Abstract
Past failures of monocultures, caused by wind-throw or insect damages, and ongoing climate change currently strongly stimulate research into mixed-species stands. So far, the focus has mainly been on combinations of species with obvious complementary functional traits. However, for any generalization, a broad overview of the mixing reactions of functionally different tree species in different mixing proportions, patterns and under different site conditions is needed, including assemblages of species with rather similar demands on resources such as light. Here, we studied the growth of Scots pine and oak in mixed versus monospecific stands on 36 triplets located along a productivity gradient across Europe, reaching from Sweden to Spain and from France to Georgia. The set-up represents a wide variation in precipitation (456–1250 mm year−1), mean annual temperature (6.7–11.5 °C) and drought index by de Martonne (21–63 mm °C−1). Stand inventories and increment cores of trees stemming from 40- to 132-year-old, fully stocked stands on 0.04–0.94-ha-sized plots provided insight into how species mixing modifies stand growth and structure compared with neighbouring monospecific stands. On average, the standing stem volume was 436 and 360 m3 ha−1 in the monocultures of Scots pine and oak, respectively, and 418 m3 ha−1 in the mixed stands. The corresponding periodical annual volume increment amounted to 10.5 and 9.1 m3 ha−1 year−1 in the monocultures and 10.5 m3 ha−1 year−1 in the mixed stands. Scots pine showed a 10% larger quadratic mean diameter (p < 0.05), a 7% larger dominant diameter (p < 0.01) and a 9% higher growth of basal area and volume in mixed stands compared with neighbouring monocultures. For Scots pine, the productivity advantages of growing in mixture increased with site index (p < 0.01) and water supply (p < 0.01), while for oak they decreased with site index (p < 0.01). In total, the superior productivity of mixed stands compared to monocultures increased with water supply (p < 0.10). Based on 7843 measured crowns, we found that in mixture both species, but especially oak, had significantly wider crowns (p < 0.001) than in monocultures. On average, we found relatively small effects of species mixing on stand growth and structure. Scots pine benefiting on rich, and oak on poor sites, allows for a mixture that is productive and most likely climate resistant all along a wide ecological gradient. We discuss the potential of this mixture in view of climate change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There are many reasons for systematically analysing as many different tree species mixtures as possible. Recent research has revealed that mixed-species stands can be more stable in view of biotic or abiotic disturbances (Bauhus et al. 2017; del Río et al. 2017; Jactel and Brockerhoff 2007), more resilient after damages (Metz et al. 2016; Pretzsch et al. 2013a, b) and more productive due to competition reduction or facilitation (Jactel et al. 2018; Liang et al. 2016; Pretzsch et al. 2017), and may provide a broader supply of ecological and socio-economical services (Biber et al. 2015; Felton et al. 2016; Gamfeldt et al. 2013; Griess and Knoke 2013; Heinrichs et al. 2019). Selected tree species mixtures such as Norway spruce (Picea abies (L.) H. Karst)/European beech (Fagus sylvatica L.) or Scots pine (Pinus sylvestris L.)/European beech and other mixtures including European beech are very well analysed (Knoke et al. 2008; Pretzsch and Schütze 2009); however, there is still a long way for forest science to establish a solid theory of mixing effects and general rules of species behaviour in mixture or even guidelines for combining and thinning tree species or functional groups of species (Forrester 2014). A basis for any generalization or theory building is a broad overview of the mixing reactions of functionally different tree species in different mixing proportions, patterns and under different site conditions.
Potential for synergy arises in particular when mixing species with complementary resource use and access above-ground (Ammer 2019; Forrester et al. 2018; Pretzsch 2014) or below-ground (Augusto et al. 2002; Caldwell et al. 1998; Rothe and Binkley 2001); to what extent this potential for synergy can be exploited depends in addition on the respective site conditions. Greater crown canopy packing in mixed stands due to species differences in crown morphology and light ecology has been identified as an important cause of overyielding (i.e. the mixed stand produces more than is expected from the monocultures) (Pretzsch and Schütze 2016; Williams et al. 2017). There are many studies about combinations of light demanding with shade-tolerant tree species which suggest overyielding due to increased light interception and possibly higher light use efficiency (Forrester et al. 2018; Jactel et al. 2018). When mixed species have similar traits, such as pine mixtures, the potential for synergy is lower and underyielding or neutral effects may be more common (Aguirre et al. 2019). However, small differences in functional traits can result in overyielding too (Riofrío et al. 2017). Combinations of species with more similar light ecology but complementary root space exploitation, such as Scots pine and oak, were analysed at selected sites, but so far not over a broader range of site conditions (Bello et al. 2019).
Here, we studied the growth of Scots pine (P. sylvestris L.) and oak, the latter comprising both sessile oak (Quercus petraea (Matt.) Liebl.) and pedunculate oak (Q. robur L.), in mixed versus monospecific stands on 36 triplets located along a productivity gradient across Europe. The taxonomic status of the two mentioned oak species has since long been subjected to ongoing discussions and repeated reassessment (Aas 1991). Sessile and pedunculate oak have either been described as two distinct species, Q. petraea (Matt.) Liebl. and Q. robur L., respectively, or are currently placed within the species Quercus robur L. as two subspecies Q. r. petraea and Q. r. robur (Roloff et al. 2008, pp. 506–507). To avoid possible taxonomic pitfalls, we either use “oak” as a generic term summarizing both species, or their colloquial names to distinguish species/subspecies with “sessile oak” to the petraea type and “pedunculate oak” referring to the robur type, respectively.
Scots pine and oak are economically very important tree species in Europe, valued for a wide range of end-uses, ranging from construction timber over furniture to pulp and paper in the case of Scots pine (Houston Durrant et al. 2016) and focusing on higher-end timber-frame building, furniture, flooring and veneer applications in the case of oak (Eaton et al. 2016). Under climate change, ecosystem disturbances such as severe droughts and wildfires are likely to increase in frequency and intensity (IPCC 2013). Against this background, Scots pine and oak are widely considered to be promising tree species that allow forest managers to reduce risks associated with climate change (Spellmann et al. 2011) and to provide an option to manage for multiple values. There are also many indications that this mixture has been quite natural and common in the past (Björse and Bradshaw 1998). Scots pine is well protected against drought, owing to its imbedded stomata and waxy layer on the epidermis (Krakau et al. 2013), although its sensitivity against increased temperatures is not fully clear yet. Scots pine can regulate its transpiration in an early stage of drought. Oak on the other hand is known to keep its stomata open longer during drought and utilize its deep-reaching taproots, thereby improving the water availability under drought (Praciak 2013). While Scots pine seems to perform better in spring droughts, oak showed higher resistance in years with longer summer drought events (Merlin et al. 2015; Vanhellemont et al. 2019). Scots pine is a tree species of the continental climate, well adapted to colder and nutrient-poor sites. Its climate envelope covers a temperature amplitude of roughly − 4 to 14 °C and a precipitation range of some 400 to 1300 mm (Kölling 2007). The distribution range of oak is limited to warmer sites with mean annual temperatures of 1 to 15 °C, with annual precipitation totals similar to Scots pine between 300 and 1300 mm (Kölling 2007). In mixture, both species currently cover an area of approximately 1.3 × 106 ha in Europe, with a potential distribution area of 35 × 106 ha (calculated based on Brus et al. 2012). It is likely that this species combination will increase in popularity. First, it will increase because both species are economically important and are assigned to have the potential for adaptive forest management and, second, because the two other tree species prevailing in Central Europe, Norway spruce and European beech seem to suffer already from the ongoing climate changes. However, in many European regions Scots pine and oak are disadvantaged as other light-demanding species since close-to-nature management schemes promote more shade-tolerant species such as beech (Pach et al. 2018) which can be considered the most competitive tree species in Central Europe (Leuschner et al. 2006). Silvicultural interventions to increase light availability are often required to ensure the continuance of oak in particular (Mölder et al. 2019). This is even more important in mixture with beech (von Lüpke and Hauskeller-Bullerjahn 2004).
Despite the significant potential of Scots pine and oak mixtures for adaptive forest management, they have so far only been studied regionally, revealing inconsistent results. The Gisburn experiment (Brown 1992) revealed positive mixing effects in terms of productivity for young Scots pine–oak stands in England. Lu et al. (2016, 2018) reported overyielding of Scots pine–oak mixtures on permanent field plots in the Netherlands; here, overyielding increased on poor soils which can be seen as in line with the stress gradient hypothesis (Bertness and Callaway 1994). According to a recent study by Steckel et al. (2019), utilizing a triplet transect spanning from Southern Germany to Eastern Denmark, mixing of Scots pine and oak resulted in a higher annual volume productivity than expected from monocultures. It amounted on average to 14% and increased with annual water supply. Similar results were observed for Iberian pine–oak mixtures (Jucker et al. 2014) where overyielding increased in wet years. Using inventory data in France, Toïgo et al. (2015) were able to confirm overyielding for oak, but found no significant overyielding on the stand level in Scots pine–oak mixtures. In those cases, where overyielding of Scots pine–oak mixtures was found, it has mainly been attributed to complementary light use, stemming from differences in shade tolerance (even though both species are classified as light demanding), leaf phenology and crown architecture (Steckel et al. 2019). Similarly, complementary resource use resulting from differences in depth of water uptake may also play a role for the observed mixing effects (Bello et al. 2019). However, the question of how Scots pine and oak interact along a wide range of different site conditions has not been sufficiently answered yet.
The objective of this study was therefore, to analyse the effect of mixing Scots pine with oak on tree and stand growth along a pedo-climatic gradient. Based on 36 triplets of mixed and monospecific stands of these species across Europe, we tried to answer the following questions:
-
1.
How do mixed stands differ from the monospecific stands in terms of mean height, mean stem diameter, volume stock, stand density and periodical annual growth of stand basal area and stem volume?
