol 20:31–43. https://doi.org/10.1017/S0266467404006157
Giacomini HC, Galetti M (2013) An index for defaunation. Biol Conserv 163:33–41. https://doi.org/10.1016/j.biocon.2013.04.007
González-Maya JF, Martínez-Meyer E, Medellín R, Ceballos G (2017) Distribution of mammal functional diversity in the Neotropical realm. PloS One 12(4):e0175931. https://doi.org/10.1371/journal.pone.0175931
Graipel ME, Cherem JJ, Monteiro-Filho ELA, Carmignotto AP (2017) Mamíferos da Mata Atlântica. In: Monteiro-Filho ELA, Conte CE (eds) Revisões em Zoologia: Mata Atlântica. Ed. UFPR, Curitiba, pp 391–482
Harmsen BJ, Foster RJ, Silver S, Ostro L, Doncaster CP (2010) Differential use of trails by forest mammals and the implications for camera-trap studies: A case study from Belize. Biotropica 42:126–133. https://doi.org/10.1111/j.1744-7429.2009.00544.x
Herkt KMB, Skidmore AK, Fahr J (2017) Macroecological conclusions based on IUCN expert maps: A call for caution. Glob Ecol Biogeogr 26(8):1–12. https://doi.org/10.1111/geb.12601
Hoskins AJ, Bush A, Gilmore J et al (2016) Downscaling land-use data to provide global 30” estimates of five land-use classes. Ecol Evol 6:3040–3055. https://doi.org/10.1002/ece3.2104
Hsieh TC, Ma KH, Chao A (2020) iNEXT: iNterpolation and EXTrapolation for species diversity. R package version 2.0.20. https://cran.r-project.org/web/packages/iNEXT/index.html
IBGE (2019) Instituto Brasileiro de Geografia e Estatística: Consulta estados. https://www.ibge.gov.br/cidades-e-estados/pr.html
IUCN (2019) The IUCN Red List of Threatened Species. Version 2019-2. https://www.iucnredlist.org
Jorge MLSP, Galetti M, Ribeiro MC, Ferraz KMPMB (2013) Mammal defaunation as surrogate of trophic cascades in a biodiversity hotspot. Biol Conserv 163:49–57. https://doi.org/10.1016/j.biocon.2013.04.018
Kurten EL (2013) Cascading effects of contemporaneous defaunation on tropical forest communities. Biol Conserv 163:22–32. https://doi.org/10.1016/j.biocon.2013.04.025
Kurten EL, Wright SJ, Carson WP, Palmer TM (2015) Hunting alters seedling functional trait composition in a Neotropical forest. Ecology 96:1923–1932. https://doi.org/10.1890/14-1735.1
Lanz B, Dietz S, Swanson T (2018) The expansion of modern agriculture and global biodiversity decline: an integrated assessment. Ecol Econ 144:260–277. https://doi.org/10.1016/j.ecolecon.2017.07.018
Lyra-Jorge MC, Ciochetti G, Pivello VR, Meirelles ST (2008) Comparing methods for sampling large- and medium-sized mammals: camera traps and track plots. Eur J Wildl Res 54:739–744. https://doi.org/10.1007/s10344-008-0205-8
Maack R (2017) Geografa Física do Estado do Paraná, 4th edn. Editora UEPG, Ponta Grossa
MacKenzie NJ, Royle A, Pollock KH, Bailey LL, Hines J (2017) Occupancy estimation and modeling, 2nd edn. Elsevier, London
Magioli M, Ferraz KMPM, de B, Setz EZF, et al (2016) Connectivity maintain mammal assemblages functional diversity within agricultural and fragmented landscapes. Eur J Wildl Res 62:431–446. https://doi.org/10.1007/s10344-016-1017-x
Markl JS, Schleuning M, Forget PM et al (2012) Meta-analysis of the effects of human disturbance on seed dispersal by animals. Conserv Biol 26:1072–1081. https://doi.org/10.1111/j.1523-1739.2012.01927.x
Mbachu HI, Nduka EC, Nja ME (2012) Designing a pseudo R-squared goodness-of-fit measure in generalized linear models. J Math Res 4:148–154. https://doi.org/10.5539/jmr.v4n2p148
Melo FPL, Arroyo-Rodrıguez V, Fahrig L et al (2013) On the hope for biodiversity-friendly tropical landscapes. Trends Ecol Evol 28:461–468. https://doi.org/10.1016/j.tree.2013.01.001
Mittermeier RA, Robles-Gil P, Hoffmann M et al (2004) Hotspots revisited. CEMEX, Mexico
Newbold T, Hill LNHSLL, Contu S et al (2015) Global effects of land use on local terrestrial biodiversity. Nature 520:45–50. https://doi.org/10.1038/nature14324
