Abstract
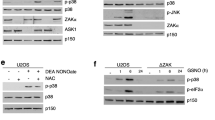
Copper is an essential micronutrient involved in many redox reactions in human cells. However, a high concentration of copper, intake from the environment or abnormal accumulation within cells because of genetic mutation, leads to cell toxicity. This is attributable to oxidative damage, altered gene expression, and functional impairment of the mitochondria. Copper stress also alters the morphology of the nucleolus, but the process has not been fully elucidated. In this study, cells were treated with copper sulfate at 3–9 ppm and examined if a high dose of copper would block ribosome biogenesis. With the incorrect distribution of nucleolar proteins nucleophosmin and fibrillarin to the nucleoplasm, ribosomal RNA (rRNA) processing was impaired; 34S rRNA from an abnormal A2 cut increased, and downstream pre-rRNAs decreased. The under-accumulation of 60S subunits was detected using sucrose gradients. From transcriptome analysis, ribosome synthesis–related genes were misregulated. Blockage in ribosome synthesis under copper-treatment induced nucleolar stress and triggered p53-independent apoptosis pathways. Thus, nucleolar stress is one cause of cell death under copper exposure.









Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
National Research Council (U.S.). Subcommittee on the Tenth Edition of the RDAs., National Institutes of Health (U.S.), National Research Council (U.S.). Committee on Dietary Allowances (1989) Recommended dietary allowances. National Academy Press, Washington, D.C.
Castresana J, Lubben M, Saraste M, Higgins DG (1994) Evolution of cytochrome-oxidase, an enzyme older than atmospheric oxygen. EMBO J 13:2516–2525
Fukai T, Ushio-Fukai M (2011) Superoxide dismutases: role in redox signaling, vascular function, and diseases. Antioxid Redox Sign 15:1583–1606
Klinman JP (2006) The copper-enzyme family of dopamine beta-monooxygenase and peptidylglycine alpha-hydroxylating monooxygenase: resolving the chemical pathway for substrate hydroxylation. J Biol Chem 281:3013–3016
Czlonkowska A, Litwin T, Dusek P, et al. (2018) Wilson disease. Nat Rev Dis Primers 4
Rodriguez-Castro KI, Hevia-Urrutia FJ, Sturniolo GC (2015) Wilson’s disease: A review of what we have learned. World J Hepatol 7:2859–2870
Nayak NC, Chitale AR (2013) Indian childhood cirrhosis (ICC) & ICC-like diseases: The changing scenario of facts versus notions. Indian J Med Res 137:1029–1042
Flemming CA, Trevors JT (1989) Copper toxicity and chemistry in the environment - a review. Water Air Soil Poll 44:143–158
National Research Council (U.S.). Committee on Copper in Drinking Water (2000) Copper in drinking water. National Academy Press, Washington, D.C.
Brewer GJ (2015) Copper-2 ingestion, plus increased meat eating leading to increased copper absorption, are major factors behind the current epidemic of alzheimer’s disease. Nutrients 7:10053–10064
Squitti R, Barbati G, Rossi L et al (2006) Excess of nonceruloplasmin serum copper in AD correlates with MMSE, CSF beta-amyloid, and h-tau. Neurology 67:76–82
Squitti R, Ghidoni R, Siotto M et al (2014) Value of serum nonceruloplasmin copper for prediction of mild cognitive impairment conversion to alzheimer disease. Ann Neurol 75:574–580
Hureau C, Faller P (2009) A beta-mediated ROS production by Cu ions: Structural insights, mechanisms and relevance to Alzheimer’s disease. Biochimie 91:1212–1217
Valko M, Jomova K, Rhodes CJ, Kuca K, Musilek K (2016) Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch Toxicol 90:1–37
Tao TY, Gitlin JD (2003) Hepatic copper metabolism: insights from genetic disease. Hepatology 37:1241–1247
Aston NS, Watt N, Morton IE, Tanner MS, Evans GS (2000) Copper toxicity affects proliferation and viability of human hepatoma cells (HepG2 line). Hum Exp Toxicol 19:367–376
Cecconi I, Scaloni A, Rastelli G et al (2002) Oxidative modification of aldose reductase induced by copper ion - Definition of the metal-protein interaction mechanism. J Biol Chem 277:42017–42027
Hatori Y, Clasen S, Hasan NM, Barry AN, Lutsenko S (2012) Functional partnership of the copper export machinery and glutathione balance in human cells. J Biol Chem 287:26678–26687
Gaetke LM, Chow-Johnson HS, Chow CK (2014) Copper: toxicological relevance and mechanisms. Arch Toxicol 88:1929–1938
Hernandezperez O, Luna G, Mercado E, Delgado N, Rosado A (1989) Effect of cupric ions on the initiation protein-synthesis rate in the human-endometrium. Contraception 40:751–761
