Abstract
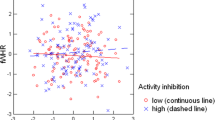
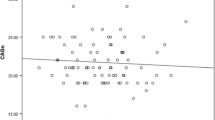
Previous research has linked the facial width-to-height ratio (FWHR) to a host of psychological and behavioral characteristics, primarily in men. In two studies, we examined novel links between FWHR and sex drive. In Study 1, a sample of 145 undergraduate students revealed that FWHR positively predicted sex drive. There were no significant FWHR × sex interactions, suggesting that FWHR is linked to sexuality among both men and women. Study 2 replicated and extended these findings in a sample of 314 students collected from a different Canadian city, which again demonstrated links between the FWHR and sex drive (also in both men and women), as well as sociosexuality and intended infidelity (men only). Internal meta-analytic results confirm the link between FWHR and sex drive among both men and women. These results suggest that FWHR may be an important morphological index of human sexuality.





Similar content being viewed by others
Notes
FWHR remained a significant predictor of sex drive when ethnicity (dichotomized as Caucasian vs. non-Caucasian) was included as a covariate, b = 1.32, SE = .53, t(137) = 2.49, p = .014, partial-r = .21 and when BMI was included as a covariate, b = 1.43, SE = .57, t(136) = 2.52, p = .013, partial-r = .21.
FWHR remained a significant predictor of sex drive when ethnicity (dichotomized as Caucasian vs. non-Caucasian) was included as a covariate, b = .79, SE = .30, t(309) = 2.64, p = .009, partial-r = .15. Unfortunately, body mass index was not collected in Study 2, and thus we could not control for this variable. When the sample was restricted to include only heterosexual participants, results were not meaningfully different from those presented for the full sample.
References
Anderi, C., Hahn, T., Schmidt, A., Moldenhauer, H., Notebaert, K., Clément, C. C., & Windmann, S. (2016). Facial width-to-height ratio predicts psychopathic traits in males. Personality and Individual Differences, 88, 99–101. doi:10.1016/j.paid.2015.08.057.
Anderson, R. A., Bancroft, J., & Wu, F. C. (1992). The effects of exogenous testosterone on sexuality and mood of normal men. Journal of Clinical Endocrinology and Metabolism, 75, 1503–1507.
Arnett, J. J. (2000). Emerging adulthood: A theory of development from the late teens through the twenties. American Psychologist, 55, 469–480. doi:10.1037/0003-066X.55.5.469.
Arnocky, S., Bird, B. M., & Perilloux, C. (2014). An evolutionary perspective on characteristics of physical attractiveness in humans. In A. Rennolds (Ed.), Psychology of interpersonal perception and relationships (pp. 115–155). New York, NY: NOVA Publishers.
Bagatell, C. J., Heiman, J. R., River, J. E., & Bremner, W. J. (1994). Effects of endogenous testosterone and estradiol on sexual behavior in normal young men. Journal of Clinical Endocrinology and Metabolism, 78, 711–716. doi:10.1210/jc.78.3.711.
Baumeister, R. F. (2000). Gender differences in erotic plasticity: The female sex drive as socially flexible and responsive. Psychological Bulletin, 126, 347–374. doi:10.1037/0033-2909.126.3.347.
Baumeister, R. F., Catanese, K. R., & Vohs, K. D. (2001). Is there a gender difference in strength of sex drive? Theoretical views, conceptual distinctions, and a review of relevant evidence. Personality and Social Psychology Review, 5, 242–273. doi:10.1207/S15327957PSPR0503_5.
Bird, B. M., Cid, V., Geniole, S. N., Welker, K. M., Zilioli, S., Maestripieri, D., … Carré, J. M. (2016). Does facial width-to-height ratio map onto variability in men’s testosterone concentrations? Evolution and Human Behavior, 37, 392–398. doi:10.1016/j.evolhumbehav.2016.03.004.
Campbell, B. C., Prossinger, H., & Mbzivo, M. (2005). Timing of pubertal maturation and the onset of sexual behavior among Zimbabwe school boys. Archives of Sexual Behavior, 34, 505–516. doi:10.1007/s10508-005-6276-7.
Cappelletti, M., & Wallen, K. (2016). Increasing women’s sexual desire: The comparative effectiveness of estrogens and androgens. Hormones and Behavior, 78, 178–193. doi:10.1016/j.yhbeh.2015.11.003.
