Abstract
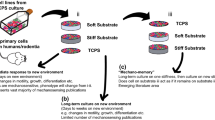
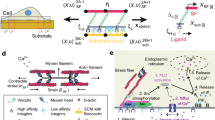
Understanding the role of mechanophenotype in competitive adherence of cells to other cells versus underlying substrates can inform such processes as tissue development, cancer progression, and wound healing. This study investigated how mechanophenotype, defined by whole-cell, elastic/viscoelastic properties for the perinuclear region, and cellular assembly are intertwined through the mechanosensing process. Atomic force microscopy was used to characterize the temporal elastic/viscoelastic properties of individual and assembled fibroblasts grown on substrates with elastic moduli above, below, or similar to whole-cell mechanophenotypes measured for three, genetically modified cell lines. All cells were at their most compliant immediately after plating but transitioned to distinct, stiffer mechanophenotypes by Day 1 after acclimation. This mechanical state, and cellular assembly/morphology, did not change significantly over the following three days of testing, regardless of substrate compliance or cellular organization (multi-cell nodules/plaques or single cells). Interestingly, cells formed 3D nodules when attached to substrates with elastic moduli less than their own but spread readily on substrates with moduli equal to or greater than their own, suggesting a preference to adhere to the stiffest surface sensed (substrate or cell). This suggests that inherent mechanophenotype plays a role as a competing surface during microenvironment mechanosensing and subsequent cell–cell-substrate organization.





Similar content being viewed by others
Abbreviations
- AFM:
-
Atomic force microscopy
- COL-I:
-
Collagen type-1
- dnRhoA:
-
dnRhoA (T19 N) - GFP transfected WI-38 cells
- ECM:
-
Extracellular matrix
- E elastic :
-
Elastic modulus
- E 0 :
-
Instantaneous modulus
- E R :
-
Relaxed modulus
- FN:
-
Fibronectin
- LN:
-
Laminin
- PAAm:
-
Polyacrylamide
- WI-38:
-
WI-38 VA-13 subline 2RA
- β-Actin:
-
β-Actin-GFP transfected WI-38 cells
- µ :
-
Apparent viscosity
References
Aizawa, H., M. Sameshima, and I. Yahara. A green fluorescent protein-actin fusion protein dominantly inhibits cytokinesis, cell spreading, and locomotion in Dictyostelium. Cell Struct. Funct. 22(3):335–345, 1997.
Amin, E., B. N. Dubey, S. C. Zhang, L. Gremer, R. Dvorsky, J. M. Moll, M. S. Taha, L. Nagel-Steger, R. P. Piekorz, A. V. Somlyo, and M. R. Ahmadian. Rho-kinase: regulation, (dys)function, and inhibition. Biol. Chem. 394(11):1399–1410, 2013.
Brafman, D. A. Constructing stem cell microenvironments using bioengineering approaches. Physiol. Genomics 45(23):1123–1135, 2013.
Brieher, W. M., and A. S. Yap. Cadherin junctions and their cytoskeleton(s). Curr. Opin. Cell Biol. 25(1):39–46, 2013.
Choidas, A., A. Jungbluth, A. Sechi, J. Murphy, A. Ullrich, and G. Marriott. The suitability and application of a GFP-actin fusion protein for long-term imaging of the organization and dynamics of the cytoskeleton in mammalian cells. Eur. J. Cell Biol. 77(2):81–90, 1998.
Darling, E. M., and D. Di Carlo. High-throughput assessment of cellular mechanical properties. Annu. Rev. Biomed. Eng. 17:35–62, 2015.
Darling, E. M., M. Topel, S. Zauscher, T. P. Vail, and F. Guilak. Viscoelastic properties of human mesenchymally-derived stem cells and primary osteoblasts, chondrocytes, and adipocytes. J. Biomech. 41(2):454–464, 2008.
Darling, E. M., S. Zauscher, J. A. Block, and F. Guilak. A thin-layer model for viscoelastic, stress-relaxation testing of cells using atomic force microscopy: do cell properties reflect metastatic potential? Biophys. J . 92(5):1784–1791, 2007.
Di Carlo, D. A mechanical biomarker of cell state in medicine. Jala 17(1):32–42, 2012.
Engler, A., L. Bacakova, C. Newman, A. Hategan, M. Griffin, and D. Discher. Substrate compliance versus ligand density in cell on gel responses. Biophys. J . 86(1 Pt 1):617–628, 2004.
Engler, A. J., S. Sen, H. L. Sweeney, and D. E. Discher. Matrix elasticity directs stem cell lineage specification. Cell 126(4):677–689, 2006.
Georges, P. C., and P. A. Janmey. Cell type-specific response to growth on soft materials. J. Appl. Physiol. (1985) 98(4):1547–1553, 2005.
Ghosh, P. M., N. Ghosh-Choudhury, M. L. Moyer, G. E. Mott, C. A. Thomas, B. A. Foster, N. M. Greenberg, and J. I. Kreisberg. Role of RhoA activation in the growth and morphology of a murine prostate tumor cell line. Oncogene 18(28):4120–4130, 1999.
Gilchrist, C. L., E. M. Darling, J. Chen, and L. A. Setton. Extracellular matrix ligand and stiffness modulate immature nucleus pulposus cell-cell interactions. PLoS ONE 6(11):e27170, 2011.
Gonzalez-Cruz, R. D., V. C. Fonseca, and E. M. Darling. Cellular mechanical properties reflect the differentiation potential of adipose-derived mesenchymal stem cells. Proc. Natl. Acad. Sci. USA 109(24):E1523–E1529, 2012.
