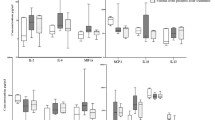
In diffuse large B-cell lymphoma, bone marrow involvement is rarely diagnosed. We compared the properties of bone marrow stromal progenitor cells and the concentration of fibroblast CFU in patients with diffuse large B-cell lymphoma without bone marrow involvement and in healthy donors. It was found that the properties of multipotent mesenchymal stromal cells in patients in the debut of the disease differed considerably from those in healthy donors. In particular, the total cell production in patients was significantly higher than in donors. In multipotent mesenchymal stromal cells of patients, some cell parameters were changes; the mean fluorescence intensity of the adhesion molecule ICAM1 on the cell surface was increased. The mean fluorescence intensity of mesenchymal stromal cell markers (HLA-ABC, CD73 and CD90) was significantly elevated. The relative expression of BMP4, MMP2, FGFR1, and ICAM1 genes in mesenchymal stromal cell was reduced, while the expression of FGFR2 gene was enhanced. Despite the absence of proven involvement of the bone marrow, the properties of mesenchymal stromal cells, the components in the stromal microenvironment niche regulating hemopoiesis are altered in patients with diffuse large B-cell lymphoma.
Similar content being viewed by others
References
Adrian K, Bernhard MK, Breitinger HG, Ogilvie A. Expression of purinergic receptors (ionotropic P2X1-7 and metabotropic P2Y1-11) during myeloid differentiation of HL60 cells. Biochim. Biophys Acta. 2000;1492(1):127-138.
Armitage JO. The changing classification of non-Hodgkin’s lymphomas. CA Cancer J. Clin. 1997;47(6):323-325.
Beltran BE, Quiñones P, Morales D, Malaga JM, Chavez JC, Sotomayor EM, Castillo JJ. Response and survival benefit with chemoimmunotherapy in Epstein-Barr virus-positive diffuse large B-cell lymphoma. Hematol. Oncol. 2018;36(1):93-97.
Chen Q, Yuan Y, Chen T. Morphology, differentiation and adhesion molecule expression changes of bone marrow mesenchymal stem cells from acute myeloid leukemia patients. Mol. Med. Rep. 2014;9(1):293-298.
Chen Y, Cheung KM, Kung HF, Leong JC, Lu WW, Luk KD. In vivo new bone formation by direct transfer of adenoviralmediated bone morphogenetic protein-4 gene. Biochem. Biophys. Res. Commun. 2002;298(1):121-127.
Ding BS, Cao Z, Lis R, Nolan DJ, Guo P, Simons M, Penfold ME, Shido K, Rabbany SY, Rafii S. Divergent angiocrine signals from vascular niche balance liver regeneration and fibrosis. Nature. 2014;505:97-102.
Liu YF, Zhang SY, Chen YY, Shi K, Zou B, Liu J, Yang Q, Jiang H, Wei L, Li CZ, Zhao M, Gabrilovich DI, Zhang H, Zhou J. ICAM-1 deficiency in the bone marrow niche impairs quiescence and repopulation of hematopoietic stem cells. Stem Cell Rep. 2018;11(1):258-273.
Passaro D, Di Tullio A, Abarrategi A, Rouault-Pierre K, Foster K, Ariza-McNaughton L, Montaner B, Chakravarty P, Bhaw L, Diana G, Lassailly F, Gribben J, Bonnet D. Increased vascular permeability in the bone marrow microenvironment contributes to disease progression and drug response in acute myeloid leukemia. Cancer Cell. 2017;32(3):324-341.e6.
Ríus C, Smith JD, Almendro N, Langa C, Botella LM, Marchuk DA, Vary CP, Bernabéu C. Cloning of the promoter region of human endoglin, the target gene for hereditary hemorrhagic telangiectasia type 1. Blood. 1998;92(12):4677-4690.
Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat. Prot. 2008;3(6):1101-1108.
Shipounova IN, Petinati NA, Bigildeev AE, Drize NJ, Sorokina TV, Kuzmina LA, Parovichnikova EN, Savchenko VG. Alterations of the bone marrow stromal microenvironment in adult patients with acute myeloid and lymphoblastic leukemias before and after allogeneic hematopoietic stem cell transplantation. Leuk. Lymphoma. 2017;58(2):408-417.
Shipounova IN, Petrova TV, Svinareva DA, Momotuk KS, Mikhailova EA, Drize NI. Alterations in hematopoietic microenvironment in patients with aplastic anemia. Clin. Transl. Sci. 2009;2(1):67-74.
World Health Organization, International Agency for Research on Cancer WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Swerdlow SH, ed. Lyon, 2017.
Wilkie AO, Patey SJ, Kan SH, van den Ouweland AM, Hamel BC. FGFs, their receptors, and human limb malformations: clinical and molecular correlations. Am. J. Med. Genet. 2002;112(3):266-278.
Yahata T, Ibrahim AA, Muguruma Y, Eren M, Shaffer AM, Watanabe N, Kaneko S, Nakabayashi T, Dan T, Hirayama N, Vaughan DE, Miyata T, Ando K. TGF-β-induced intracellular PAI-1 is responsible for retaining hematopoietic stem cells in the niche. Blood. 2017;130(21):2283-2294
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 1, pp. 33-36, January, 2019
Rights and permissions
About this article
Cite this article
Fastova, E.A., Magomedova, A.U., Petinati, N.A. et al. Bone Marrow Multipotent Mesenchymal Stromal Cells in Patients with Diffuse Large B-Cell Lymphoma. Bull Exp Biol Med 167, 150–153 (2019). https://doi.org/10.1007/s10517-019-04480-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-019-04480-6