Carré, J. M. (2014). Social status, facial structure, and assertiveness in brown capuchin monkeys. Frontiers in Psychology, 5, 567. doi:10.3389/fpsyg.2014.00567.
Carré, J. M., & McCormick, C. M. (2008). In your face: Facial metrics predict aggressive behaviour in the laboratory and in varsity and professional hockey players. Proceedings of the Royal Society B: Biological Sciences, 275, 2651–2656. doi:10.1098/rspb.2008.0873.
Carré, J. M., McCormick, C. M., & Hariri, A. R. (2011). The social neuroendocrinology of human aggression. Psychoneuroendocrinology, 36, 935–944. doi:10.1016/j.psyneuen.2011.02.001.
Carré, J. M., McCormick, C. M., & Mondloch, C. J. (2009). Facial structure is a reliable cue of aggressive behavior. Psychological Science, 20, 1194–1198. doi:10.1111/j.1467-9280.2009.02423.x.
Carvalho, J., & Nobre, P. (2010). Predictors of women’s sexual desire: The role of psychopathology, cognitive-emotional determinants, relationship dimensions, and medical factors. Journal of Sexual Medicine, 7, 928–937. doi:10.1111/j.1743-6109.2009.01568.x.
Carvalho, J., & Nobre, P. (2011). Predictors of men’s sexual desire: The role of psychological, cognitive-emotional, relational, and medical factors. Journal of Sex Research, 48, 254–262. doi:10.1080/00224491003605475.
Cumming, G. (2013). Understanding the new statistics: Effect sizes, confidence intervals, and meta-analysis. New York: Routledge.
Davidson, J. M., Camargo, C. A., & Smith, E. R. (1979). Effects of androgen on sexual behavior in hypogonadal men. Journal of Clinical Endocrinology and Metabolism, 48, 955–958. doi:10.1210/jcem-48-6-955.
Davis, S. R., & Tran, J. (2001). Testosterone influences libido and well being in women. Trends in Endocrinology and Metabolism, 12, 33–37. doi:10.1016/S1043-2760(00)00333-7.
DeLecce, T. L., Polheber, J. P., & Matchock, R. L. (2014). Sociosexual orientation and 2D:4D ratios in women: Relationship to men’s desirability ratings as a long-term pair bond. Archives of Sexual Behavior, 43, 319–327. doi:10.1007/s10508-013-0201-2.
Dixson, B. J. W., Sulikowski, D., Gouda-Vossos, A., Rantala, M. J., & Brooks, R. C. (2016). The masculinity paradox: Facial masculinity and beardedness interact to determine women’s ratings of men’s facial attractiveness. Journal of Evolutionary Biology, 29, 2311–2320. doi:10.1111/jeb.12958.
Edelstein, R. S., Chopik, W. J., & Kean, E. L. (2011). Sociosexuality moderates the association between testosterone and relationship status in men and women. Hormones and Behavior, 60, 248–255. doi:10.1016/j.yhbeh.2011.05.007.
Eisenegger, C., Haushofer, J., & Fehr, E. (2011). The role of testosterone in social interaction. Trends in Cognitive Sciences, 15, 263–271. doi:10.1016/j.tics.2011.04.008.
Gallup, A. C., White, D. D., & Gallup, D. D. (2007). Handgrip strength predicts sexual behavior, body morphology, and aggression in male college students. Evolution and Human Behavior, 28, 423–429. doi:10.1016/j.evolhumbehav.2007.07.001.
Geniole, S. N., Denson, T. F., Dixson, B. J., Carré, J. M., & McCormick, C. M. (2015). Evidence from meta-analyses of the facial width-to-height ratio as an evolved cue of threat. PLoS ONE, 10, e0132726. doi:10.1371/journal.pone.0132726.
Geniole, S. N., Keyes, A. E., Carré, J. M., & McCormick, C. M. (2014). Fearless dominance mediates the relationship between the facial width-to-height ratio and cheating. Personality and Individual Differences, 57, 59–64. doi:10.1016/j.paid.2013.09.023.
Goetz, A. T., & Causey, K. (2009). Sex differences in perceptions of infidelity: Men often assume the worst. Evolutionary Psychology. doi:10.1177/147470490900700208.
