Abstract
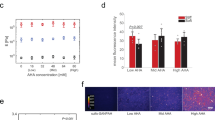
Human stem cells hold significant potential for the treatment of various diseases. However, their use as a therapy is hampered because of limited understanding of the mechanisms by which they respond to environmental stimuli. Efforts to understand extracellular biophysical cues have demonstrated the critical roles of geometrical and mechanical signals in determining the fate of stem cells. The goal of this study was to explore the interplay between cell polarity and matrix stiffness in stem cell lineage specification. We hypothesize that confining cells to asymmetric extracellular matrix islands will impart polarity at a single-cell level and will interact with mechanical signals to define the lineage of stem cells. To test these hypotheses, we employed microcontact printing to create patterned symmetric and asymmetric hydrogel islands of soft and hard surface stiffness. Human mesenchymal stem cells (hMSCs) were confined to these islands at the single-cell level and given the ability to differentiate along adipogenic or osteogenic routes. Our results demonstrated that cell polarity defines the lineage specification of hMSCs only on islands with low stiffness. Insight gained from this study provides a rational basis for designing stem cell cultures to enhance tissue engineering and regenerative medicine strategies.





Similar content being viewed by others
References
Ben-Yair, R., N. Kahane, and C. Kalcheim. Lgn-dependent orientation of cell divisions in the dermomyotome controls lineage segregation into muscle and dermis. Development 138:4155–4166, 2011.
Chang, Y. C., P. Nalbant, J. Birkenfeld, Z. F. Chang, and G. M. Bokoch. Gef-h1 couples nocodazole-induced microtubule disassembly to cell contractility via rhoa. Mol. Biol. Cell. 19:2147–2153, 2008.
Chen, C. S., M. Mrksich, S. Huang, G. M. Whitesides, and D. E. Ingber. Geometric control of cell life and death. Science. 276:1425–1428, 1997.
Chen, C. S., M. Mrksich, S. Huang, G. M. Whitesides, and D. E. Ingber. Micropatterned surfaces for control of cell shape, position, and function. Biotechnol. Prog. 14:356–363, 1998.
Drubin, D. G., and W. J. Nelson. Origins of cell polarity. Cell. 84:335–344, 1996.
Engler, A. J., S. Sen, H. L. Sweeney, and D. E. Discher. Matrix elasticity directs stem cell lineage specification. Cell. 126:677–689, 2006.
Guilak, F., D. M. Cohen, B. T. Estes, J. M. Gimble, W. Liedtke, and C. S. Chen. Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell. 5:17–26, 2009.
Harris, G. M., M. E. Piroli, and E. Jabbarzadeh. Deconstructing the effects of matrix elasticity and geometry in mesenchymal stem cell lineage commitment. Adv. Funct. Mater. 24:2396–2403, 2014.
Harris, G. M., T. Shazly, and E. Jabbarzadeh. Deciphering the combinatorial roles of geometric, mechanical, and adhesion cues in regulation of cell spreading. PLoS ONE. 8:e81113, 2013.
Horvitz, H. R., and I. Herskowitz. Mechanisms of asymmetric cell division: two bs or not two bs, that is the question. Cell. 68:237–255, 1992.
Ishizaki, T., M. Uehata, I. Tamechika, J. Keel, K. Nonomura, M. Maekawa, and S. Narumiya. Pharmacological properties of y-27632, a specific inhibitor of rho-associated kinases. Mol. Pharmacol. 57:976–983, 2000.
James, A. W. Review of signaling pathways governing msc osteogenic and adipogenic differentiation. Scientifica (Cairo). 684736:2013, 2013.
Kamakura, S., M. Nomura, J. Hayase, Y. Iwakiri, A. Nishikimi, R. Takayanagi, Y. Fukui, and H. Sumimoto. The cell polarity protein minsc regulates neutrophil chemotaxis via a noncanonical g protein signaling pathway. Dev. Cell. 26:292–302, 2013.
Kaushik, R., F. Yu, W. Chia, X. Yang, and S. Bahri. Subcellular localization of lgn during mitosis: evidence for its cortical localization in mitotic cell culture systems and its requirement for normal cell cycle progression. Mol. Biol. Cell. 14:3144–3155, 2003.