-
2.
How do mixing effects depend on the site characteristics?
-
3.
Does the crown allometry differ between trees in mixed and monospecific stands?
Materials and methods
Material
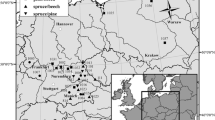
The consortium established a total of 36 triplets (Table 1, Fig. 1), reaching from Spain in the South to Sweden in the North and from France in the West to Georgia in the East. The triplets transect covers a wide variation in site conditions (Supplement Table 1), with precipitation ranging from 456 to 1250 mm year−1 and mean annual temperature from 6.7 to 11.5 °C. On some of the locations, two triplets were established in close proximity to each other in order to allow for future thinning experiments that are subject to a subsequent study. Triplets are sets of three plots: two monospecific stands of Scots pine and oak, and one mixed-species stand with both species.
Location of the 36 triplet groups of mixed and monospecific stands of Scots pine and oak established and sampled in 2017. The triplets are spread over 13 countries: Austria (AT_1), Belgium (BE_1), The Czech Republic (CZ_1-2), Denmark (DK_1), France (FR_1-2), Georgia (GE_1), Germany (DE_1-5), Latvia (LV_1), Lithuania (LT_1-2), Poland (PL_1-4), Slovakia (SK_1), Spain (ES_1-4) and Sweden (SE_1-2). Note that selected locations entail two triplets (AT_1, DE_1, DE_3, LT_1, PL_1, PL_2, PL_3, PL_4, ES_1 and ES_4) (see supplement Tables 1 and 2)
The plot size depended on the local conditions and ranged from 0.04 to 0.94 ha. The plots represent even-aged, fully stocked stands and have a mono-layered structure. We selected plots in stands that had not been thinned or at least not thinned within the last 10 years, so that they approximately represent the site-specific maximum stand density. Before the final selection and acceptance of the triplets, all project partners investigated the stand history as far backwards as possible in order to contribute to the study with unmanaged or, at most, slightly managed plots. In the mixed stands, the trees of the two species were arranged mainly in mixtures of individual trees or small groups. The mixing proportions were calculated by the weighted SDI (see “Data evaluation” section) and amounted to 19–85% for Scots pine and correspondingly 15–81% for oak.
Based on local site mappings, only plots of similar site conditions were chosen for the triplets. This is an important precondition since the monospecific plots were used as reference for the quantification of any over- or underyielding of mixed versus monospecific stands. On the plots, we measured dendrometric state variables at the tree as well as at the stand level and took increment cores.
In order to retrospectively calculate the tree and stand growth, we aimed to extract increment cores from at least 15 sample trees per species per plot. On average, 28 and 27 trees were cored in the mixed and monospecific stands, respectively (Supplement Table 2). The sample trees covered the whole diameter range of the corresponding species on the plots. The cores were extracted at 1.30 m stem height from the north and east directions; they reached back to the pith of the trees. For further evaluation, the annual ring widths on the cores were measured, the dendrometric time series cross-dated and synchronized, and arithmetic means of the annual ring widths from the cores in north and east directions calculated. For a more detailed description of the sampling and measuring procedure, see Heym et al. (2017, 2018) and Steckel et al. (2019).
Data preparation
According to the DESER-Norm 1993 (Johann 1993; Pretzsch 2009, pp. 181–222), we calculated the quadratic mean tree diameter, dominant diameter, height of the tree with the quadratic mean diameter and dominant tree height (dq, do, hq, ho), stand basal area (BA) and standing volume stock per hectare (V) for the current survey in 2017 and also for 2012. The evaluation for 2012, i.e. retrospectively for the last 5 years, is explained briefly in the next paragraph (see also Heym et al. (2017, 2018) for further details). Based on the reconstructed stand characteristics in 2012, the mean periodical stand basal area growth PAIBA and stem volume growth of the stand PAIV were calculated for 2012–2017 as PAIBA2012–2017 = BA2012 − BA2017 + BAremoval and PAIV2012–2017 = V2012 − V2017 + Vremoval. BAremoval and Vremoval represent the basal area and stem volume of the trees that had naturally dropped out in the period 2012–2017 and were calculated on the basis of stumps (by species-specific functions d1.3 = f(dstump) parameterized on the basis of sample trees in the stands) and standing dead trees inventoried on the plots. All statements on volume stock and volume increment refer to standing merchantable stem volume (> 7 cm at the smaller end).
For reconstruction of the stand characteristics in 2012, diameters and heights were reconstructed based on growth rate reads from increment cores. In order to reconstruct the diameter at breast height over bark in 2012, linear regression models (OLS regression) \(\ln ({\text{id}}_{2017 - 2012} ) = a_{0} + a_{1} \times \ln (d_{2017} )\) were fitted for each plot and species. In this equation, id2012–2017 represents the total stem diameter increment of the period 2012–2017, d2017 the stem diameter in 2017, and a0 and a1 the intercept and slope. By using this model, the tree diameter in 2012 of all non-cored trees was estimated (d2012 = d2017 − id2017–2012). For all cored trees, the diameter was estimated based on measured tree ring widths.
For reconstruction of the individual tree heights of Scots pine and oak back to 2012, we applied the system of uniform height curves developed for European beech by Kennel (1972, pp. 77–80) which was parameterized also for Scots pine and oak by Franz et al. (1973, pp. 91–99). By using the uniform height curves, all tree heights, h, were estimated in dependency of the available stem diameters, d, by the Petterson (1955) formula \(h = 1.3 + \left( {d/(b_{1} \times d + b_{0} )} \right)^{3}\). The parameters b0 and b1 depend on stand age, quadratic mean tree diameter, dq, of the stand and the height of the tree with the mean diameter as follows: stand age and dq in 2012 were calculated based on the records from 2017 and the results of the diameter reconstruction. For reconstruction of the mean tree height in 2012, we used the corresponding site-specific height–age curves of the yield tables by Wiedemann (1943) and Jüttner (1955) for Scots pine and oak, respectively. Both yield tables are based on data from long-term experiments of the former Prussian Forest Research Station in Eberswalde. This network of experiments was established at the end of the nineteenth century and reached from high-quality sites in the south of Germany to very poor sites in east Prussia. This broad foundation is the reason why both tables are used till present in many countries involved in this study. Both yield tables represent moderate thinning and fit better to our stands than other available tables for heavy thinning. Any flaws in site indexing by using the tables by Wiedemann (1943) and Jüttner (1955) should be minor, as our plots are on average 72–73 years (Scots pine) and 73–77 years (oak) old. This means that the height extrapolation to age 100 is relatively small and uncritical as it reaches only 20–30 years and that in an age phase where the height growth is just about 1 m per decade (e.g. 3–4 m growth from age 70 to 100 in the case of yield class I.).
Based on the individual trees’ diameters and heights in year n, dn and hn, and their species-specific form factors, fn (Franz 1971), we calculated the stem volume in 2017 (\(v_{2017} = d_{2017}^{2} \times \pi /4 \times h_{2017} \times f_{2017}\)) and 2012 (\(v_{2012} = d_{2012}^{2} \times \pi /4 \times h_{2012} \times f_{2012}\)). Mean periodical volume increment within the 5-year period 2012–2017 was calculated accordingly (\({{iv}} = (v_{2017} - v_{2012} )/5\)). The applied form factors f and fn−5 depend on stem diameter and tree height at the beginning and end of each period. In this way, the changes of the form factors between 2012 and 2017 were taken into account. Mean periodical basal area increment (bai) was derived analogously. Stand-level PAIV and PAIBA were subsequently computed from the summation of single-tree values and up-scaled to one hectare.
The volume of the removed dead trees during the period 2012–2017 and the estimation of their stem volume and volume growth were based on the number, diameter, age of stumps and annual size growth of the mean tree. In the case of standing dead trees, volume was based on the diameter and height in 2017.
Minor proportions (< 10%) of other conifers were assigned to Scots pine and analogously other deciduous trees to oak. Stand data for 2017 and 2012 and the estimated growth characteristics in the 5-year period 2012–2017 were used for the subsequent evaluation or mixing effects.
Data evaluation
Nomenclature for quantifying mixing effects on stand growth
Stand productivity was quantified by the mean periodical stand basal area growth (PAIBA, m2 ha−1 year−1) and stem volume growth (PAIV, m3 ha−1 year−1) in the 5-year period 2012–2017. The results on volume production in mixed versus monospecific stands are essential for management planning and silvicultural decisions. However, it is more fraught with assumptions than stand basal area growth since calculating stem volume production requires the reconstruction of height growth and assumptions about the stem form factors, as described above. In contrast, stand basal area growth requires the measured stand characteristics and increment core measurements only. So, we report both PAIV as it is the more management-relevant property for quantifying stand productivity and PAIBA as it is more flawless for quantifying mixing effects.
Stand productivity of species 1 (Scots pine) and 2 (oak) in the monocultures was named p1 and p2. The productivities of the mixed stands in total were named p1,2. The productivity of species 1 and 2 in the mixed stands are pp1,(2) and pp(1),2, so that \(p_{1,2} = pp_{1,(2)} + pp_{(1),2}\). The mixing portions of species 1 and 2 are named m1 and m2, respectively, i.e. \(m_{1} + m_{2} = 1\). For the calculation of m1 and m2, see the next paragraph. The productivities of the species 1 and 2 in the mixed stand projected to a hectare are p1,(2) and p(1),2; they are calculated according to \(p_{1,(2)} = pp_{1,(2)} /m_{1}\) and \(p_{(1),2} = pp_{(1),2} /m_{2} ,\) respectively.