Goetz, S. M., Shattuck, K. S., Miller, R. M., Campbell, J. A., Lozoya, E., Weisfeld, G. E., & Carré, J. M. (2013). Social status moderates the relationship between facial structure and aggression. Psychological Science, 24, 2329–2334. doi:10.1177/0956797613493294.
González-Álvarez, J. (2017). Perception of sexual orientation from facial structure: A study with artificial face models. Archives of Sexual Behavior, 46, 1251–1260. doi:10.1007/s10508-016-0929-6.
Gray, P. B. (2003). Marriage, parenting, and testosterone variation among Kenyan Swahili men. American Journal of Physical Anthropology, 122, 279–286. doi:10.1002/ajpa.10293.
Halpern, C. T., Udry, J. R., Campbell, B., Suchindran, C., & Mason, G. A. (1994). Testosterone and religiosity as predictors of sexual attitudes and activity among adolescent males: A biosocial model. Journal of Biosocial Science, 26, 217–234. doi:10.1017/S0021932000021258.
Halpern, C. T., Udry, J. R., & Suchindran, C. (1997). Testosterone predicts initiation of coitus in adolescent females. Psychosomatic Medicine, 59, 161–171. doi:10.1097/00006842-199703000-00008.
Halpern, C. T., Udry, J. R., & Suchindran, C. (1998). Monthly measures of salivary testosterone predict sexual activity in adolescent males. Archives of Sexual Behavior, 27, 445–465. doi:10.1023/A:1018700529128.
Haselhuhn, M. P., Ormiston, M. E., & Wong, E. M. (2015). Men’s facial width-to-height ratio predicts aggression: A meta-analysis. PLoS ONE, 10, e0122637. doi:10.1371/journal.pone.0122637.
Haselhuhn, M. P., & Wong, E. M. (2012). Bad to the bone: Facial structure predicts unethical behaviour. Proceedings of the Royal Society of London B: Biological Sciences, 279, 571–576. doi:10.1098/rspb.2011.1193.
Hayes, A. F. (2013). Introduction to mediation, moderation, and conditional process analysis. New York, NY: Guilford.
Hayes, A. F. (n.d.). Macro and script rules and frequently asked questions. Retrieved July 5, 2016 from http://www.afhayes.com/macrofaq.html.
Hehman, E., Leitner, J. B., Deegan, M. P., & Gaertner, S. L. (2013). Facial structure is indicative of explicit support for prejudicial beliefs. Psychological Science, 24, 289–296. doi:10.1177/0956797612451467.
Hodges-Simeon, C. R., Hanson Sobraske, K. N., Samore, T., Gurven, M., & Gaulin, S. J. C. (2016). Facial width-to-height ratio (fWHR) is not associated with adolescent testosterone levels. PLoS ONE, 11, e0153083. doi:10.1371/journal.pone.0153083.
Isidori, A. M., Giannetta, E., Gianfrilli, D., Greco, E. A., Bonifacio, V., Aversa, A., … Lenzi, A. (2005). Effects of testosterone on sexual function in men: Results of a meta-analysis. Clinical Endocrinology, 63, 381–394. doi:10.1111/j.1365-2265.2005.02350.x.
Jannini, E. A., Screponi, E., Carosa, E., Pepe, M., Giudice, F. L., Trimarchi, F., et al. (1999). Lack of sexual activity from erectile dysfunction is associated with a reversible reduction in serum testosterone. International Journal of Andrology, 22, 385–392. doi:10.1046/j.1365-2605.1999.00196.x.
Kingsberg, S. (2007). Testosterone treatment for hypoactive sexual desire disorder in postmenopausal women. Journal of Sexual Medicine, 4, 227–234. doi:10.1111/j.1743-6109.2007.00449.x.
Kwan, M., Greenleaf, W. J., Mann, J., Crapo, L., & Davidson, J. M. (1983). The nature of androgen action on male sexuality: A combined laboratory-self-report study on hypogonadal men. Journal of Clinical Endocrinology and Metabolism, 57, 557–562. doi:10.1210/jcem-57-3-557.
Lefevre, C. E., Lewis, G. J., Bates, T. C., Dzhelyova, M., Coetzee, V., Deary, I. J., & Perrett, D. I. (2012). No evidence for sexual dimorphism of facial width-to-height ratio in four large adult samples. Evolution and Human Behavior, 33, 623–627. doi:10.1016/j.evolhumbehav.2012.03.002.