Nolte S, Müller F, Schuerch M et al (2013) Does livestock grazing affect sediment deposition and accretion rates in salt marshes? Estuar Coast Shelf Sci 135:296–305. https://doi.org/10.1016/j.ecss.2013.10.026
Núñez-regueiro MM, Branch L, Fletcher RJ et al (2015) Spatial patterns of mammal occurrence in forest strips surrounded by agricultural crops of the Chaco region, Argentina. Biol Conserv 187:19–26. https://doi.org/10.1016/j.biocon.2015.04.001
O’Farrill G, Galetti M, Campos-Arceiz A (2013) Frugivory and seed dispersal by tapirs: An insight on their ecological role. Integr Zool 8:4–17. https://doi.org/10.1111/j.1749-4877.2012.00316.x
Oksanen J, Blanchet G, Kindt R et al (2013) Vegan: Community Ecology Package. R package version 2.0–7. http://CRAN.R-project.org/package=vegan
Paglia AP, Fonseca GAB, Rylands AB et al (2012) Annotated checklist of Brazilian mammals. Occas Pap Conserv Biol 6:1–76
PARANÁ (2010) Lista de Espécies de Mamíferos pertencentes à Fauna Silvestre Ameaçadas de Extinção no Estado do Paraná: Decreto № 3.148 de 2004. https://bit.ly/2Qirj9m
Paviolo A, De Angelo C, Ferraz KMPMB et al (2016) A biodiversity hotspot losing its top predator: The challenge of jaguar conservation in the Atlantic Forest of South America. Sci Rep 6:1–16. https://doi.org/10.1038/srep37147
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Koppen-Geiger climate classificatio. Hydrol Earth Syst Sci 11:1643–1644. https://doi.org/10.1002/ppp.421
Pereira AD, Pires Adelino JR, Garcia DAZ et al (2020) Modeling the geographic distribution of Myocastor coypus (Mammalia, Rodentia) in Brazil: establishing priority areas for monitoring and an alert about the risk of invasion. Stud Neotrop Fauna Environ 55:139–148. https://doi.org/10.1080/01650521.2019.1707419
Peres CA (1990) Effects of hunting on Western Amazonian Primate Communities. Biol Conserv 54:47–59. https://doi.org/10.1016/0006-3207(90)90041-M
Peres CA, Emilio T, Schietti J et al (2016) Dispersal limitation induces long-term biomass collapse in overhunted Amazonian forests. Proc Natl Acad Sci USA 113:892–897. https://doi.org/10.1073/pnas.1516525113
Polishchuk LV (2010) The three-quarter-power scaling of extinction risk in Late Pleistocene mammals, and a new theory of the size selectivity of extinction. Evol Ecol Res 12:1–22
Püttker T, Crouzeilles R, Almeida-gomes M et al (2020) Indirect effects of habitat loss via habitat fragmentation: A cross-taxa analysis of forest-dependent species. Biol Conserv 241:108368. https://doi.org/10.1016/j.biocon.2019.108368
Redford KH (2009) The Empty of neotropical forest where the vegetation still appears intact. Sci York 42:412–422
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ribeiro MC, Metzger JP, Martensen AC et al (2009) The Brazilian Atlantic Forest: How much is left, and how is the remaining forest distributed? Implications for conservation. Biol Conserv 142:1141–1153. https://doi.org/10.1016/j.biocon.2009.02.021
Rondinini C, Di Marco M, Chiozza F, Santulli G et al (2011) Global habitat suitability models of terrestrial mammals. Phil Trans R Soc B 366:2633–2641. https://doi.org/10.1098/rstb.2011.0113
Schmidt BR (2005) Monitoring the distribution of pond-breeding amphibians when species are detected imperfectly. Aquat Conserv 15:681–692. https://doi.org/10.1002/aqc.740
Schrama M, Heijning P, Bakker JP et al (2013) Herbivore trampling as an alternative pathway for explaining differences in nitrogen mineralization in moist grasslands. Oecologia 172:231–243. https://doi.org/10.1007/s00442-012-2484-8
Srbek-Araujo AC, Chiarello AG (2013) Influence of camera-trap sampling design on mammal species capture rates and community structures in southeastern Brazil. Biota Neotrop 13:51–62. https://doi.org/10.1590/s1676-06032013000200005