For quantifying the stand density and mixing proportions m1 and m2, we used the stand density index (SDI) by Reineke (1933). For monocultures, this index \({\text{SDI}} = N \times (25/d_{\text{q}} )^{ - 1.605}\) is based on the allometric relationship between tree number, N, and quadratic mean diameter of a stand, dq. Calculating a combined SDI of species in a mixed stand and the comparison of SDI values of different species must consider the species-specific growing area requirements and levels of the SDI. For this purpose, we first calculated the species-specific SDI values for the fully stocked monospecific stands of each triplet (\({\text{SDIMAX}}_{1}\),\({\text{SDIMAX}}_{2}\)). Then, we used these SDI values as proxies for the maximum stand densities at the respective sites and derived equivalence coefficients \(e_{2 \Rightarrow 1} = {\text{SDIMAX}}_{1} /{\text{SDIMAX}}_{2}\) and \(e_{1 \Rightarrow 2} = {\text{SDIMAX}}_{2} /{\text{SDIMAX}}_{1}\) for converting the SDI from one species to the other. Then, we used the equivalence coefficients to calculate a common density measure for species 1 and 2 in the mixed stand (\({\text{SDI}}_{{\underline{1} ,2}} = {\text{SDI}}_{1,(2)} + {\text{SDI}}_{(1),2} \times e_{2 \Rightarrow 1}\)). The underlining of 1 in \({\text{SDI}}_{{\underline{1} ,2}}\) indicates that the stand density of the mixed stand is standardized on species 1. In this way, the density of a given mixed stand can be compared with the density of the monospecific stands belonging to the triplet. Finally, the resulting \({\text{SDI}}_{{\underline{1} ,2}}\) value was used to calculate the relative density of the mixed-species stand in relation to the monoculture:
RD is a measure for over- or under-density of the mixed-species stands of the triplet compared with its neighbouring monocultures.
The stand density standardized to species 1 (\({\text{SDI}}_{{\underline{1} ,2}} = {\text{SDI}}_{1,(2)} + {\text{SDI}}_{(1),2} \times e_{2 \Rightarrow 1}\)) and the shares (\({\text{SDI}}_{1,(2)} /{\text{SDI}}_{{\underline{1} ,2}}\) resp. \({\text{SDI}}_{(1),2} \times e_{2 \Rightarrow 1} /{\text{SDI}}_{{\underline{1} ,2}}\)) of species 1 and 2 in the standardized SDI result in the following formulas for calculating the mixing proportions m1 and m2 of species 1 and 2:
Dirnberger and Sterba (2014) and Huber et al. (2014) presented similar approaches which consider the species-specific growing space requirements for calculating the mixing proportions.
Mean and dominant tree characteristics of the two monospecific plots of the triplets were compared with each other, e.g. hq1 versus hq2, dq1 versus dq2, …., ho1 versus ho2. The ratios Rhq = hq1/hq2 − 1, etc., quantify the relationship between both species in the monospecific stands and indicate positive (+y) or negative (−y) performance of species 1 over species 2. Differences between species mean sizes in mixed versus monospecific stands were tested analogously.
For analysing differences between standing volume, V, of the mixed stand and the monocultures, their volume V1,2 was compared with the weighted mean \(\hat{V}_{1,2} = V_{1} \times m_{1} + V_{2} \times m_{2}\) of the two neighbouring monocultures. For analogous comparison at the species level, the standing volumes VV1,(2) and VV(1),2 (V1,2 = VV1,(2) + VV(1),2) of the mixed stands were first up-scaled to one hectare, using the mixing proportions m1 and m2. Then, we compared VV1,(2) and VV(1),2 with the standing volume of the monocultures by the ratios RV1,(2) = VV1,(2)/m1/V1 and RV(1),2 = VV(1),2/m2/V2.
For quantifying over- or underyielding of the mixed versus monospecific stands, we used the mean periodic volume and basal area growth in the 5-year period 2012–2017. According to Pretzsch et al. (2010, 2013a), we first calculated the relative productivity, \(RP_{1,2} = p_{1,2} /\overset{\lower0.5em\hbox{$\smash{\scriptscriptstyle\frown}$}}{p}_{1,2}\), between mixed-species stands and monocultures for the stands as a whole. The relative productivity is the observed productivity of the mixed stand \(p_{1,2}\) divided by the weighted mean productivity \(\overset{\lower0.5em\hbox{$\smash{\scriptscriptstyle\frown}$}}{p}_{1,2}\) of the two monospecific stands. The latter was derived from the productivity of both species in the neighbouring monocultures, \(p_{1}\) and \(p_{2}\), and the mixing proportions \(m_{1}\) and \(m_{2}\) according to \(\overset{\lower0.5em\hbox{$\smash{\scriptscriptstyle\frown}$}}{p}_{1,2} = m_{1} \times p_{1} + m_{2} \times p_{2}\). Secondly, the relative productivity (RP) of species 1 and 2 in mixed versus monospecific stands was analysed by the ratios \({\text{RP}}_{1,(2)} = pp_{1,(2)} /m_{1} /p_{1}\) and \({\text{RP}}_{(1),2} = pp_{(1),2} /m_{2} /p_{2}\). Note that \(pp_{1,(2)}\) and \(pp_{(1),2}\) are the contributions of the productivity of species 1 and 2 to the mixed stand which adds up to \(p_{1,2}\) (\(p_{1,2} = pp_{1,(2)} + pp_{(1),2}\)). In contrast, the productivities \(p_{1,(2)}\) and \(p_{(1),2}\) are the contributions of both species to the mixed stand scaled up to 1 hectare (\(p_{1,(2)} = pp_{1,(2)} /m_{1}\) and \(p_{(1),2} = pp_{(1),2} /m_{2}\)).
For analysing dependencies between overyielding or underyielding and site fertility, we derived the height of the trees at the age of 100 years with the quadratic mean diameter, hq, as site index (SI) (see Pretzsch 2009, pp. 200–203 for the definition and calculation of hq). For that purpose, we used the height curve systems of the yield tables of Wiedemann (1943) and Jüttner (1955) for Scots pine and oak, respectively.
For characterizing the water supply at the 36 sites (see Supplement Table 1), we calculated the de Martonne index (1926) for each triplet based on the climate data from 1985 to 2015 [M = annual precipitation (mm)/(mean annual temperature (°C) +10)]. High de Martonne indices indicate sufficient water supply of trees, whereas low indices mean likelihood of drought. We used the index of de Martonne because of its minimal data requirement and wide use.
For analysing differences in crown extension, we calculated the tree’s crown projection area (CPA), expressed as a circular crown based on the squared mean of the 4 or 8 measured crown radii per tree. For CPA, we analysed the differences between mixed and monospecific stands at individual tree level.
Statistical analysis and models
For the statistical analyses to compare mixed and monospecific stand-level values, we applied linear mixed-effects models with nested random effects on country as proxy for the biogeographical zones and on location, i.e. triplet group level in order to consider autocorrelation effects on these levels. The number of the models refers to the results in Tables 2, 3 and 4. For analysing the differences between mixed and monospecific stand attributes, we used terms such as \({{hq_{\text{mix}} } \mathord{\left/ {\vphantom {{hq_{\text{mix}} } {hq_{\text{mono}} }}} \right. \kern-0pt} {hq_{\text{mono}} }} - 1\) for comparing the mean heights. Any positive or negative differences directly indicate the relative difference between mixed and monospecific stands. For example, \({{hq_{\rm mix} } \mathord{\left/ {\vphantom {{hq_{\rm mix} } {hq_{\rm mono} }}} \right. \kern-0pt} {hq_{\rm mono} }} - 1 = 0.20\) indicates that the trees are by 20% taller in the mixed stands than in the monoculture.
Model 1: \({{ho_{{{\text{mix}},ijk}} } \mathord{\left/ {\vphantom {{ho_{{{\text{mix}},ijk}} } {ho_{{{\text{mono}},ijk}} }}} \right. \kern-0pt} {ho_{{{\text{mono}},ijk}} }} - 1 = a + b_{i} + b_{ij} + \varepsilon_{ijk}\)
resp. \({{do_{{{\text{mix}},ijk}} } \mathord{\left/ {\vphantom {{do_{{{\text{mix}},ijk}} } {do_{{{\text{mono}},ijk}} }}} \right. \kern-0pt} {do_{{{\text{mono}},ijk}} }} - 1 = a + b_{i} + b_{ij} + \varepsilon_{ijk}\) and analogous approaches based on \({{hq_{mix,ijk} } \mathord{\left/ {\vphantom {{hq_{mix,ijk} } {hq_{mono,ijk} }}} \right. \kern-0pt} {hq_{mono,ijk} }} - 1\) and \({{dq_{{{\text{mix}},ijk}} } \mathord{\left/ {\vphantom {{dq_{{{\text{mix}},ijk}} } {dq_{{{\text{mono}},ijk}} }}} \right. \kern-0pt} {dq_{{{\text{mono}},ijk}} }} - 1\).
Analogous models were used for testing the mean tree characteristics hq and dq of the monospecific stand against each other. The only fixed effect required in this model is the intercept a. If it is significantly greater or lower than zero, a mixture effect was identified as explained above. The indices i, j and k represent the levels country, triplet group and triplet. The variable b represents random effects on the indicated levels. Model 1 was applied for Scots pine and oak, respectively; the variables refer to the corresponding species (Table 2).
Model 2: \({{{\text{SDI}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{SDI}}_{{{\text{obs}},ijk}} } {{\text{SDI}}_{{{ \exp },ijk}} }}} \right. \kern-0pt} {{\text{SDI}}_{{{ \exp },ijk}} }} - 1 = a + b_{i} + b_{ij} + \varepsilon_{ijk}\).
The only fixed effect required in this model is also intercept a. If it is significantly greater or lower than zero, a mixture effect was identified. The indices i, j and k represent the levels country, triplet group and triplet. The variable b represents random effects on the indicated levels. Model 2 has been applied for the total stand (Table 2).