Lefevre, C. E., Lewis, G. J., Perrett, D. I., & Penke, L. (2013). Telling facial metrics: Facial width is associated with testosterone levels in men. Evolution and Human Behavior, 34, 273–279. doi:10.1016/j.evolhumbehav.2013.03.005.
Lefevre, C. E., Wilson, V. A. D., Morton, F. B., Brosnan, S. F., Paukner, A., & Bates, T. C. (2014). Facial width-to-height ratio relates to alpha status and assertive personality in capuchin monkeys. PLoS ONE, 9, e93369. doi:10.1371/journal.pone.0093369.
Lewis, G. J., Lefevre, C. E., & Bates, T. C. (2012). Facial width-to-height ratio predicts achievement drive in US presidents. Personality and Individual Differences, 52, 855–857. doi:10.1016/j.paid.2011.12.030.
Massar, K., & Buunk, A. P. (2009). The effect of a subliminally primed context on intrasexual competition depends on individual differences in sex drive. Journal of Research in Personality, 43, 691–694. doi:10.1016/j.jrp.2009.02.004.
McCoy, N. L., & Davidson, J. M. (1985). A longitudinal study of the effects of menopause on sexuality. Maturitas, 7, 203–210. doi:10.1016/0378-5122(85)90041-6.
Ostovich, J. M., & Sabini, J. (2004). How are sociosexuality, sex drive, and lifetime number of sexual partners related? Personality and Social Psychology Bulletin, 30, 1255–1266. doi:10.1177/0146167204264754.
Özener, B. (2012). Facial width-to-height ratio in a Turkish population is not sexually dimorphic and is unrelated to aggressive behavior. Evolution and Human Behavior, 33, 169–173. doi:10.1016/j.evolhumbehav.2011.08.001.
Penke, L., & Asendorpf, J. B. (2008). Beyond global sociosexual orientations: A more differentiated look at sociosexuality and its effects on courtship and romantic relationships. Journal of Personality and Social Psychology, 95, 1113–1135. doi:10.1037/0022-3514.95.5.1113.
Persky, H., Lief, H. I., Strauss, D., Miller, W. R., & O’Brien, C. P. (1978). Plasma testosterone level and sexual behavior of couples. Archives of Sexual Behavior, 7, 157–173. doi:10.1007/BF01542376.
Puts, D. A., Pope, L. E., Hill, A. K., Cardenas, R. A., Welling, L. L., Wheatley, J. R., & Breedlove, S. M. (2015). Fulfilling desire: Evidence for negative feedback between men’s testosterone, sociosexual psychology, and sexual partner number. Hormones and Behavior, 70, 14–21. doi:10.1016/j.yhbeh.2015.01.006.
Roney, J. R., & Simmons, Z. L. (2013). Hormonal predictors of sexual motivation in natural menstrual cycles. Hormones and Behavior, 63, 636–645. doi:10.1016/j.yhbeh.2013.02.013.
Schiavi, R. C., White, D., Mandeli, J., & Levine, A. C. (1997). Effects of testosterone administration on sexual behavior and mood in men with erectile dysfunction. Archives of Sexual Behavior, 26, 231–241. doi:10.1023/A:1024518730222.
Schreiner-Engel, P., Schiavi, R. C., Smith, H., & White, D. (1981). Sexual arousability and the menstrual cycle. Psychosomatic Medicine, 43, 199–214. doi:10.1097/00006842-198106000-00002.
Simon, J., Braunstein, G., Nachtigall, L., Utian, W., Katz, M., Miller, S., et al. (2005). Testosterone patch increases sexual activity and desire in surgically menopausal women with hypoactive sexual desire disorder. Journal of Clinical Endocrinology and Metabolism, 90, 5226–5233. doi:10.1210/jc.2004-1747.
Sisk, C. L. (2016). Hormone-dependent adolescent organization of socio-sexual behaviors in mammals. Current Opinion in Neurobiology, 38, 63–68. doi:10.1016/j.conb.2016.02.004.
Skorska, M. N., Geniole, S. N., Vrysen, B. M., McCormick, C. M., & Bogaert, A. F. (2015). Facial structure predicts sexual orientation in both men and women. Archives of Sexual Behavior, 44, 1377–1394. doi:10.1007/s10508-014-0454-4.
Snyder, P. J., Bhasin, S., Cunningham, G. R., Matsumoto, A. M., Stephens-Shields, A. J., Cauley, J. A., … Ellenberg, S. S. (2016). Effects of testosterone treatment in older men. New England Journal of Medicine, 374, 611–624. doi:10.1056/NEJMoa1506119.