Pytharopoulou S, Kournoutou GG, Leotsinidis M, Georgiou CD, Kalpaxis DL (2013) Cadmium versus copper toxicity: Insights from an integrated dissection of protein synthesis pathway in the digestive glands of mussel Mytilus galloprovincialis. J Hazard Mater 260:263–271
Viarengo A, Pertica M, Mancinelli G, Capelli R, Orunesu M (1980) Effects of copper on the uptake of amino-acids, on protein-synthesis and on atp content in different tissues of mytilus-galloprovincialis lam. Mar Environ Res 4:145–152
Um HY, Kong HG, Lee HJ et al (2013) Altered gene expression and intracellular changes of the viable but nonculturable state in ralstonia solanacearum by copper treatment. Plant Pathology J 29:374–385
Kournoutou GG, Giannopoulou PC, Sazakli E, Leotsinidis M, Kalpaxis DL (2017) Oxidative damage of 18S and 5S ribosomal RNA in digestive gland of mussels exposed to trace metals. Aquat Toxicol 192:136–147
Oguz EO, Abban G, Kutlubay R, Turgut S, Enli Y, Erdogan D (2007) Transmission electron microscopy study of the effects of cadmium and copper on fetal rat liver tissue. Biol Trace Elem Res 115:127–135
Aliza D, Tey CL, Ismail IS, Kuah MK, Shu-Chien AC, Muhammad TST (2012) The ribosomal protein L19 mRNA is induced by copper exposure in the swordtail fish, Xiphophorus helleri. Mol Biol Rep 39:4823–4829
Awad D, Prattes M, Kofler L et al (2019) Inhibiting eukaryotic ribosome biogenesis. BMC Biol 17:46
McIntosh KB, Bonham-Smith PC (2005) The two ribosomal protein L23A genes are differentially transcribed in Arabidopsis thaliana. Genome 48:443–454
Tsang T, Posimo JM, Gudiel AA, Cicchini M, Feldser DM, Brady DC (2020) Copper is an essential regulator of the autophagic kinases ULK1/2 to drive lung adenocarcinoma. Nat Cell Biol 22:412–424
Konarikova K, Perdikaris GA, Gbelcova H et al (2016) Autophagy in MCF-7 cancer cells induced by copper complexes. Pharmacol Rep 68:1221–1224
Tanida I, Ueno T, Kominami E (2008) LC3 and autophagy. Methods Mol Biol 445:77–88
Ossareh-Nazari B, Bonizec M, Cohen M et al (2010) Cdc48 and Ufd3, new partners of the ubiquitin protease Ubp3, are required for ribophagy. EMBO Rep 11:548–554
Lindstrom MS (2011) NPM1/B23: a multifunctional chaperone in ribosome biogenesis and chromatin remodeling. Biochem Res Int 2011:195209
Rodriguez-Corona U, Sobol M, Rodriguez-Zapata LC, Hozak P, Castano E (2015) Fibrillarin from archaea to human. Biol Cell 107:159–174
Tafforeau L, Zorbas C, Langhendries JL et al (2013) The complexity of human ribosome biogenesis revealed by systematic nucleolar screening of pre-rrna processing factors. Mol Cell 51:539–551
James A, Wang YB, Raje H, Rosby R, DiMario P (2014) Nucleolar stress with and without p53. Nucleus 5:402–426
Yang K, Yang J, Yi J (2018) Nucleolar Stress: hallmarks, sensing mechanism and diseases. Cell Stress 2:125–140
Kruse JP, Gu W (2009) Modes of p53 regulation. Cell 137:609–622
Yogosawa S, Yoshida K (2018) Tumor suppressive role for kinases phosphorylating p53 in DNA damage-induced apoptosis. Cancer Sci 109:3376–3382
Donati G, Brighenti E, Vici M et al (2011) Selective inhibition of rRNA transcription downregulates E2F–1: a new p53-independent mechanism linking cell growth to cell proliferation. J Cell Sci 124:3017–3028
Iadevaia V, Caldarola S, Biondini L et al (2010) PIM1 kinase is destabilized by ribosomal stress causing inhibition of cell cycle progression. Oncogene 29:5490–5499
Li J, Yu L, Zhang HZ et al (2009) Down-regulation of pescadillo inhibits proliferation and tumorigenicity of breast cancer cells. Cancer Sci 100:2255–2260
Baker ZN, Cobine PA, Leary SC (2017) The mitochondrion: a central architect of copper homeostasis. Metallomics 9:1501–1512
Cobine PA, Pierrel F, Winge DR (2006) Copper trafficking to the mitochondrion and assembly of copper metalloenzymes. Bba-Mol Cell Res 1763:759–772
Xu WJ, Barrientos T, Andrews NC (2013) Iron and copper in mitochondrial diseases. Cell Metab 17:319–328
Liddell JR, White AR (2018) Nexus between mitochondrial function, iron, copper and glutathione in Parkinson’s disease. Neurochem Int 117:126–138
Bustos RI, Jensen EL, Ruiz LM et al (2013) Copper deficiency alters cell bioenergetics and induces mitochondrial fusion through up-regulation of MFN2 and OPA1 in erythropoietic cells. Biochem Bioph Res Co 437:426–432
Shao YZ, Zhao HJ, Wang Y, Liu JJ, Zong H, Xing MW (2019) Copper-mediated mitochondrial fission/fusion is associated with intrinsic apoptosis and autophagy in the testis tissues of chicken. Biol Trace Elem Res 188:468–477
