Abstract
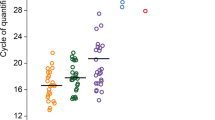
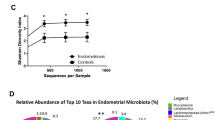
This study aimed to characterise the microbial community within the endometrial cavity and endocervix in women with menorrhagia or dysmenorrhea. Paired endocervical and endometrial biopsy samples were collected from women undergoing operative hysteroscopy and/or laparoscopy. Samples were cohorted based on pathology, indications for surgery, and histological dating of the endometrium. Samples were interrogated for the presence of microbial DNA using a two-step next generation sequencing technology approach to exploit the V5–V8 regions of the 16S rRNA gene. Pyrosequencing revealed that the endocervix and endometrium share a minor microbial community, but that each site harbours a separate and distinct microbial population (p = 0.024). This was also the case for women with menorrhagia and dysmenorrhea (p = 0.017). Lactobacillus spp. were the most abundant microbial taxa present in 50% of the cohorts, and across all endocervical groups. Members of the genera Prevotella, Fusobacterium and Jonquetella were the most abundant taxa identified in samples collected from nulliparous women. It can be concluded that the female upper genital tract is not sterile. Microbial community profiling revealed differences in the endometrial microbial community profiles for: (1) the endocervix compared to the endometrium, and (2), women with menorrhagia versus dysmenorrhea. The distinct microbial community profiles in these women may offer insight into the pathology and clinical management of dysfunctional menstrual bleeding.





Similar content being viewed by others
References
Andrews WW, Goldenberg RL, Hauth JC, Cliver SP, Conner M, Goepfert AR (2005) Endometrial microbial colonization and plasma cell endometritis after spontaneous or indicated preterm versus term delivery. Am J Obstet Gynecol 193(3 Pt 1):739–745
Ansbacher R, Boyson WA, Morris JA (1967) Sterility of the uterine cavity. Am J Obstet Gynecol 99(3):394–396
Aslim B, Kilic E (2006) Some probiotic properties of vaginal lactobacilli isolated from healthy women. Jpn J Infect Dis 59(4):249–253
Bollinger CC (1964) Bacterial flora of the nonpregnant uterus: a new culture technic. Obstet Gynecol 23:251–255
Bragg L, Stone G, Imelfort M, Hugenholtz P, Tyson GW (2012) Fast, accurate error-correction of amplicon pyrosequences using Acacia. Nat Methods 9(5):425–426
Calman RM, Gibson J (1954) The bacteriology of the puerperal uterus. J Obstet Gynaecol Br Emp 61(5):623–627
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27(16):2194–2200
Eschenbach DA, Patton DL, Meier A, Thwin SS, Aura J, Stapleton A, Hooton TM (2000) Effects of oral contraceptive pill use on vaginal flora and vaginal epithelium. Contraception 62(3):107–112
Fettweis JM, Serrano MG, Girerd PH, Jefferson KK, Buck GA (2012) A new era of the vaginal microbiome: advances using next-generation sequencing. Chem Biodivers 9(5):965–976
Friberg J, Confino E, Suarez M, Gleicher N (1987) Chlamydia trachomatis attached to spermatozoa recovered from the peritoneal cavity of patients with salpingitis. J Reprod Med 32(2):120–122
Gomez CI, Stenback WA, James AN, Criswell BS, Williams RP (1979) Attachment of Neisseria gonorrhoeae to human sperm. Microscopical study of trypsin and iron. Br J Vener Dis 55(4):245–255
Greer FR (2010) Vitamin K the basics—what’s new? Early Hum Dev 86(1, Supplement):43–47
Hill MJ (1997) Intestinal flora and endogenous vitamin synthesis. Eur J Cancer Prev 6(Suppl 1):S43–S45
Hillier S, Witkin S, Krohn M, Watts D, Kiviat N, Eschenbach D (1993) The relationship of amniotic fluid cytokines and preterm delivery, amniotic fluid infection, histologic chorioamnionitis, and chorioamnion infection. Obstet Gynecol 81(6):941–948
Hite KE, Hesseltine HC, Goldstein L (1947) A study of the bacterial flora of the normal and pathologic vagina and uterus. Am J Obstet Gynecol 53(2):233–240
Holmes KK, Sparling PF, Mardh P, Lemon ST, Stamm SE, Piot P, Wasserheit JN (eds) (2008) Sexually transmitted diseases. McGraw-Hill, New York
Holt JG, Krieg NR, Sneath PHA, Staley JT, Williams ST (eds) (2010) Bergey’s manual of systematic bacteriology. Williams and Wilkins, Baltimore
Jacobsson B, Pernevi P, Chidekel L, Jorgen Platz-Christensen J (2002) Bacterial vaginosis in early pregnancy may predispose for preterm birth and postpartum endometritis. Acta Obstet Gynecol Scand 81(11):1006–1010
Keith LG, Berger GS, Edelman DA, Newton W, Fullan N, Bailey R, Friberg J (1984) On the causation of pelvic inflammatory disease. Am J Obstet Gynecol 149(2):215–224
Korn AP, Bolan G, Padian N, Ohm-Smith M, Schachter J, Landers DV (1995) Plasma cell endometritis in women with symptomatic bacterial vaginosis. Obstet Gynecol 85(3):387–390
Kuczynski J, Stombaugh J, Walters WA, Gonzalez A, Caporaso JG, Knight R (2012) Using QIIME to analyze 16S rRNA gene sequences from microbial communities. Curr Protoc Microbiol Chapter 1: Unit 1E 5
