Abstract
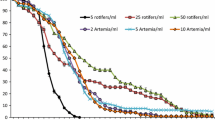
Metamorphosis induction cues (by chemical mediation or direct contact) were tested in Paracentrotus lividus using three different macroalgae treatments: Corallina sp., Palmaria palmata, and Laminaria digitata. Higher percentages of metamorphosis were reached in Paracentrotus lividus larvae by direct contact with a Palmaria palmata substrate.





Similar content being viewed by others
References
Andrew N, Agatsuma Y, Ballesteros E et al (2002) Status and management of world sea urchin fisheries. Oceanogr Mar Biol Annu Rev 40:343–425
Barillé-Boyer AL, Gruet Y, Barillé L, Harin N (2004) Temporal changes in community structure of tide pools following the “Erika” oil spill. Aquat Living Resour 17:323–328
Basuyaux O, Blin J-L (1998) Use of maize as a food source for sea urchins in a recirculating rearing system. Aquac Int 6:233–247
Boudouresque CF (1987) Colloque international sur Paracentrotus lividus et les oursins comestibles. GIS POSIDONIE, Faculté des sciences de Luminy
Boudouresque CF, Verlaque M (2013) Paracentrotus lividus. In: Lawrence JM (ed) Sea urchins: biology and ecology, 3rd edn. Elsevier, Amsterdam, pp 297–327
Brundu G, Monleón LV, Vallainc D, Carboni S (2016) Effects of larval diet and metamorphosis cue on survival and growth of sea urchin post-larvae (Paracentrotus lividus; Lamarck, 1816). Aquaculture 465:265–271
Cameron RA, Schroeter SC (1980) Sea urchin recruitment: effect of substrate selection on juvenile distribution. Mar Ecol Prog Ser 2:243–247
Carboni S, Kelly MS, Hughes AD, Vignier J, Atack T, Migaud H (2014) Evaluation of flow through culture technique for commercial production of sea urchin (Paracentrotus lividus) larvae. Aquac Res 45:768–772
Conand C, Sloan N (1989) World fisheries for echinoderms. In: Caddy JF (ed) Marine invertebrate fisheries: their assessment and management. Wiley, Hoboken, pp 647–663
Cook E, Kelly MS (2007) Effect of variation in the protein value of the red macroalga Palmaria palmata on the feeding, growth and gonad composition of the sea urchins Psammechinus miliaris and Paracentrotus lividus (Echinodermata). Aquaculture 270:207–217
Cook EJ, Kelly MS (2009) Co-culture of the sea urchin Paracentrotus lividus and the edible mussel Mytilus edulis L. on the west coast of Scotland, United Kingdom. J Shellfish Res 28:553–559
Cook E, Hughes AD, Orr H et al (2007) Influence of dietary protein on essential fatty acids in the gonadal tissue of the sea urchins Psammechinus miliaris and Paracentrotus lividus (Echinodermata). Aquaculture 273:586–594
De la Uz S, Carrasco JF, Rodríguez C, Anadon N (2013) Metamorphosis, growth and survival of early juveniles of Paracentrotus lividus (Echinodermata: Echinoidea): effects of larval diet and settlement inducers. Cah Biol Mar 54:691–695
Dworjanyn SA, Pirozzi I (2008) Induction of settlement in the sea urchin Tripneustes gratilla by macroalgae, biofilms and conspecifics: a role for bacteria? Aquaculture 274:268–274
FAO (2016) FAO yearbook. Fishery and Aquaculture Statistics. 2014. Rome, Italy
García-Bueno N, Turpin V, Cognie B, Dumay J, Morançais M, Amat M, Pédron JM, Atucha AM, Fleurence J, Decottignies P (2016) Can the European abalone Haliotis tuberculata survive on an invasive algae? A comparison of the nutritional value of the introduced Grateloupia turuturu and the native Palmaria palmata, for the commercial European abalone industry. J Appl Phycol 28:2427–2433
Gosselin P, Jangoux M (1996) Induction of metamorphosis in Paracentrotus lividus larvae (Echinodermata, Echinoidea). Oceanol Acta 19:293–296
Grosjean P (2001) Growth model of the reared sea urchin Paracentrotus lividus. Dissertation, Université Libre de Bruxelles
Grosjean P, Spirlet C, Gosselin P et al (1998) Land-based, closed-cycle echiniculture of Paracentrotus lividus (Lamarck) (Echinoidea: Echinodermata): a long-term experiment at a pilot scale. J Shellfish Res 17:1523–1531