Srbek-Araujo AC, Chiarello AG (2017) Population status of the jaguar Panthera onca in one of its last strongholds in the Atlantic Forest. Oryx 51:246–253. https://doi.org/10.1017/S0030605315001222
Stoner C, Caro T, Mduma S et al (2007) Changes in large herbivore populations across large areas of Tanzania. Afr J Ecol 45:202–215. https://doi.org/10.1111/j.1365-2028.2006.00705.x
Takeda AK, Takeda IJM, Farago PV (2001) Unidades de conservação da região dos campos gerais. Paraná Publ UEPG – Biol Heal Sci 7:57–78. https://doi.org/10.5212/publicatio
Taylor RA, Ryan SJ, Brashares JS, Johnson LR (2016) Hunting, food subsidies, and mesopredator release: the dynamics of crop-raiding baboons in a managed landscape. Ecology 97:951–960. https://doi.org/10.1890/15-0885.1
Terborgh J (2008) The big things that run the world-a sequel to E. O. Wilson. Conserv Biol 2:402–403
Tobler MW, Pitman RL, Mares R, Powell G (2008) An evaluation of camera traps for inventorying large- and medium-sized terrestrial rainforest mammals. Anim Conserv 11:169–178. https://doi.org/10.1111/j.1469-1795.2008.00169.x
Trigo TC, Schneider A, De Oliveira TG et al (2013) Molecular data reveal complex hybridization and a cryptic species of Neotropical wild cat. Curr Biol 23:2528–2533. https://doi.org/10.1016/j.cub.2013.10.046
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Weckel M, Giuliano W, Silver S (2006) Jaguar (Panthera onca) feeding ecology: Distribution of predator and prey through time and space. J Zool 270:25–30. https://doi.org/10.1111/j.1469-7998.2006.00106.x
Wilcoxon F (1945) Individual comparisons by ranking methods. Biometrics Bull 1:80–83
Wilkie DS, Bennett EL, Peres CA, Cunningham AA (2011) The empty forest revisited. Ann N Y Acad Sci 1223:120–128. https://doi.org/10.1111/j.1749-6632.2010.05908.x
Wilson MC, Chen XY, Corlett RT et al (2016) Habitat fragmentation and biodiversity conservation: key findings and future challenges. Landsc Ecol 31:219–227. https://doi.org/10.1007/s10980-015-0312-3
Zimbres B, Peres CA, Machado RB (2017) Terrestrial mammal responses to habitat structure and quality of remnant riparian forests in an Amazonian cattle-ranching landscape. Biologia (Bratisl) 206:283–292. https://doi.org/10.1016/j.biocon.2016.11.033
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
We thank the Postgraduate Program in Biological Sciences of the Universidade Estadual de Londrina and the Coordination for the Upgrading of Higher Education Personnel (CAPES-Funding Code 1689817), for logistical and financial support. JAB is supported by postdoctoral fellowships grants 2018-05970-1 and 2019-11901-5, São Paulo Research Foundation (FAPESP). We thank two anonymous reviewers for their valuable contribution to this manuscript.
Funding
Coordination for the Upgrading of Higher Education Personnel (CAPES). JAB is supported by the São Paulo Research Foundation (FAPESP) postdoctoral fellowship grants 2018-05970-1 and 2019-11901-5.
Author information
Authors and Affiliations
Contributions
ADP, JAB, and MLO conceived the ideas; SB and ADP conducted the data collection; ADP and JAB analyzed the data (defaunation index, taxonomic diversity, rarefaction, and generalized linear mixed model); ADP led the writing with assistance from JAB, SB, and MLO.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Karen E. Hodges.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pereira, A.D., Bogoni, J.A., Bazilio, S. et al. Mammalian defaunation across the Devonian kniferidges and meridional plateaus of the Brazilian Atlantic Forest. Biodivers Conserv 30, 4005–4022 (2021). https://doi.org/10.1007/s10531-021-02288-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-021-02288-3