Model 3: \({{{\text{PAIBA}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIBA}}_{{{\text{obs}},ijk}} } {{\text{PAIBA}}_{\exp ,ijk} }}} \right. \kern-0pt} {{\text{PAIBA}}_{\exp ,ijk} }} - 1 = a + b_{i} + b_{ij} + \varepsilon_{ijk}\) and analogous approach based on \({{{\text{PAIV}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIV}}_{{{\text{obs}},ijk}} } {\text{PAIV}}}} \right. \kern-0pt} {\text{PAIV}}}_{{{ \exp },ijk}} - 1\).
Also here, the only fixed effect required in this model is the intercept a. If it is significantly greater or lower than zero, a mixture effect was identified. The indices i, j and k represent the levels country, triplet group and triplet. The variable b represents random effects on the indicated levels. Model 3 has been applied at species level and for total stand (Table 2).
Model 4: \({{{\text{PAIBA}}_{{{\text{obs,}}ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIBA}}_{{{\text{obs,}}ijk}} } {{\text{PAIBA}}_{\exp ,ijk} }}} \right. \kern-0pt} {{\text{PAIBA}}_{\exp ,ijk} }} - 1 = a_{0} + a_{1} \times SI_{ijk} + b_{i} + b_{ij} + \varepsilon_{ijk}\) and analogous approach based on \({{{\text{PAIV}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIV}}_{{{\text{obs}},ijk}} } {\text{PAIV}}}} \right. \kern-0pt} {\text{PAIV}}}_{\exp ,ijk} - 1\).
\({\text{SI}}_{ijk}\) is the site index at age 100 years per triplet. Applying Model 4 at species level, \({\text{SI}}_{ijk}\) refers to the site index of the corresponding species (derived from the monospecific stands). In the case of the total stand, Model 4 was applied using \({\text{SI}}_{ijk}\) as site index in a first model version from Scots pine and in a second model version from oak (both derived from the monospecific stands), respectively (Table 3, Supplement Table 3). The indices i, j and k represent the levels country, triplet group and triplet. The fixed-effects parameter is a; the variable b represents the random effects on the indicated levels.
Model 5: \({{{\text{PAIBA}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIBA}}_{{{\text{obs}},ijk}} } {{\text{PAIBA}}_{\exp ,ijk} }}} \right. \kern-0pt} {{\text{PAIBA}}_{\exp ,ijk} }} - 1 = a_{0} + a_{1} \times hq_{{{\text{S}} . {\text{pine}},ijk}} /hq_{{{\text{oak}},ijk}} + b_{i} + b_{ij} + \varepsilon_{ijk}\) and analogous approach based on \({{{\text{PAIV}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIV}}_{{{\text{obs}},ijk}} } {\text{PAIV}}}} \right. \kern-0pt} {\text{PAIV}}}_{\exp ,ijk} - 1\).
The independent variable \(hq_{{{\text{S}} . {\text{pine}},ijk}} /hq_{{{\text{oak}},ijk}}\) refers to the mixed stand and quantifies any lead in height of Scots pine in relation to oak. The indices i, j and k represent the levels country, triplet group and triplet. The fixed-effects parameter is a; the variableb represents the random effects on the indicated levels. Model 5 has been applied at species level and total stand (Table 3, Supplement Table 3).
Model 6: \({{{\text{PAIBA}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIBA}}_{{{\text{obs}},ijk}} } {{\text{PAIBA}}_{\exp ,ijk} }}} \right. \kern-0pt} {{\text{PAIBA}}_{\exp ,ijk} }} - 1 = a_{0} + a_{1} \times M_{ijk} + b_{i} + b_{ij} + \varepsilon_{ijk}\) and analogous approach based on \({{{\text{PAIV}}_{{{\text{obs}},ijk}} } \mathord{\left/ {\vphantom {{{\text{PAIV}}_{{{\text{obs}},ijk}} } {\text{PAIV}}}} \right. \kern-0pt} {\text{PAIV}}}_{\exp ,ijk} - 1\).
\(M_{ijk}\) represents the index of de Martonne that increases with increasing water supply. The indices i, j and k represent the levels country, triplet group and triplet. The fixed-effects parameter is a; the variable b represents the random effects on the indicated levels. Model 6 has been applied at species level and total stand (Table 3, Supplement Table 3).
Another model was fitted in order to explore mixture effects on the tree-level allometry between crown projection area, CPA and stem diameter at breast height, d:
Model 7: \(\ln ({\text{CPA}}_{ijklm} ) = a_{0} + a_{1} \times \ln (d_{ijklm} ) + a_{2} \times {\text{mix}}_{ijkl} + a_{3} \times \ln (d_{ijklm} ) \times {\text{mix}}_{ijkl} + b_{i} + b_{ij} + b_{ijk} + b_{ijkl} + \varepsilon_{ijklm}\). The fixed effect variable mix is categorical with mix = 0 for monocultures as reference and mix = 1 for mixed-species stands. The model in general can be seen as a typical log-linear allometric relation between size variables (CPA, d) with mixture effects on both, the intercept and the allometric slope. The fixed effects parameters are \(a_{0} - a_{3}\). The random effects \(b_{i}\), \(b_{j}\), \(b_{k}\) and \(b_{l}\) cover the levels country (i), triplet group (j), triplet (k) and plot (l). The level of the single tree is represented by the index m. Model 7 was applied for Scots pine and oak, respectively (Table 4).
The statistical software R 3.4.1 (R Core Team 2018) was used for all calculations, in particular the function lme from the package nlme (Pinheiro et al. 2017).
Results
Quadratic mean tree height (hq) on monoculture was on average only 1.0 m higher for Scots pine (22.7 m) than for oak (21.7 m) (Table 1, Fig. 2). Stand density as expressed by the SDI was higher in Scots pine monocultures (890 N ha−1) compared with oak monocultures (733 N ha−1) (Table 1, Fig. 2). The same was true for standing stock, which on average amounted to 436 m3 ha−1 in Scots pine and 360 m3 ha−1 in oak monocultures. Scots pine was the more productive species with an average PAIV of 10.5 m3 ha−1 year−1 compared with oak (9.1 m3 ha−1 year−1) (Table 1).
Growth and yield of Scots pine (x-axis) compared to oak (y-axis) in the monospecific plots of the 36 triplets. Values on the bisector line indicate equality of the stand characteristics of both species. a Quadratic mean height, hq (m), b stand density index, SDI (trees ha−1), c standing merchantable (> 7 cm at the smaller end) stem volume, V (m3 ha−1), and d mean periodic increment of the stand volume, PAIV (m3 ha−1 year−1), during the 5 years before the stand inventory. Small empty symbols represent the observed values, and large filled symbols indicate the mean values of all 36 triplets
Mixing reactions at the stand level
Tree heights and diameters of Scots pine tend to be superior in mixed compared to monospecific stands (Table 2). In the case of dq and do, the differences were statistically significant (Fig. 3). On the contrary, the tree dimensions of oak tended to be inferior in mixed compared to monospecific stands, but differences were never significant (Model 1). There were no differences in stand density between mixed and monospecific stands (Model 2). PAIBA and PAIV at the species and stand level were higher in mixed compared to monospecific stands (Model 3). This is indicated by the fact that the estimates of fixed effects were all greater than a = 0. In the case of PAIV of Scots pine, we found a significant (p < 0.10) superiority by 9.1% in mixed stands. In the subsequent tables, we only report the fixed effects of the models 1–7.
Quadratic mean stem diameter dq (a), dominant diameter do (b), periodic stand basal area increment PAIBA (c), and periodic stand volume increment PAIV (d) of Scots pine in the mixed stands compared with the monocultures. Small empty symbols represent the observed values, and large filled symbols indicate the mean values of all 36 triplets
Dependency of mixing effects on stand and site variables
Any dependency of mixing effects on stand and site variables is shown for the PAIBA. In contrast to PAIV, it is not based on any hypotheses of retrospective height and form factor development (see “Data evaluation” section). The site index showed opposing effects on Scots pine and oak (Table 3, Fig. 4). The positive mixing effect on Scots pine increased with its species-specific site index (p < 0.01). On the other hand, the benefit of oak was cancelled out by increasing site index of oak (p < 0.01). In other words, oak benefited more from the mixture on poor sites. There were also significant increases in the mixing effect with increasing site indices at the total stand level. Interestingly, oak also significantly benefited when growing in stands where Scots pine showed superior height growth (Table 3, Model 5). With increasing de Martonne index, both Scots pine (p < 0.01) and the mixed stand in total (p < 0.05) benefited from growing in mixture (Table 3, Model 6). Note that there was an underyielding for SI values below 24 m for Scots pine and for SI values above 30 m for oak. There was also an underyielding for Scots pine below a Martonne index of 30 and for the total stand below a Martonne index of 30, too.
Effect of the species-specific site index and the index of de Martonne on the growth of mixed versus monospecific stands of Scots pine and oak: a the relative basal area increment of Scots pine in mixed versus monospecific stands increases with increasing site index of Scots pine. b The relative basal area increment of oak in mixed versus monospecific stands decreases with increasing site index of oak. c The relative basal area increment of Scots pine in mixed versus monospecific stands increases with increasing index of de Martonne. d The relative basal area increment of the mixed stand in total versus the weighted mean of the monospecific stands increases with increasing index of de Martonne
Effects of species mixing on the tree structure
Both species showed a significant change of tree allometry in mixed versus monospecific stands (Table 4, Model 7). In the case of Scots pine, the CPA was slightly higher in monospecific stands for small trees and the positive mixing effect increased with tree diameter (Fig. 5). In the case of oak, the effect was stronger for trees of all diameters and it did not change very much with increasing size. Note that the mixing effect displayed in Table 4 is composed of two components, the effect of the dummy variable mix and the interaction between mix and stem diameter.