Li SW, Zhao HJ, Wang Y et al (2017) The inflammatory responses in Cu-mediated elemental imbalance is associated with mitochondrial fission and intrinsic apoptosis in Gallus gallus heart. Chemosphere 189:489–497
Puno MR, Patel NA, Moller SG, Robinson CV, Moody PCE, Odell M (2013) Structure of Cu(I)-Bound DJ-1 reveals a biscysteinate metal binding site at the homodimer interface: insights into mutational inactivation of DJ-1 in parkinsonism. J Am Chem Soc 135:15974–15977
Macomber L, Imlay JA (2009) The iron-sulfur clusters of dehydratases are primary intracellular targets of copper toxicity. P Natl Acad Sci USA 106:8344–8349
Brancaccio D, Gallo A, Piccioli M, Novellino E, Ciofi-Baffoni S, Banci L (2017) [4Fe-4S] Cluster Assembly in Mitochondria and Its Impairment by Copper. J Am Chem Soc 139:719–730
Kimura S, Suzuki T (2015) Iron-sulfur proteins responsible for RNA modifications. Biochim Biophys Acta 1853:1272–1283
Yarunin A, Panse VG, Petfalski E, Dez C, Tollervey D, Hurt EC (2005) Functional link between ribosome formation and biogenesis of iron-sulfur proteins. EMBO J 24:580–588
Kispal G, Sipos K, Lange H et al (2005) Biogenesis of cytosolic ribosomes requires the essential iron-sulphur protein Rli1p and mitochondria. EMBO J 24:589–598
Burkhead JL, Ralle M, Wilmarth P, David L, Lutsenko S (2011) Elevated copper remodels hepatic RNA processing machinery in the mouse model of wilson’s disease. J Mol Biol 406:44–58
Han DMR, Choi MR, Jung KH et al (2012) Proteomic analysis of the copper ion-induced stress response in a human embryonic carcinoma cell line. Int J Toxicol 31:397–406
Eyckmans M, Benoot D, Van Raemdonck GA et al (2012) Comparative proteomics of copper exposure and toxicity in rainbow trout, common carp and gibel carp. Comp Biochem Physiol Part D Genomics Proteomics 7:220–232
Yu HT, Wang D, Zou LY et al (2018) Proteomic alterations of brain subcellular organelles caused by low-dose copper exposure: implication for Alzheimer’s disease. Arch Toxicol 92:1363–1382
Acknowledgments
We thank the Join Center for Instruments and Researches, and the College of Bioresources and Agriculture of National Taiwan University, as well as the staff of the Technology commons, College of Life Science, National Taiwan University for technical support. We thank Dr. Louis Grillet for editing this article. This work was financially supported by the Ministry of Science and Technology of Taiwan under contracts No. MOST 106-2313-B-002-031-MY3, MOST 109-2313-B-002-023-MY3 and National Taiwan University Career Development Project (108L7862; 109L7862; 110L7862).
Author information
Authors and Affiliations
Contributions
KYL conceived and coordinated the study and wrote the paper. CHC, YTC, and YWY designed, performed, and analyzed the experiments. All the authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest with the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10495_2021_1692_MOESM1_ESM.tif
Supplementary file1 Fig S1. The cell cycle of HEK293T cells is unaffected by Cu(II) exposure Cells treated with different concentrations of Cu(II) were stained with propidium iodide (PI) and analyzed by flow cytometry (n=3) (TIF 5104 kb)
10495_2021_1692_MOESM2_ESM.tif
Supplementary file2 Fig S2 Cell viability of MDA-MB-231 revealed by MTT assay. MDA-MB-231 cells were treated with different concentrations of Cu(II) for 24 h and cell viability was examined by MTT assay (n=6) (TIF 3823 kb)
10495_2021_1692_MOESM3_ESM.tif
Supplementary file3 Fig S3 The protein levels of p53, p21, p27, and E2F1 in HEK293T treated with Cu(II). The protein levels of p53, phospho-p53 (Ser46), phopsho-p53(Ser15), p21, p27, and E2F1 detected by Western blot from three independent experiments were shown (TIF 7584 kb)
10495_2021_1692_MOESM4_ESM.tif
Supplementary file4 Fig S4. KEGG pathway enrichment of 3 ppm Cu(II) DEGs. A graphical representation of the 20 most represented KEGG pathways in DEGs of cells treated with 3 ppm Cu(II). Gene ratio is the ratio of DEGs counts to this pathway in the annotated genes counts. Q-value is the adjusted p-value after multiple hypothesis testing, and its range is [0, 1]. When the Q-value is close to zero, this means the enrichment is more significant (TIF 6951 kb)
Rights and permissions
About this article
Cite this article
Chen, CH., Chou, YT., Yang, YW. et al. High-dose copper activates p53-independent apoptosis through the induction of nucleolar stress in human cell lines. Apoptosis 26, 612–627 (2021). https://doi.org/10.1007/s10495-021-01692-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-021-01692-y