Stirrat, M., & Perrett, D. I. (2012). Face structure predicts cooperation: Men with wider faces are more generous to their in-group when out-group competition is salient. Psychological Science, 23, 718–722. doi:10.1177/0956797611435133.
Tinlin, R. M., Watkins, C. D., Welling, L. L., DeBruine, L. M., Al-Dujaili, E. A., & Jones, B. C. (2013). Perceived facial adiposity conveys information about women’s health. British Journal of Psychology, 104, 235–248. doi:10.1111/j.2044-8295.2012.02117.x.
Travison, T. G., Morley, J. E., Araujo, A. B., O’Donnell, A. B., & McKinlay, J. B. (2006). The relationship between libido and testosterone levels in aging men. Journal of Clinical Endocrinology and Metabolism, 91, 2509–2513. doi:10.1210/jc.2005-2508.
Udry, J. R., Talbert, L. M., & Morris, N. M. (1986). Biosocial foundations for adolescent female sexuality. Demography, 23, 217–230.
Valentine, K. A., Li, N. P., Penke, L., & Perrett, D. I. (2014). Judging a man by the width of his face: The role of facial ratios and dominance in mate choice at speed-dating events. Psychological Science, 25, 806–811. doi:10.1177/0956797613511823.
van Anders, S. M., Hamilton, L. D., Schmidt, N., & Watson, N. V. (2007). Associations between testosterone secretion and sexual activity in women. Hormones and Behavior, 51, 477–482. doi:10.1016/j.yhbeh.2007.01.003.
van Anders, S. M., Hamilton, L. D., & Watson, N. V. (2007). Multiple partners are associated with higher testosterone in North American men and women. Hormones and Behavior, 51, 454–459. doi:10.1016/j.yhbeh.2007.01.002.
van Anders, S. M., & Watson, N. V. (2006). Relationship status and testosterone in North American heterosexual and non-heterosexual men and women: Cross-sectional and longitudinal data. Psychoneuroendocrinology, 31, 715–723. doi:10.1016/j.psyneuen.2006.01.008.
Verdonck, A., Gaethofs, M., Carels, C., & de Zegher, F. (1999). Effects of low-dose testosterone treatment on craniofacial growth in boys with delayed puberty. European Journal of Orthodontics, 21, 137–143. doi:10.1093/ejo/21.2.137.
Wallen, K. (1995). The evolution of female sexual desire. In P. Abramson & S. Pinkerton (Eds.), Sexual nature, sexual culture (pp. 57–79). Chicago, IL: University of Chicago Press.
Welker, K. M., Bird, B. M., & Arnocky, S. (2016). Commentary: Facial width-to-height ratio is not associated with adolescent testosterone levels. Frontiers in Evolutionary Psychology and Neuroscience, 7, 1745. doi:10.3389/fpsyg.2016.01745.
Weston, E. M., Friday, A. E., & Liò, P. (2007). Biometric evidence that sexual selection has shaped the hominin face. PLoS ONE, 2, e710. doi:10.1371/journal.pone.0000710.
Whitehouse, A. J., Gilani, S. Z., Shafait, F., Mian, A., Tan, D. W., Maybery, M. T., … Eastwood, P. (2015). Prenatal testosterone exposure is related to sexually dimorphic facial morphology in adulthood. Proceedings of the Royal Society B: Biological Sciences, 282, 20151351.
Wilson, V., Lefevre, C. E., Morton, F. B., Brosnan, S. F., Paukner, A., & Bates, T. C. (2014). Personality and facial morphology: Links to assertiveness and neuroticism in capuchins (Sapajus [Cebus] apella). Personality and Individual Differences, 58, 89–94. doi:10.1016/j.paid.2013.10.008.
Acknowledgements
Funding support came from the Canadian Institutes of Health Research, Canada Research Chairs Program (Grant Nos. #950-203794; #950-229048).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included each study.
Rights and permissions
About this article
Cite this article
Arnocky, S., Carré, J.M., Bird, B.M. et al. The Facial Width-to-Height Ratio Predicts Sex Drive, Sociosexuality, and Intended Infidelity. Arch Sex Behav 47, 1375–1385 (2018). https://doi.org/10.1007/s10508-017-1070-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10508-017-1070-x