Beneficiary and benefactor
For forest management, overyielding of both species may be of special interest; an additional cubic metre of oak wood might be as welcome as an additional one of Scots pine wood. For understanding the stand dynamics and coexistence of the associated species, it may also be important to analyse the extent to which it is beneficial to grow in interspecific compared to intraspecific neighbourhoods. A comparison between RP1,(2) and RP(1),2 may show the species-specific benefits of growing in mixture. In the case of Scots pine and oak, the comparison between RP of one and the other species is most revealing since both species have very similar stand productivities in terms of stem volume (10 m3 ha−1 year−1) and similar wood densities of around 550–650 kg m−3 (Knigge and Schulz 1966, p. 135), which means that overyielding of, for example, 1.10 in both cases means a plus of 1 m3 ha−1 year−1 or approximately 0.55–0.65 t ha−1 year−1.
By plotting the relative productivity of oak and pine in the mixed stand compared to the monoculture, it becomes obvious how both species perform when growing in mixture (Fig. 6).
Relative productivity in terms of PAIBA for oak and pine in mixed stands compared to monoculture. Points in quarter I and III indicate over- or underyielding, respectively, for both species. Points in quarter IV indicate strong relative productivity advantage of oak over Scots pine in mixtures and points in II indicate advantage of Scots pine over oak. The large grey point indicates the mean mixing reaction on stand productivity
If both species react similarly to mixing, points are located close along the ascending line. Their RP1,(2) and RP(1),2 combination lies on or close to the increasing bisector line. All points lying above the decreasing line indicate overyielding at the stand level. The points below the decreasing line represent underyielding. The wider the range to above or below, the more pronounced the species response. Points in quarter I and III indicate over- or underyielding for both species, respectively. Points in quarter IV and II indicate a strong relative advantage of species 1 over 2 and species 2 over 1, respectively.
In 25 out of the total 36 cases (69%), both species lie above the decreasing line and indicate overyielding at the stand level. Fourteen cases (39%) and also the mean value (large grey point) lie in quadrant I and indicate a benefit of both species by the mixture, i.e. the advantage of one species is not gained at the expense of the other. In 5 out of 36 cases (14%), the values are in quadrant III and indicate a disadvantage for both species. In 17 out of 36 cases (47%), the points lie in quadrant IV or II, indicating that the benefit of one species is gained at the expense of the other. In 7 cases (19%), Scots pine is the beneficiary and oak the benefactor (points in quadrant II). In 10 cases (28%), it is the other way around (points in quadrant IV).
Regarding pros and cons of mixed-species stands in forest practice, it may be also interesting that in 11 out of the 36 cases (~ 31%) the basal area growth of the mixed stand of Scots pine and oak exceeded even the growth of the monoculture with the highest growth of the respective triplet (transgressive overyielding). The analogous statistic for the volume growth was 12 out of the total 36 cases (~ 33%). For 8 out of 36 cases (~ 22%), transgressive overyielding has been observed for both basal area and volume increment.
Discussion
Growth and yield effects of tree species mixing are of interest for both ecological analysis of tree species interactions (change in access, uptake and use efficiency of resources) and economical decisions in favour of mixed-species stands in forest practice.
We established the triplets in mainly 40–90-year-old stands (70 years on average) when they are in the phase between the culmination of the annual and mean annual volume increment and represent both species’ productivity well. Until that age, the observed stands have had sufficient time to develop their typical stand structures, to modify their stand density, alter the composition of the humus layer and the conditions of the mineral soil, and to adapt stem morphology and crown allometry for the identification of the surrounding trees.
Compared to other mixtures, the Scots pine and oak stands of this study showed only minor increases in productivity, stand density and species-specific changes in size growth. On average, the standing stem volume was 436 and 360 m3 ha−1 in the monocultures of Scots pine and oak, respectively, and 418 m3 ha−1 in the mixed stands. The corresponding periodical annual volume increment amounted to 10.5 and 9.1 m3 ha−1 year−1 in the monocultures and 10.5 m3 ha−1 year−1 in the mixed stands. Scots pine showed a 10 resp. 7% larger quadratic mean and dominant stem diameter in mixed stands and tended to achieve a 9% higher growth of basal area and volume compared with neighbouring monocultures. In total, the superior productivity of mixed stands compared to monocultures increased with water supply. In mixture, both species had 5–10% wider crowns than in monocultures (Fig. 5).
The mean productivity differences between mixed and monospecific stands of Scots pine and oak were smaller than in mixtures of Norway spruce and European beech, sessile oak and European beech, or Scots pine and European beech (Pretzsch et al. 2010, 2013a, b, 2015). Mixtures of light-demanding species (Scots pine, European larch (Larix decidua Mill.), sessile oak) and shade-tolerant species (European beech, silver fir (Abies alba Mill.)) can show overyielding of stem volume growth of 20–30% (Jactel et al. 2018). Mixing of deep-rooting broadleaved species (European beech) and shallow-rooting conifers (Norway spruce) can result in overyielding by 10–20% (Pretzsch 2018). The mixture of nitrogen-fixing species (red alder (Alnus rubra Bong.), acacia spec.) and non-nitrogen-fixing species (Norway spruce, eucalyptus spec.) may even enable overyielding by up to 50% (DeBell et al. 1989; Forrester et al. 2006). However, different traits do not necessarily result in overyielding and even if there are no obvious differences between the species there can be mixing effects as shown by Donoso et al. (2011), Forrester and Smith (2012), Sharma et al. (2008), or Staudhammer et al. (2009).
Figure 6 underlines that in the majority of cases stand productivity was higher in mixed compared to monospecific stands of Scots pine and oak and that the roles of both species are rather balanced. In some cases, it was Scots pine and in a similar number of cases oak that benefited more from the mixture. In 39% of the cases, and also on average, the observations indicated a benefit of both species by the mixture. Certainly, overyielding at the stand or species level does not at all indicate a reduced level of competition; it rather indicates that both species compete with each other, but on a higher level of stand productivity. For both species, it is still essential which is the more vital, faster growing and more competitive one. However, Fig. 6 reveals that even in fully stocked stands none of the two species is so overwhelmingly superior that it might endanger the presence of the other one.
This pattern of stand overyielding among triplets (Fig. 6) is in line with other studies. Compared with other mixtures, the productivity gains in mixtures of Scots pine and oak were either significant but minor (Brown 1992; Lu et al. 2016, 2018) or there was no superiority of mixed versus monospecific stands found at all (Toïgo et al. 2015). However, Toïgo et al. (2018) found that the less shade-tolerant an admixed species was, the higher was the positive effect on Q. petraea productivity. Using a modelling approach, Perot and Picard (2012) also reported overyielding in this mixture. Steckel et al. (2019), utilizing a triplet transect spanning from Southern Germany to Eastern Denmark, found that the mixing of Scots pine and oak resulted in a higher annual volume productivity which amounted to 14% on average and increased with annual water supply. Actually, the latter work represented a subset of the gradient in this study, encompassing sites that are rather well supplied with water and nutrients. In line with the findings achieved in the former study, we also found higher overyielding on well-supplied sites, but the gradient of the study in hand reached even further to warm and dry and cold and moist sites (Supplement Table 1).
Similar to our findings, previous studies also indicate some variability on species overyielding and benefactor/beneficiary roles. Lu et al. (2018) and Toïgo et al. (2015) found that oak was the main benefactor in Dutch and French Scots pine–oak mixtures, respectively. del Río et al. (2013), based on a small sample from Northern Spain, showed that only Scots pine benefited in the mixture, whereas Brown (1992) reported positive mixing effect for the two species (quarter 1 in Fig. 6) in the Gisburn experiment in England.
Obviously, Scots pine and oak are less complementary compared with mixtures of shade-tolerant and light-demanding species or nitrogen-fixing and non-nitrogen-fixing species. However, there are some differences in traits between Scots pine and oak, which generate the potential for synergy. How this potential is exploited varies strongly depending on the site conditions along the transect across Europe.
The minimal light requirement of shade leaves/needles in relation to light above canopy (100%) amounts to 10% for Scots pine and 4–7% for sessile/common oak (Ellenberg and Leuschner 2010, pp. 103–105), i.e. oaks can survive with less light below Scots pine. The difference in light compensation point of 27 to 17 μmol m−2 s−1 for Scots pine and oak, respectively, substantiates the higher shade tolerance of oak (Ellenberg and Leuschner 2010, pp. 103–105). The light compensation points refer to sun leaves/needles in summer for Amax (i.e. when light-saturated photosynthesis occurs under normal CO2 concentration). The wider crown extension in mixture, especially in the case of oak (del Río et al. 2019), suggests a competition reduction by spatial niche complementary as shown by Barbeito et al. (2017) and Bayer et al. (2013) for other mixtures of conifers and broadleaved trees. In comparison with the monocultures, oak may grow slightly shaded by Scots pine. The latter, on the other hand, seem to grow slightly higher since it may be less impaired by other pines in the lower canopy when growing in mixture with oak. The same but only weak spatial niche complementary might apply in the root space (Bello et al. 2019). Both mechanisms generate the potential for a higher resource uptake, photosynthetic capacity and growth. In addition, there might be a temporal complementary of water use, as the evergreen Scots pine can better utilize water in spring when oak is still leafless (Goisser et al. 2016). This might explain that oak growing in mixture with Scots pine was found to suffer more from spring droughts, while Scots pine seems to be more affected by summer droughts (Merlin et al. 2015).
