Abstract
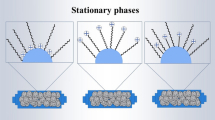
The retention behavior of melamine (MEL), ammeline (AMN), ammelide, and cyanuric acid on various hydrophilic interaction liquid chromatography (HILIC) stationary phases was studied. Using fully optimized electrospray conditions and ammonium formate buffer pH 4/acetonitrile (20:80 v/v) as a mobile phase, precursor and product ions for each compound were identified. For the LC separation study, the effect of the following parameters was investigated: the type of buffer, the effect of the pH of ammonium formate buffer, the dilution percentage of the standard solutions in the vial, the stability of the standard solutions, and the column equilibration time. The retention and separation of melamine and its hydrolysis products on several HILIC columns was also investigated and retention models were proposed. Retention mechanisms were discussed for all the compounds. When the percentage of acetonitrile in the mobile phase was increased, the retention times of ammelide, ammeline and melamine were shifted to higher values, while the retention time of cyanuric acid did not change significantly. The separation of compounds with isobaric transitions (MEL, AMN) was achieved on four HILIC columns, namely TSKgel Amide-80, Luna HILIC, XBridge HILIC, and ZIC-HILIC (at either 10/90 or 15/85 ammonium formate buffer pH 4/ACN).




Similar content being viewed by others
References
Muniz R, Ceballos SG, Rosales-Martinez D, Conzalo-Lumbreras R, Santos-Montes A, Cubedo-Fernandez A, Izquierdo-Hornillos RC (2008) Anal Bioanal Chem 392:523–531
Crossley SJ, Petersen B, Baines J, WHO (2009) (http://www.who.int/foodsafety/fs_management/Melamine_4.pdf). Accessed 02 July 2011)
Commission Regulation (EC) No 1135/2009, Off J. Eur Union 2009, L 311, 3–5
Deng XJ, Guo DH, Zhao SZ, Han L, Sheng YG, Yi XH (2010) J Chromatogr B 878:2839–2844
Tittlemier SA (2010) Food Addit Contam 27:129–145
Heller DN, Nochetto GB (2008) Rapid Commun Mass Spectrom 22:3624–3639
Wu Y-T, Huang C-M, Lin C–C, Ho W-A, Lin L-C, Chiu T-F, Tarng D-C, Lin C-H, Tsai T-H (2009) J Chromatogr A 1216:7595–7601
Zhou J, Zhao J, Xue X, Zhang J, Chen F, Li Y, Wu L, Li C, Mi J (2009) J Chromatogr B 877:4164–4170
Xia J, Zhou N, Zhou C, Chen B, Wu Y, Yao S (2010) J Sep Sci 33:2688–2697
Alpert A (1990) J Chromatogr A 499:177–196
Hemström P, Irgum K (2006) J Sep Sci 29:1784–1821
Jin G, Guo Z, Zhang F, Xue X, Jin Y, Liang X (2008) Talanta 76:522–527
Karatapanis AE, Fiamegos YC, Stalikas CD (2010) Chromatographia 71:751–759
Karatapanis AE, Fiamegos YC, Stalikas CD (2011) J Chromatogr A 1218:2871–2879
Dejagher B, Mangelings D, Heyden YV (2008) J Sep Sci 31:1438–1448
Jandera P (2008) J Sep Sci 31:1421–1437
Jandera P (2011) Anal Chim Acta 692:1–25
Maragou NC, Thomaidis NS, Koupparis MA (2011) J Am Soc Mass Spectrom 22:1826–1838
Ruta J, Rudaz S, McCalley DV, Veuthey J-L, Guillarme D (2010) J Chromatogr A 1217:8230–8240
Ali MS, Ghori M, Rafiuddin S, Khatri AR (2007) J Pharm Biomed Anal 43:158–167
Dai X, Qian X, Gong B, Wei Y (2011) Chromatographia 73:865–870
Nesterenko EP, Nesterenko PN, Paull B (2009) Anal Chim Acta 652:3–21
McCalley DV (2010) J Chromatogr A 1217:3408–3417
Lämmerhofer M, Richter M, Wu J, Nogueira R, Bicker W, Linder W (2008) J Sep Sci 31:2572–2588
Ikegami T, Tomomatsu K, Takubo H, Horie K, Tanaka N (2008) J Chromatogr A 1184:474–503
Chirita R-I, Westa C, Zubrzycki S, Finaru A-L, Elfakir C (2011) J Chromatogr A 1218:5939–5963
Taylor T, Heckendorf A (2011) The theory of hydrophilic interaction chromatography, ChromAcademy (available at: http://www.chromacademy.com/)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kokotou, M.G., Thomaidis, N.S. Behavior and Retention Models of Melamine and Its Hydrolysis Products. Chromatographia 75, 457–467 (2012). https://doi.org/10.1007/s10337-012-2228-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-012-2228-2