Kilian, K. A., B. Bugarija, B. T. Lahn, and M. Mrksich. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc. Natl. Acad. Sci. USA. 107:4872–4877, 2010.
Kilian, K. A., and M. Mrksich. Directing stem cell fate by controlling the affinity and density of ligand-receptor interactions at the biomaterials interface. Angew Chem Int Ed Engl. 51:4891–4895, 2012.
Kimura, K., M. Ito, M. Amano, K. Chihara, Y. Fukata, M. Nakafuku, B. Yamamori, J. Feng, T. Nakano, K. Okawa, A. Iwamatsu, and K. Kaibuchi. Regulation of myosin phosphatase by rho and rho-associated kinase (rho-kinase). Science. 273:245–248, 1996.
Knoblich, J. A. Mechanisms of asymmetric stem cell division. Cell. 132:583–597, 2008.
Knoblich, J. A. Asymmetric cell division: recent developments and their implications for tumour biology. Nat. Rev. Mol. Cell Biol. 11:849–860, 2010.
Konno, D., G. Shioi, A. Shitamukai, A. Mori, H. Kiyonari, T. Miyata, and F. Matsuzaki. Neuroepithelial progenitors undergo lgn-dependent planar divisions to maintain self-renewability during mammalian neurogenesis. Nat. Cell Biol. 10:93–101, 2008.
Krtolica, A., O. Genbacev, C. Escobedo, T. Zdravkovic, A. Nordstrom, D. Vabuena, A. Nath, C. Simon, K. Mostov, and S. J. Fisher. Disruption of apical-basal polarity of human embryonic stem cells enhances hematoendothelial differentiation. Stem Cells. 25:2215–2223, 2007.
Lechler, T., and E. Fuchs. Asymmetric cell divisions promote stratification and differentiation of mammalian skin. Nature. 437:275–280, 2005.
Lee, J., A. A. Abdeen, T. H. Huang, and K. A. Kilian. Controlling cell geometry on substrates of variable stiffness can tune the degree of osteogenesis in human mesenchymal stem cells. J. Mech. Behav. Biomed. Mater. 38:209–218, 2014.
Lee, J., A. A. Abdeen, and K. A. Kilian. Rewiring mesenchymal stem cell lineage specification by switching the biophysical microenvironment. Sci. Rep. 4:5188, 2014.
Lee, M., and V. Vasioukhin. Cell polarity and cancer–cell and tissue polarity as a non-canonical tumor suppressor. J. Cell Sci. 121:1141–1150, 2008.
Ling, L., V. Nurcombe, and S. M. Cool. Wnt signaling controls the fate of mesenchymal stem cells. Gene. 433:1–7, 2009.
McBeath, R., D. M. Pirone, C. M. Nelson, K. Bhadriraju, and C. S. Chen. Cell shape, cytoskeletal tension, and rhoa regulate stem cell lineage commitment. Dev. Cell. 6:483–495, 2004.
Mooney, D., L. Hansen, J. Vacanti, R. Langer, S. Farmer, and D. Ingber. Switching from differentiation to growth in hepatocytes: control by extracellular matrix. J. Cell. Physiol. 151:497–505, 1992.
Morrison, S. J., and J. Kimble. Asymmetric and symmetric stem-cell divisions in development and cancer. Nature. 441:1068–1074, 2006.
Mukherjee, S., J. Kong, and D. J. Brat. Cancer stem cell division: when the rules of asymmetry are broken. Stem Cells Dev. 24:405–416, 2015.
Nelson, C. M., and C. S. Chen. Cell-cell signaling by direct contact increases cell proliferation via a pi3 k-dependent signal. FEBS Lett. 514:238–242, 2002.
Peng, R., X. Yao, B. Cao, J. Tang, and J. Ding. The effect of culture conditions on the adipogenic and osteogenic inductions of mesenchymal stem cells on micropatterned surfaces. Biomaterials. 33:6008–6019, 2012.
Pham, K., F. Sacirbegovic, and S. M. Russell. Polarized cells, polarized views: asymmetric cell division in hematopoietic cells. Front. Immunol. 5:26, 2014.