The relative superiority of pine with increasing water supply (Fig. 4) may result from its better adaptation to moist and cold climate, whereas oak may benefit from warmer and drier conditions as its niche reaches further into continental and Mediterranean climate conditions. Despite these species differences, overyielding at stand level increased with water supply, in accordance with the general trend found in the meta-analysis by Jactel et al. (2018). The causal explanation might be that on sites with sufficient water and nutrient supply light becomes the limiting factor of growth and light-related complementarity of mixed species may be more useful than on poor sites. On poor sites with limitation of below-ground resources, light complementarity may be less useful as the water and nutrient supply does not allow full light exploitation.
As the climatic envelopes of both species reach up to 14–15 °C mean annual temperature and 300–400 mm annual precipitation, the mixture of Scots pine and oak may become even more important under future warmer and drier climate (Kölling 2007). Our results showed that for Scots pine the advantages of growing in mixture increased with site index and water supply; for oak, they decreased with site index. This resulted in a rather constant level of overyielding at the stand level, but may indicate that under increased stress levels due to global warming, oak may be more competitive than Scots pine when growing together.
With a current area of approximately 1.3 × 106 ha and a potential area of 35 × 106 ha (Brus et al. 2012), the mixture of Scots pine and oak is important. Certainly, there are other reasons beyond growth and yield that question mixtures with oaks. Morphological traits in particular qualify oak as habitat trees (Dieler et al. 2017; Horak et al. 2014; Johnson et al. 2009) and significantly increase diversity of cryptogam species (Preikša et al. 2015). On the other hand, the morphological variability of oak may reduce its wood quality when cultivated in mixed-species stands (Benneter et al. 2018; Pretzsch and Rais 2016).
It was shown that species associated with Scots pine decrease in mixture with European beech, since the latter strongly determines microclimate (Heinrichs et al. 2019). As a result, for example the composition of herbaceous plant species in mixtures of beech and conifers was more similar to pure beech stands than to conifer stands (Heinrichs et al. 2019). We assume that similar negative effects on species richness by mixing tree species are unlikely to occur in a similar way in Scots pine–oak mixtures since the light regimes in monocultures of the two species do not differ as much as they do between oak and beech or Scots pine and beech. Finally, the mixture of Scots pine and oak appears almost alternativeless when aiming to create economically and ecologically valuable mixtures of Scots pine with broadleaved deciduous in the regions beyond the range of European beech.
Both species have shown relatively similar height development; in the first decades, Scots pine may grow slightly quicker. However, in mature stands their height growth is still very synchronous and the reported maximum final heights of 48–50 m are also rather similar (Ellenberg and Leuschner 2010, pp. 103–105). On good-quality sites, both species reach a height of about 30 m at the age of 120 years; Scots pine may be harvested at that point in time, whereas oak proceeds until age 150–200 years reaching tree heights of 30–35 m. From a silvicultural point of view, the different rotation periods of Scots pine and oak provide the option to avoid clear-cuts during the final harvest. When Scots pine is removed by one or more shelterwood cuttings, natural tree regeneration could be established depending on the density of the remaining mature oak (Dobrowolska 2006). On the contrary, regeneration of oak under Scots pine is more uncertain, especially when wild herbivores (such as roe deer and red deer) are present in the forest (Danell et al. 2003; Gill 1992). Nevertheless, the stands on most of the triplets were naturally regenerated and the fact that both tree species are present in the upper canopy until age 60–80 without strong silvicultural interventions underlines this balanced competition. Rather similar height development and light ecology means that both species can be mixed in individual tree or group mixtures. However, it seems that the mutual beneficial effect of mixing the two species is the highest in intimate mixtures (Ngo Bieng et al. 2013). In conclusion, mixtures of Scots pine and oak seem to be a reliable mixture that needs no frequent interventions to keep a desired stand composition, though relatively low gains in yield can be expected. However, with ongoing climate changes it may become less important that mixtures increases stand productivity; the mixture should also work well under harsh conditions which seem to be the case for Scots pine and oak.
References
Aas G (1991) Kreuzungsversuche mit Stiel-und Traubeneichen (Quercus robur L. und Q. petraea Mattl. Liebl.). Allg Forst Jagdztg 162:141–145
Aguirre A, del Río M, Condés S (2019) Productivity estimations for monospecific and mixed pine forests along the Iberian Peninsula aridity gradient. Forests 10:430
Ammer C (2019) Diversity and forest productivity in a changing climate. New Phytol 221:50–66
Augusto L, Ranger J, Binkley D, Rothe A (2002) Impact of several common tree species of European temperate forests on soil fertility. Ann For Sci 59(3):233–253
Barbeito I, Dassot M, Bayer D, Collet C, Drössler L, Löf M, del Rio M, Ruiz-Peinado R, Forrester DI, Bravo-Oviedo A, Pretzsch H (2017) Terrestrial laser scanning reveals differences in crown structure of Fagus sylvatica in mixed vs. pure European forests. For Ecol Manag 405:381–390
Bauhus J, Forrester DI, Gardiner B, Jactel H, Vallejo R, Pretzsch H (2017) Ecological stability of mixed-species forests. In: Pretzsch H, Forrester DI, Bauhus J (eds) Mixed-species forests. Ecology and Management. Springer, Berlin, pp 337–382
Bayer D, Seifert S, Pretzsch H (2013) Structural crown properties of Norway spruce (Picea abies [L.] Karst) and European beech (Fagus sylvatica [L.]) in mixed versus pure stands revealed by terrestrial laser scanning. Trees 27(4):1035–1047
Bello J, Hasselquist NJ, Vallet P, Kahmen A, Perot T, Korboulewsky N (2019) Complementary water uptake depth of Quercus petraea and Pinus sylvestris in mixed stands during an extreme drought. Plant Soil 437:93–115. https://doi.org/10.1007/s11104-019-03951-z
Benneter A, Forrester DI, Bouriaud O et al (2018) Tree species diversity does not compromise stem quality in major European forest types. For Ecol Manag 422:323–337
Bertness MD, Callaway R (1994) Positive interactions in communities. Trends Ecol Evol 9:191–193
Biber P, Borges JG, Moshammer R, Barreiro S, Botequim B, Brodrechtová Y, Brukas V, Chirici G, Cordero-Debets R, Corrigan E, Eriksson LO, Favero M, Galev E, Garcia-Gonzalo J, Hengeveld G, Kavaliauskas M, Marchetti M, Marques S, Mozgeris G, Navrátil R, Nieuwenhuis M, Orazio C, Paligorov I, Pettenella D, Sedmák R, Smrecek R, Stanislovaitis A, Tomé M, Trubins R, Tucek J, Vizzarri M, Wallin I, Pretzsch H, Sallnäs O (2015) How sensitive are ecosystem services in European forest landscapes to silvicultural treatment? Forests 6(5):1666–1695
Björse G, Bradshaw R (1998) 2000 years of forest dynamics in southern Sweden: suggestions for forest management. For Ecol Manag 104:15–26
Brown AHF (1992) Functioning of mixed-species stands at Gisburn, NW England. The ecology of mixed-species stands of trees. In: Cannel MGR, Malcolm DC, Robertson PA (eds) The ecology of mixed-species stands of trees. Blackwell, Oxford, pp 125–150
Brus DJ, Hengeveld GM, Walvoort DJJ, Goedhart PW, Heidema AH, Nabuurs GJ (2012) Statistical mapping of tree species over Europe. Eur J For Res 131:145–157
Caldwell MM, Dawson TE, Richards JH (1998) Hydraulic lift: consequences of water efflux from the roots of plants. Oecologia 113(2):151–161
Danell K, Bergström R, Edenius L, Ericsson G (2003) Ungulates as drivers of tree population dynamics at module and genet levels. For Ecol Manag 181:67–76. https://doi.org/10.1016/S0378-1127(03)00116-6
de Martonne E (1926) Une novelle fonction climatologique: L’indice d’aridité. La Météorologie 21:449–458
DeBell DS, Whitesell CD, Schubert TH (1989) Using N2-fixing Albizia to increase growth of Eucalyptus plantations in Hawaii. For Sci 35(1):64–75
del Río M, Condés S, Sterba H (2013) Productividad en masas mixtas vs. masas puras: influencia de la espesura en la interacción entre especies. In: Actas del 6°Congreso Forestal Español 6CFE01-121:13. Sociedad Española de Ciencias Forestales, Pontevedra
del Río M, Pretzsch H, Ruiz-Peinado R, Ampoorter E, Annighöfer P, Barbeito I, Bielak K, Brazaitis G, Coll L, Drössler L, Fabrika M, Forrester DI, Heym M, Hurt V, Kurylyak V, Löf M, Lombardi F, Madrickiere E, Matovic B, Mohren F, Motta R, den Ouden J, Pack M, Ponette Qu, Schütze G, Skrzyszewski J, Sramek V, Sterba H, Stojanovic D, Svoboda M, Zlatanov TM, Bravo-Oviedo A (2017) Species interactions increase the temporal stability of community productivity in Pinus sylvestris-Fagus sylvatica mixtures across Europe. J Ecol 105(4):1032–1043