Hadfield MG, Paul VJ (2001) Natural chemical cues for settlement and metamorphosis of marine invertebrate larvae. In: McClintock JB, Baker BJ (eds) Marine chemical ecology. CRC Press LLC, Boca Raton, pp 431–461
Harrold C, Pearse JS (1987) The ecological role of echinoderms in kelp forests. In: Jangoux M, Lawrence JM (eds) Echinoderm studies. CRC Press, Boca Raton, pp 137–233
Kalvass PE, Hendrix JM (1997) The California red sea urchin, Strongylocentrotus franciscanus, fishery: catch, effort, and management trends. Mar Fish Rev 59:1–17
Li J-Y, Rahim S, Satuito CG, Kitamura H (2004) Characterization of the active substances in water conditioned by the coralline red alga Corallina pilulifera as inducers of metamorphosis in larvae of the sea urchin Anthocidaris crassispina. Sessile Organisms 21:41–46
Liu H, Kelly MS, Cook EJ, Black K, Orr H, Zhu JX, Dong SL (2007) The effect of diet type on growth and fatty-acid composition of sea urchin larvae, I. Paracentrotus lividus (Lamarck, 1816) (Echinodermata). Aquaculture 264:247–262
Orvain F, Martinez A, Desoche E, Claquin P (2015) Chemical interaction between epilitic microphytobenthic biofilm and larval development of the sea urchin Paracentrotus lividus. M. Boutouil & S. Leboulanger, ESITC Caen, pp 239–247
Pearce CM, Scheibling RE (1990) Induction of metamorphosis of larvae of the green sea urchin, Strongylocentrotus droebachiensis, by coralline red algae. Biol Bull 179:304–311
Pearce CM, Scheibling RE (1991) Effect of macroalgae, microbial films, and conspecifics on the induction of metamorphosis of the green sea urchin Strongylocentrotus droebachiensis (Müller). J Exp Mar Biol Ecol 147:147–162
Pearse JS (2006) Ecological role of purple sea urchins. Science 314:940–941
Rial D, Rial P, Casal A, Costoya N, Costas D (2018) Induction of settlement, growth and survival of juveniles of Paracentrotus lividus. Aquaculture 483:16–20
Swanson RL, Williamson JE, De Nys R et al (2004) Induction of settlement of larvae of the sea urchin Holopneustes purpurascens by histamine from a host alga. Biol Bull 206:161–172
Swanson RL, Byrne M, Prowse TAA, Mos B, Dworjanyn SA, Steinberg PD (2012) Dissolved histamine: a potential habitat marker promoting settlement and metamorphosis in sea urchin larvae. Mar Biol 159:915–925
Vadas RL, Beal B, Dowling T, Fegley JC (2000) Experimental field tests of natural algal diets on gonad index and quality in the green sea urchin, Strongylocentrotus droebachiensis: a case for rapid summer production in post-spawned animals. Aquaculture 182:115–135
Valentine JF, Heck KL (1991) The role of sea urchin grazing in regulating subtropical seagrass meadows: evidence from field manipulations in the northern Gulf of Mexico. J Exp Mar Biol Ecol 154:215–230
Zupo V, Glaviano F, Caramiello D, Mutalipassi M (2018) Effect of five benthic diatoms on the survival and development of Paracentrotus lividus post-larvae in the laboratory. Aquaculture 495:13–20
Acknowledgements
The authors wish to thank Clément Chauvin for his work during experiments and Richard Chagneau for providing the living resources. They are also grateful to Carol Robins for correcting the English of this communication.
Funding
This study was supported by the European project “Tools for Assessment and Planning of Aquaculture Sustainability” funded by the EU H2020 research and innovation program under Grant Agreement No 678396.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants performed by any of the authors.
All applicable international and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Castilla-Gavilán, M., Turpin, V., Buzin, F. et al. Optimizing metamorphosis in Paracentrotus lividus aquaculture using alternative macroalgae species to Corallina sp.. Aquacult Int 26, 1511–1518 (2018). https://doi.org/10.1007/s10499-018-0305-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-018-0305-8