Kunin V, Engelbrektson A, Ochman H, Hugenholtz P (2010) Wrinkles in the rare biosphere: pyrosequencing errors can lead to artificial inflation of diversity estimates. Environ Microbiol 12(1):118–123
Levi M, Keller TT, van Gorp E, ten Cate H (2003) Infection and inflammation and the coagulation system. Cardiovasc Res 60(1):26–39
Levi M, van der Poll T, Schultz M (2012) New insights into pathways that determine the link between infection and thrombosis. Neth J Med 70(3):114–120
Liu B, Gibbons T, Ghodsi M, Treangen T, Pop M (2011) Accurate and fast estimation of taxonomic profiles from metagenomic shotgun sequences. BMC Genomics 12(Suppl 2):S4
Lusher JM (1999) Systemic causes of excessive uterine bleeding. Semin Hematol 36(3 Suppl 4):10–20
McDonald D, Price MN, Goodrich J, Nawrocki EP, DeSantis TZ, Probst A, Andersen GL, Knight R, Hugenholtz P (2012) An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J 6(3):610–618
Moller BR, Kristiansen FV, Thorsen P, Frost L, Mogensen SC (1995) Sterility of the uterine cavity. Acta Obstet Gynecol Scand 74(3):216–219
Pelzer ES, Allan JA (2012) The isolation and identification of microorganisms in the reproductive environment: the potential impact on the IVF culture system and on IVF outcomes. J Clin Embryol 15(3):44–53
Pelzer ES, Allan JA, Waterhouse MA, Ross T, Beagley KW, Knox CL (2013) Microorganisms within human follicular fluid: effects on IVF. PLoS ONE 8(3):e59062
Ravel J, Gajer P, Abdo Z, Schneider GM, Koenig SS, McCulle SL, Karlebach S, Gorle R, Russell J, Tacket CO, Brotman RM, Davis CC, Ault K, Peralta L, Forney LJ (2011) Vaginal microbiome of reproductive-age women. Proc Natl Acad Sci USA 108(Suppl 1):4680–4687
Romero R, Gomez R, Chaiworapongsa T, Conoscenti G, Kim JC, Kim YM (2001) The role of infection in preterm labour and delivery. Paediatr Perinat Epidemiol 15(Suppl 2):41–56
Romero R, Espinoza J, Mazor M (2004) Can endometrial infection/inflammation explain implantation failure, spontaneous abortion, and preterm birth after in vitro fertilization? Fertil Steril 82(4):799–804
Santiago GL, Tency I, Verstraelen H, Verhelst R, Trog M, Temmerman M, Vancoillie L, Decat E, Cools P, Vaneechoutte M (2012) Longitudinal qPCR study of the dynamics of L. crispatus, L. iners, A. vaginae, (sialidase positive) G. vaginalis, and P. bivia in the vagina. PLoS ONE 7(9):e45281
Sharpe K, Karovitch A, Claman P, Suh K (2006) Transvaginal oocyte retrieval for in virto fertilization complicated by ovarian abscess during pregnancy. Fertil Steril 86(1):11–13
Sonnex C (1998) Influence of ovarian hormones on urogenital infection. Sex Transm Infect 74(1):11–19
Spence MR, Blanco LJ, Patel J, Brockman MT (1982) A comparative evaluation of vaginal, cervical and peritoneal flora in normal, healthy women: a preliminary report. Sex Transm Dis 9(1):37–40
Speroff L, Fritz MA (2005) Clinical gynecologic endocrinology and infertility. Lippincott Williams & Wilkins, Philadelphia
Svenstrup HF, Fedder J, Abraham-Peskir J, Birkelund S, Christiansen G (2003) Mycoplasma genitalium attaches to human spermatozoa. Hum Reprod 18(10):2103–2109
Swidsinski A, Verstraelen H, Loening-Baucke V, Swidsinski S, Mendling W, Halwani Z (2013) Presence of a polymicrobial endometrial biofilm in patients with bacterial vaginosis. PLoS ONE 8(1):e53997
Verstraelen H, Verhelst R, Claeys G, De Backer E, Temmerman M, Vaneechoutte M (2009) Longitudinal analysis of the vaginal microflora in pregnancy suggests that L. crispatus promotes the stability of the normal vaginal microflora and that L. gasseri and/or L. iners are more conducive to the occurrence of abnormal vaginal microflora. BMC Microbiol 9:116
White JR, Nagarajan N, Pop M (2009) Statistical methods for detecting differentially abundant features in clinical metagenomic samples. PLoS Comput Biol 5(4):e1000352
Willner D, Haynes MR, Furlan M, Schmieder R, Lim YW, Rainey PB, Rohwer F, Conrad D (2012) Spatial distribution of microbial communities in the cystic fibrosis lung. ISME J 6(2):471–474
Wolner-Hanssen P, Mardh PA (1984) In vitro tests of the adherence of Chlamydia trachomatis to human spermatozoa. Fertil Steril 42(1):102–107
Acknowledgements
The authors wish to thank Wesley Hospital theatre staff who facilitated collection of the genital tract samples. We wish to acknowledge the Australian Centre for Ecogenomics for 454 pyrosequencing and Professor Philip Hugenholtz and Dr Fiona May. This work was performed in the Thorsen Group Women’s Health Laboratory.
Funding
This work was performed in the Thorsen Group Women’s Health Laboratory and was supported by a Wesley Research Institute Grant (2011-18).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No competing financial interests exist.
Ethical approval
All procedures performed in studies involving human participant were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Pelzer, E.S., Willner, D., Buttini, M. et al. A role for the endometrial microbiome in dysfunctional menstrual bleeding. Antonie van Leeuwenhoek 111, 933–943 (2018). https://doi.org/10.1007/s10482-017-0992-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-017-0992-6