del Río M, Bravo-Oviedo A, Ruiz-Peinado R, Condés S (2019) Tree allometry variation in response to intra- and inter-specific competitions. Trees 33:121–138
Dieler J, Uhl E, Biber P, Müller J, Rötzer T, Pretzsch H (2017) Effect of forest stand management on species composition, structural diversity, and productivity in the temperate zone of Europe. Eur J For Res 136(4):739–766
Dirnberger GF, Sterba H (2014) A comparison of different methods to estimate species proportions by area in mixed stands. For Syst 23:534–546
Dobrowolska D (2006) Oak natural regeneration and conversion processes in mixed Scots pine stands. Forestry 79(5):503–513
Donoso PJ, Muñoz AA, Thiers O, Soto DP, Donoso C (2011) Effects of aspect and type of competition on the early performance of Nothofagus dombeyi and Nothofagus nervosa in a mixed plantation. Can J For Res 41:1075–1081
Eaton E, Caudullo G, Oliveira S, de Rigo D (2016) Quercus robur and Quercus petraea in Europe: distribution, habitat, usage and threats. In: San-Miguel-Ayanz J, de Rigo D, Caudullo G, Houston Durrant T, Mauri A (eds) European atlas of forest tree species. Publication Office of the European Union, Luxembourg, pp e01c6df+
Ellenberg H, Leuschner C (2010) Vegetation Mitteleuropas mit den Alpen in ökologischer, dynamischer und historischer Sicht. Eugen Ulmer, Stuttgart, p 1334
Felton A, Nilsson U, Sonesson J, Felton AM, Roberge JM, Ranius T, Drössler L (2016) Replacing monocultures with mixed-species stands: ecosystem service implications of two production forest alternatives in Sweden. Ambio 45(2):124–139
Forrester DI (2014) The spatial and temporal dynamics of species interactions in mixed-species forests: from pattern to process. For Ecol Manag 312:282–292
Forrester DI, Smith RGB (2012) Faster growth of Eucalyptus grandis and Eucalyptus pilularis in mixed-species stands than monocultures. For Ecol Manag 286:81–86
Forrester DI, Bauhus J, Cowie AL, Vanclay JK (2006) Mixed-species plantations of Eucalyptus with nitrogen-fixing trees: a review. For Ecol Manag 233(2–3):211–230
Forrester DI, Ammer C, Annighöfer PJ, Barbeito I, Bielak K, Bravo-Oviedo A, Hurt V (2018) Effects of crown architecture and stand structure on light absorption in mixed and monospecific Fagus sylvatica and Pinus sylvestris forests along a productivity and climate gradient through Europe. J Ecol 106(2):746–760
Franz F (1971) Funktionen und Tabellen der Derbholzformhöhen für die wichtigsten Baumarten in Bayern. München, Manuskriptdruck, unveröffentlicht
Franz F, Bachler J, Deckelmann E, Kennel E, Kennel R, Schmidt A, Wotschikowsky U (1973) Bayerische Waldinventur 1970/71. Inventurabschnitt I: Großrauminventur Aufnahme- und Auswertungsverfahren. Forstliche Versuchsanstalt München, vol 11
Gamfeldt L, Snäll T, Bagchi R, Jonsson M, Gustafsson L, Kjellander P, Mikusiński G (2013) Higher levels of multiple ecosystem services are found in forests with more tree species. Nat Commun 4:1340
Gill RMA (1992) A review of damage by mammals in north temperate forests: 3. Impact on trees and forests. Forestry (Oxf.) 65:363–388. https://doi.org/10.1093/forestry/65.4.363-a
Goisser M, Geppert U, Rötzer T, Paya A, Huber A, Kerner R, Bauerle T, Pretzsch H, Pritsch K, Häberle KH, Matyssek R, Grams TEE (2016) Does belowground interaction with Fagus sylvatica increase drought susceptibility of photosynthesis and stem growth in Picea abies? For Ecol Manag 375:268–278
Griess VC, Knoke T (2013) Bioeconomic modeling of mixed Norway spruce-European beech stands: economic consequences of considering ecological effects. Eur J For Res 132(3):511–522
Heinrichs S, Ammer C, Mund M et al (2019) Landscape-scale mixtures of tree species are more effective than stand-scale mixtures for biodiversity of vascular plants. Bryophyt Lichens For 10:73
Heym M, Ruiz-Peinado R, del Río M, Bielak K, Forrester DI, Dirnberger G, Barbeito I, Brazaitis G, Ruskytke I, Coll L, Fabrika M, Drössler L, Löf M, Sterba H, Hurt V, Kurylyak V, Lombardi F, Stojanovic D, den Ouden J, Motta R, Pach M, Skrzyszewski J, Ponette Q, de Streel G, Sramek V, Cihak T, Zlatanov TM, Avdagic A, Ammer Ch, Verheyen K, Wlodzimierz B, Bravo-Oviedo A, Pretzsch H (2017) EuMIXFOR empirical forest mensuration and ring width data from pure and mixed stands of Scots pine (Pinus sylvestris L.) and European beech (Fagus sylvatica L.) through Europe. Ann For Sci 74(S9):63. https://doi.org/10.1007/s13595-017-0660-z
Heym M, Bielak K, Wellhausen K, Uhl E, Biber P, Perkins D, Steckel M, Thurm EA, Rais A, Pretzsch H (2018) A new method to reconstruct recent tree and stand attributes of temporary research plots: new opportunity to analyse mixed forest stands. IntechOpen, Conifers, Chapter 2, https://doi.org/10.5772/intechopen.79682
Horak J, Vodka S, Kout J, Halda JP, Bogusch P, Pech P (2014) Biodiversity of most dead wood-dependent organisms in thermophilic temperate oak woodlands thrives on diversity of open landscape structures. For Ecol Manag 315:80–85
Houston Durrant T, de Rigo D, Caudullo G (2016) Pinus sylvestris in Europe: distribution, habitat, usage and threats. In: San-Miguel-Ayanz J, de Rigo D, Caudullo G, Houston Durrant T, Mauri A (eds) European atlas of forest tree species. Publication Office of the European Union, Luxembourg, pp e016b94+
Huber MO, Sterba H, Bernhard L (2014) Site conditions and definition of compositional proportion modify mixture effects in Picea abies—Abies alba stands. Can J For Res 44:1281–1291
IPCC (2013) Summary for policymakers. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: the physical science basis. Contribution of working group 1 to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Jactel H, Brockerhoff EG (2007) Tree diversity reduces herbivory by forest insects. Ecol Lett 10:835–848
Jactel H, Gritti ES, Drössler L, Forrester DI, Mason WL, Morin X, Pretzsch H, Castagneyrol B (2018) Positive biodiversity–productivity relationships in forests: climate matters. Biol Lett 14(4):20170747
Johann K (1993) DESER-Norm 1993. Normen der Sektion Ertragskunde im Deutschen Verband Forstlicher Forschungsanstalten zur Aufbereitung von waldwachstumskundlichen Dauerversuchen. In: Proceedings of Dt Verb Forstl Forschungsanst, Sek Ertragskd, in Unterreichenbach-Kapfenhardt, pp 96–104
Johnson PS, Shifley SR, Rogers R (2009) The ecology and silviculture of oaks. CABI, Wallingford
Jucker T, Bouriaud O, Avacaritei D, Dănilă I, Duduman G, Valladares F et al (2014) Competition for light and water play contrasting roles in driving diversity–productivity relationships in Iberian forests. J Ecol 102:1202–1213
Jüttner O (1955) Eichenertragstafeln. In: Schober R (ed) Ertragstafeln der wichtigsten Baumarten. JD Sauerländer’s Verlag, Frankfurt am Main, pp 12-25–134-138
Kennel R (1972) Die Buchendurchforstungsversuche in Bayern von 1870 bis 1970. Forstliche Versuchsanstalt München 7:77–80
Knoke T, Ammer C, Stimm B, Mosandl R (2008) Admixing broadleaved to coniferous tree species: a review on yield, ecological stability and economics. Eur J For Res 127:89–101
Kölling C (2007) Klimahüllen für 27 Waldbaumarten. AFZ-DerWald 23:1242–1245
Knigge W, Schulz H (1966) Grundriss der Forstbenutzung. Verlag Paul Parey, Hamburg, Berlin
Krakau UK, Liesebach M, Aronen T, Lelu-Walter M-A, Schneck V (2013) Scots Pine (Pinus sylvestris L.). In: Pâques LE (ed) Forest tree breeding in Europe. Current state-of-the-art and perspectives. Springer, Dordrecht
Leuschner C, Meier IC, Hertel D (2006) On the niche breadth of Fagus sylvatica: Soil nutrient status in 50 Central European beech stands on a broad range of bedrock types. Ann For Sci 63:355–368
Liang J, Crowther TW, Picard N, Wiser S, Zhou M, Alberti G, Schulze ED, McGuire AD, Bozzato F, Pretzsch H, de-Miguel S, Paquette A, Hérault B, Scherer-Lorenzen M, Barrett CB, Glick HB, Hengeveld GM, Nabuurs GJ, Pfautsch S, Viana H, Vibrans AC, Ammer C, Schall P, Verbyla D, Tchebakova N, Fischer M, Watson JV, Chen HYH, Lei X, Schelhaas MJ, Lu H, Gianelle D, Parfenova EI, Salas C, Lee E, Lee B, Kim HS, Bruelheide H, Coomes DA, Piotto D, Sunderland T, Schmid B, Gourlet-Fleury S, Sonké B, Tavani R, Zhu J, Brandl S, Vayreda J, Kitahara F, Searle EB, Neldner VJ, Ngugi MR, Baraloto C, Frizzera L, Balazy R, Oleksyn J, Zawila-Niedźwiecki T, Bouriaud O, Bussotti F, Finér L, Jaroszewicz B, Jucker T, Valladares F, Jagodzinski AM, Peri PL, Gonmadje C, Marthy W, O’Brien T, Martin EH, Marshall AR, Rovero F, Bitariho R, Niklaus PA, Alvarez-Loayza P, Chamuya N, Valencia R, Mortier F, Wortel V, Engone-Obiang NL, Ferreira LV, Odeke DE, Vasquez RM, Lewis SL, Reich PB (2016) Positive biodiversity-productivity relationship predominant in global forests. Science 354(6309):1–12
Lu H, Mohren GMJ, den Ouden J, Goudiaby V, Sterck FJ (2016) Overyielding of temperate mixed forests occurs in evergreen–deciduous but not in deciduous–deciduous species mixtures over time in the Netherlands. For Ecol Manag 376:321–332