Rowlands, A. S., P. A. George, and J. J. Cooper-White. Directing osteogenic and myogenic differentiation of mscs: interplay of stiffness and adhesive ligand presentation. Am. J. Physiol. Cell Physiol. 295:C1037–C1044, 2008.
Simons, M., and M. Mlodzik. Planar cell polarity signaling: from fly development to human disease. Annu Rev. Genet. 42:517, 2008.
Thery, M. Micropatterning as a tool to decipher cell morphogenesis and functions. J. Cell Sci. 123:4201–4213, 2010.
Thery, M., and M. Bornens. Cell shape and cell division. Curr. Opin. Cell Biol. 18:648–657, 2006.
Thery, M., A. Jimenez-Dalmaroni, V. Racine, M. Bornens, and F. Julicher. Experimental and theoretical study of mitotic spindle orientation. Nature. 447:493–496, 2007.
Thery, M., V. Racine, M. Piel, A. Pepin, A. Dimitrov, Y. Chen, J. B. Sibarita, and M. Bornens. Anisotropy of cell adhesive microenvironment governs cell internal organization and orientation of polarity. Proc. Natl. Acad. Sci. USA. 103:19771–19776, 2006.
Vasquez, R. J., B. Howell, A. M. Yvon, P. Wadsworth, and L. Cassimeris. Nanomolar concentrations of nocodazole alter microtubule dynamic instability in vivo and in vitro. Mol. Biol. Cell. 8:973–985, 1997.
Walsh, T., H. Shahin, T. Elkan-Miller, M. K. Lee, A. M. Thornton, W. Roeb, A. Abu, S. Rayyan, K. B. Loulus, M.-C. Avraham, and M. Kanaan. Whole exome sequencing and homozygosity mapping identify mutation in the cell polarity protein gpsm2 as the cause of nonsyndromic hearing loss dfnb82. Am. J. Hum. Genet. 87:90–94, 2010.
Winer, J. P., P. A. Janmey, M. E. McCormick, and M. Funaki. Bone marrow-derived human mesenchymal stem cells become quiescent on soft substrates but remain responsive to chemical or mechanical stimuli. Tissue Eng Part A. 15:147–154, 2009.
Wodarz, A., and I. Näthke. Cell polarity in development and cancer. Nat. Cell Biol. 9:1016, 2007.
Wozniak, M. A., and C. S. Chen. Mechanotransduction in development: a growing role for contractility. Nat. Rev. Mol. Cell Biol. 10:34–43, 2009.
Wright, C. E., E. J. Kushner, Q. Du, and V. L. Bautch. Lgn directs interphase endothelial cell behavior via the microtubule network. PLoS One. 10:e0138763, 2015.
Yamashita, Y. M., H. Yuan, J. Cheng, and A. J. Hunt. Polarity in stem cell division: asymmetric stem cell division in tissue homeostasis. Cold Spring Harb Perspect Biol. 2:a001313, 2010.
Yang, S., K. Ma, Z. Geng, X. Sun, and X. Fu. Oriented cell division: new roles in guiding skin wound repair and regeneration. Biosci Rep. 35:e00280, 2015.
Yeaman, C., K. K. Grindstaff, and W. J. Nelson. New perspectives on mechanisms involved in generating epithelial cell polarity. Physiol. Rev. 79:73–98, 1999.
Zhang, D., and K. A. Kilian. The effect of mesenchymal stem cell shape on the maintenance of multipotency. Biomaterials. 34:3962–3969, 2013.
Zhong, Y., and B. Ji. Impact of cell shape on cell migration behavior on elastic substrate. Biofabrication. 5:015011, 2013.
Acknowledgments
Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number AR063338. We would like to thank Dr. Tarek Shazly and Will Torres for assisting us with the use of the Electroforce 3200.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Arash Kheradvar oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Piroli, M.E., Jabbarzadeh, E. Matrix Stiffness Modulates Mesenchymal Stem Cell Sensitivity to Geometric Asymmetry Signals. Ann Biomed Eng 46, 888–898 (2018). https://doi.org/10.1007/s10439-018-2008-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-018-2008-8