Guo, W. H., M. T. Frey, N. A. Burnham, and Y. L. Wang. Substrate rigidity regulates the formation and maintenance of tissues. Biophys. J . 90(6):2213–2220, 2006.
Hosein, R. E., S. A. Williams, K. Haye, and R. H. Gavin. Expression of GFP-actin leads to failure of nuclear elongation and cytokinesis in Tetrahymena thermophila. J. Eukaryot. Microbiol. 50(6):403–408, 2003.
Jagielska, A., A. L. Norman, G. Whyte, K. J. Vliet, J. Guck, and R. J. Franklin. Mechanical environment modulates biological properties of oligodendrocyte progenitor cells. Stem Cells Dev 21(16):2905–2914, 2012.
Kular, J. K., S. Basu, and R. I. Sharma. The extracellular matrix: structure, composition, age-related differences, tools for analysis and applications for tissue engineering. J Tissue Eng 5:2041731414557112, 2014.
Labriola, N. R., and E. M. Darling. Temporal heterogeneity in single-cell gene expression and mechanical properties during adipogenic differentiation. J. Biomech. 48(6):1058–1066, 2015.
Lo, C. M., H. B. Wang, M. Dembo, and Y. L. Wang. Cell movement is guided by the rigidity of the substrate. Biophys. J . 79(1):144–152, 2000.
McBeath, R., D. M. Pirone, C. M. Nelson, K. Bhadriraju, and C. S. Chen. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev. Cell 6(4):483–495, 2004.
Mendez, M. G., D. Restle, and P. A. Janmey. Vimentin enhances cell elastic behavior and protects against compressive stress. Biophys. J . 107(2):314–323, 2014.
Parsons, J. T., A. R. Horwitz, and M. A. Schwartz. Cell adhesion: integrating cytoskeletal dynamics and cellular tension. Nat. Rev. Mol. Cell Biol. 11(9):633–643, 2010.
Poh, Y. C., F. Chowdhury, T. S. Tanaka, and N. Wang. Embryonic stem cells do not stiffen on rigid substrates. Biophys. J . 99(2):L19–L21, 2010.
Ross, R. A., B. A. Spengler, and J. L. Biedler. Coordinate morphological and biochemical interconversion of human neuro-blastoma cells. J. Natl Cancer Inst. 71(4):741–749, 1983.
Sadick, J. S., M. E. Boutin, D. Hoffman-Kim, and E. M. Darling. Protein characterization of intracellular target-sorted, formalin-fixed cell subpopulations. Sci Rep 6:33999, 2016.
Schwarz, U. S., and M. L. Gardel. United we stand: integrating the actin cytoskeleton and cell-matrix adhesions in cellular mechanotransduction. J. Cell Sci. 125(Pt 13):3051–3060, 2012.
Sen, S., M. Dong, and S. Kumar. Isoform-specific contributions of alpha-actinin to glioma cell mechanobiology. PLoS ONE 4(12):e8427, 2009.
Sen, S., W. P. Ng, and S. Kumar. Contributions of talin-1 to glioma cell-matrix tensional homeostasis. J. R. Soc. Interface 9(71):1311–1317, 2012.
Solon, J., I. Levental, K. Sengupta, P. C. Georges, and P. A. Janmey. Fibroblast adaptation and stiffness matching to soft elastic substrates. Biophys. J . 93(12):4453–4461, 2007.
Stroka, K. M., and H. Aranda-Espinoza. Effects of morphology vs. cell-cell interactions on endothelial cell stiffness. Cell. Mol. Bioeng. 4(1):9–27, 2011.
Suresh, S. Biomechanics and biophysics of cancer cells. Acta Biomater. 3(4):413–438, 2007.
Tse, J. R., and A. J. Engler. Preparation of hydrogel substrates with tunable mechanical properties. Curr Protoc Cell Biol 10:10–16, 2010.
Wang, J. H., and J. S. Lin. Cell traction force and measurement methods. Biomech. Model. Mechanobiol. 6(6):361–371, 2007.
Weber, G. F., M. A. Bjerke, and D. W. DeSimone. Integrins and cadherins join forces to form adhesive networks. J. Cell Sci. 124(Pt 8):1183–1193, 2011.
Yeung, T., P. C. Georges, L. A. Flanagan, B. Marg, M. Ortiz, M. Funaki, N. Zahir, W. Ming, V. Weaver, and P. A. Janmey. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil Cytoskeleton 60(1):24–34, 2005.
Acknowledgments
The authors would like to thank Louise E. O. Darling for the GFP plasmids and Jessica S. Sadick and Vera Fonseca for help with western blots. This work was supported by awards from the National Institute of Health (R01 AR063642, P20 GM104937) and the National Science Foundation (CAREER CBET 1253189). The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the National Science Foundation or National Institutes of Health.
Conflict of Interest
Manisha K. Shah, Iris H. Garcia-Pak, and Eric M. Darling declare that they have no conflicts of interest.
Data Accessibility
AFM and nodule quantification data can be downloaded from 10.6084/m9.figshare.4341887.
Author information
Authors and Affiliations
Contributions
Author contributions
M.K.S. and E.M.D. designed the study, analyzed data, and wrote the manuscript. M.K.S. performed all AFM experiments and all experiments with non-transfected and transfected WI-38 cells lines. I.H.G.-P performed MG-63 and SH-SY5Y cellular assembly experiments. All authors gave final approval for publication.
Corresponding author
Additional information
Associate Editor Michael S. Detamore oversaw the review of this article.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shah, M.K., Garcia-Pak, I.H. & Darling, E.M. Influence of Inherent Mechanophenotype on Competitive Cellular Adherence. Ann Biomed Eng 45, 2036–2047 (2017). https://doi.org/10.1007/s10439-017-1841-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-017-1841-5