Lu H, Condés S, del Río M, Goudiaby V, den Ouden J, Mohren GMJ (2018) Species and soil effects on overyielding of tree species mixtures in the Netherlands. For Ecol Manag 409:105–118
Merlin M, Perot T, Perret S, Korboulewsky N, Vallet P (2015) Effects of stand composition and tree size on resistance and resilience to drought in sessile oak and Scots pine. For Ecol Manag 339:22–33
Metz J, Annighöfer P, Schall P, Zimmermann J, Kahl T, Schulze ED, Ammer C (2016) Site adapted admixed tree species reduce drought susceptibility of mature European beech. Glob Change Biol 22:903–920. https://doi.org/10.1111/gcb.13113
Mölder A, Meyer P, Nagel R-V (2019) Integrative management to sustain biodiversity and ecological continuity in Central European temperate oak (Quercus robur, Q. petraea) forests: an overview. For Ecol Manag 437:324–339
Ngo Bieng MAN, Perot T, De Coligny F, Goreaud F (2013) Spatial pattern of trees influences species productivity in a mature oak–pine mixed forest. Eur J For Res 132(5–6):841–850
Pach M, Sansone D, Ponette Q, Barreiro S, Mason B, Bravo-Oviedo A (2018) Silviculture of mixed forests: a European overview of current practices and challenges. In: Bravo-Oviedo A, Pretzsch H, del Río M (eds) Dynamics, silviculture and management of mixed forests. Springer, Cham, pp 185–253
Perot T, Picard N (2012) Mixture enhances productivity in a two-species 3 forest: evidence from a modelling approach. Ecol Res 27(1):83–94
Petterson H (1955) Die Massenproduktion des Nadelwaldes. Mitt Schwed Forstl Forschungsanst, Stockholm, vol 45 (IB), 391 p
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2017) nlme: Linear and nonlinear mixed effects models. R package version 3.1–141. https://CRAN.R-project.org/package=nlme. Accessed 1 May 2019
Praciak A (2013) The CABI encyclopedia of forest trees. CABI, Wallingford
Preikša Z, Brazaitis G, Marozas V et al (2015) Dead wood quality influences species diversity of rare cryptogams in temperate broadleaved forests. iForest 9:276–285
Pretzsch H (2009) Forest dynamics, growth and yield. Springer, Berlin, Heidelberg
Pretzsch H (2014) Canopy space filling and tree crown morphology in mixed-species stands compared with monocultures. For Ecol Manag 327:251–264
Pretzsch H (2018) Growth and structure in mixed-species stands compared with monocultures: review and perspectives. In: Bravo-Oviedo A, Pretzsch H, del Río M (eds) Dynamics, silviculture and management of mixed forests, managing forest ecosystems, vol 31. Springer, Berlin, pp 131–183
Pretzsch H, Rais A (2016) Wood quality in complex forests versus even-aged monocultures. Review and perspectives. Wood Sci Technol 50:845–880
Pretzsch H, Schütze G (2009) Transgressive overyielding in mixed compared with pure stands of Norway spruce and European beech in Central Europe: evidence on stand level and explanation on individual tree level. Eur J For Res 128(2):183–204
Pretzsch H, Schütze G (2016) Effect of tree species mixing on the size structure, density, and yield of forest stands. Eur J For Res 135:1–22
Pretzsch H, Block J, Dieler J, Dong PH, Kohnle U, Nagel J, Spellmann H, Zingg A (2010) Comparison between the productivity of pure and mixed stands of Norway spruce and European beech along an ecological gradient. Ann For Sci 67(7):712
Pretzsch H, Bielak K, Block J, Bruchwald A, Dieler J, Ehrhart HP, Kohnle U, Nagel J, Spellmann H, Zasada M, Zingg A (2013a) Productivity of mixed versus pure stands of oak (Quercus pretraea (Matt.) Liebl and Quercus robur L.) and European beech (Fagus sylvatica L.) along an ecological gradient. Eur J Forest Res 132(2):263–280
Pretzsch H, Schütze G, Uhl E (2013b) Resistance of European tree species to drought stress in mixed versus pure forests: evidence of stress release by inter-specific facilitation. Plant Biol 15:483–495
Pretzsch H, del Río M, Ammer Ch, Avdagic A, Barbeito I, Bielak K, Brazaitis G, Coll L, Dirnberger G, Drössler L, Fabrika M, Forrester DI, Godvod K, Heym M, Hurt V, Kurylyak V, Löf M, Lombardi F, Matović B, Mohren F, Motta R, den Ouden J, Pach M, Ponette Q, Schütze G, Schweig J, Skrzyszewski J, Sramek V, Sterba H, Stojanović D, Svoboda M, Vanhellemont M, Verheyen K, Wellhausen K, Zlatanov T, Bravo-Oviedo A (2015) Growth and yield of mixed versus pure stands of Scots pine (Pinus sylvestris L.) and European beech (Fagus sylvatica L.) analysed along a productivity gradient through Europe. Eur J For Res 134(5):927–947
Pretzsch H, Forrester DI, Bauhus J (2017) Mixed-species forests. Ecology and management. Springer, Berlin, p 653
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reineke LH (1933) Perfecting a stand-density index for even-aged forests. J Agric Res 46:627–638
Riofrío J, del Río M, Pretzsch H, Bravo F (2017) Changes in structural heterogeneity and stand productivity by mixing scots pine and maritime pine. For Ecol Manag 405:219–228
Roloff A, Bärtels A, Schulz B (2008) Flora der Gehölze. Bestimmung, Eigenschaften und Verwendung, 3rd edn. Ulmer, Stuttgart
Rothe A, Binkley D (2001) Nutritional interactions in mixed species forests: a synthesis. Can J For Res 31(11):1855–1870
Sharma RK, Mason EG, Sorensson CT (2008) Productivity of radiata pine (Pinus radiata D. Don.) clones in monoclonal and clonal mixture plots at age 12 years. For Ecol Manag 255:140–148
Spellmann H, Albert M, Schmidt M, Sutmöller J, Overbeck M (2011) Waldbauliche Anpassungsstrategien für veränderte Klimaverhältnisse. AFZ-Der Wald 11:19–23
Staudhammer CL, Jokela EJ, Martin TA (2009) Competition dynamics in pure- versus mixed- family stands of loblolly and slash pine in the southeastern United States. Can J For Res 39:396–409
Steckel M, Heym M, Wolff B, Reventlow DOJ, Pretzsch H (2019) Transgressive overyielding in mixed compared with monospecific Scots pine (Pinus sylvestris L.) and oak (Quercus robur L., Quercus petraea (Matt.) Liebl.) stands–Productivity gains increase with annual water supply. For Ecol Manag 439:81–96
Toïgo M, Vallet P, Perot T, Bontemps J-D, Piedallu C, Courbaud B (2015) Overyielding in mixed forests decreases with site productivity. J Ecol 103:502–512
Toïgo M, Perot T, Courbaud B, Castagneyrol B, Gégout JC, Longuetaud F, Jactel H, Vallet P (2018) Difference in shade tolerance drives the mixture effect on oak productivity. J Ecol 106(3):1073–1082
Vanhellemont M, Sousa-Silva R, Maes SL et al (2019) Distinct growth responses to drought for oak and beech in temperate mixed forests. Sci Total Environ 650:3017–3026
von Lüpke B, Hauskeller-Bullerjahn K (2004) Beitrag zur Modellierung der Jungwuchsentwicklung am Beispiel von Traubeneichen-Buchen-Mischverjüngungen. Allg Forst-u J-Ztg 175:61–69
Wiedemann E (1943) Kiefern-Ertragstafel für mäßige Durchforstung, starke Durchforstung und Lichtung. In: Wiedemann E (ed) Die Kiefer 1948. Verlag M & H Schaper, Hannover
Williams LJ, Paquette A, Cavender-Bares J, Messier C, Reich PB (2017) Spatial complementarity in tree crowns explains overyielding in species mixtures. Nat Ecol Evol 1:63
Acknowledgements
The authors wish to thank the European Union for funding the project “Mixed species forest management. Lowering risk, increasing resilience (REFORM)” (#2816ERA02S, PCIN2017-026) under the framework of Sumforest ERA-NET. All contributors thank their national funding institutions to establish, measure and analyse data from the triplets. The first author also thanks the Bayerische Staatsforsten (BaySF) for supporting the establishment of the plots and the Bavarian State Ministry for Nutrition, Agriculture, and Forestry for permanent support of the project W 07 “Long-term experimental plots for forest growth and yield research” (#7831-22209-2013). The French site (FR-1) belongs to the OPTMix experimental site (https://optmix.irstea.fr), which is supported annually by Ecofor, Allenvi, and the French national research infrastructure ANAEE-F. Research on the Lithuanian triplets was made possible by the national funding institution Research Council of Lithuania (LMTLT), agreement No. S-SUMFOREST-17-1. Thanks are also due to Ulrich Kern for the graphical artwork. Finally, we thank two anonymous reviewers for their constructive criticism.
Author information
Authors and Affiliations
Contributions
H. Pretzsch initiated the study, led the evaluation and wrote the manuscript. M. del Río initiated the study and reworked the manuscript. M. Steckel, M. Heym and P. Biber contributed to the data management, statistical evaluation and writing. C. Ammer contributed to reviewing and writing the manuscript. All authors provided the data and increment core analyses and improved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Communicated by Thomas Knoke.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Pretzsch, H., Steckel, M., Heym, M. et al. Stand growth and structure of mixed-species and monospecific stands of Scots pine (Pinus sylvestris L.) and oak (Q. robur L., Quercus petraea (Matt.) Liebl.) analysed along a productivity gradient through Europe. Eur J Forest Res 139, 349–367 (2020). https://doi.org/10.1007/s10342-019-01233-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-019-01233-y