Abstract
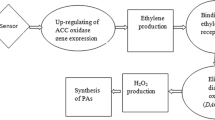
Previous studies have already highlighted the correlation between Sporisorium scitamineum pathogenicity and sugarcane polyamine accumulation. It was shown that high infectivity correlates with an increase in the amount of spermidine, spermine and cadaverine conjugated to phenols in the sensitive cultivars whereas resistant plants mainly produce free putrescine. However, these previous studies did not clarify the role of these polyamides in the disorders caused to the plant. Therefore, the purpose of this research is to clarify the effect of polyamines on the development of smut disease. In this paper, commercial polyamines were firstly assayed on smut teliospores germination. Secondly, effects were correlated to changes in endogenous polyamines after contact with defense sugarcane glycoproteins. Low concentrations of spermidine significantly activated teliospore germination, while putrescine had no activating effect on germination. Interestingly, it was observed that the diamine caused nuclear decondensation and breakage of the teliospore cell wall whereas the treatment of teliospores with spermidine did not induce nuclear decondensation or cell wall breakdown. Moreover, the number of polymerized microtubules increased in the presence of 7.5 mM spermidine but it decreased with putrescine which indicates that polyamines effects on Sporisorium scitamineum teliospore germination could be mediated through microtubules interaction. An increased production of polyamines in smut teliospores has been related to sugarcane resistance to the disease. Teliospores incubation with high molecular mass glycoproteins (HMMG) from the uninoculated resistant variety of sugarcane, Mayari 55-14, caused an increase of the insoluble fraction of putrescine, spermidine and spermine inside the teliospore cells. Moreover, the level of the soluble fraction of spermidine (S fraction) increased inside teliospores and the excess was released to the medium. The HMMG glycoproteins purified from Mayarí 55-14 plants previously inoculated with the pathogen significantly increased the levels of both retained and secreted soluble putrescine and spermidine. Polyamines levels did not increase in teliospores after incubation with HMMG produced by non resistant variety Barbados 42231 which could be related to the incapacity of these plants to defend themselves against smut disease. Thus, a hypothesis about the role of polyamines in sugarcane-smut interaction is explained.








Similar content being viewed by others
Abbreviations
- B 42231:
-
Barbados 42231 cv.
- cv:
-
Cultivar
- DAPI:
-
4,6-diamidino-2-phenylindole
- EGTA:
-
Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid
- HMMG:
-
High molecular mass glycoproteins
- HMPB:
-
High Molarity Pipes Buffer
- MMMG:
-
Mid molecular mass glycoproteins
- MT:
-
Microtubules;
- My 55-14:
-
Mayarí 55-14 cv.
- ODH:
-
Ornithine decarboxylase
- PA:
-
Polyamine
- PBS:
-
Phosphate saline buffer
- PH:
-
Insoluble conjugated polyamine fraction
- PIPES:
-
Piperazine-N,N′-bis(2-ethanesulfonic acid)
- PVP:
-
Polyvinylpyrrolidone
- PUT:
-
Putrescine
- SPD:
-
Spermidine
- SPM:
-
Spermine
- S:
-
Soluble, free polyamine fraction
- SH:
-
Soluble conjugated polyamine fraction
References
Arce L, Ríos A, Valcárcel M (1997) Selective and rapid determination of biogenicamines by capillary zone electrophoresis. Chromatographia 46:170–176
Benevenuto J, Teixeira-Silva NS, Kuramae EE, Croll D, Monteiro-Vitorello CB (2018) Comparative genomics of smut pathogens: insights from orphans and positively selected genes into host specialization. Front Plant Sci 9:660
Brauc S, de Vooght E, Claeys M, Geuns JMC, Höfte M, Angenon G (2012) Overexpression of arginase in Arabidopsis thaliana influences defence responses against Botrytis cinerea. Plant Biol 14:39–45
Carvalho G, Quecine MC, Longatto DP, Peters LP, Almeida JR, Shyton TG, Silva MML, Crestana GS, Crete S, Monteiro-Vitorello CB (2016) Sporisorium scitamineum colonisation of sugarcane genotypes susceptible and resistant to smut revealed by GFP-tagged strains. Annal Appl Biol 169:329–341
Castoldi M, Popov AV (2003) Purification of brain tubulin through two cycles of polymerization-depolymerization in a high-molarity buffer. Protein Expr Purif 32:83–88
Charnay D, Nari J, Noat G (1992) Regulation of plant cell-wall pectin methyl esterase by polyamines—Interactions with the effects of metal ions. Eur J Biochem 205:711–714
Fontaniella B, Márquez A, Rodríguez CW, Piñón D, Solas MT, Vicente C, Legaz ME (2002) A role for sugarcane glycoproteins in the resistance of sugarcane to Ustilago scitaminea. Plant Physiol Biochem 40:881–889
Gill SS, Tuteja N (2010) Polyamines and abiotic stress tolerance in plants. Plant Signal Behav 5:26–33
Grant NJ, Oriol-Audit C, Dickens MJ (1983) Supramolecular forms of actin induced by polyamines; an electron microscopic study. Eur J Cell Biol 30:67–73
Guevara-Olvera L, Xoconostle-Cázares B, Ruiz-Herrera J (1997) Cloning and disruption of the ornithine decarboxylase gene of Ustilago maydis: evidence for a role of polyamines in its dimorphic transition. Microbiology 143:2237–2245
Gupta K, Dey A, Gupta B (2013) Plant polyamines in abiotic stress responses. Acta Physiol Plant 35:2015–2036
Kadotani N, Nakayashiki H, Tosa Y, Mayama S (2003) RNA Silencing in the phytopathogenic fungus Magnaporthe oryzae. Mol Plant Microb Interact 16:769–776
Khatri M, Rajam MV (2007) Targeting polyamines of Aspergillus nidulans by siRNA specific to fungal ornithine decarboxylase gene. Med Mycol 45:211–220
Legaz ME, Pedrosa MM, Martínez M, Vicente C (1995) Soluble glycoproteins from sugarcane juice analyzed by SE-HPLC and fluorescence emission. J Chromatogr 697:329–335
Legaz ME, de Armas R, Piñón D, Vicente C (1998a) Relationships between phenolics-conjugated polyamines and sensitivity of sugarcane to smut (Ustilago scitaminea). J Exp Bot 49:1723–1728
Legaz ME, Pedrosa MM, de Armas R, Rodríguez CW, de los Ríos V, Vicente C (1998b) Separation of soluble glycoproteins from sugarcane juice by capillary electrophoresis. Anal Chim Acta 372:201–208
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Marques JPR, Appezzato-da-Glória B, Pipebring M, Massola N, Monteiro-Vitorello CB, Vieira MLC (2017) Sugarcane smut: shedding light on the development of the whip shaped sorus. Ann Bot 11:815–827
Marques JP, Hoy JW, Appezzato-da-Glória B, Viveros AF, Vieira ML, Baisakh N (2018) Sugarcane cell wall-associated defense responses to infection by Sporisorium scitamineum. Front Plant Sci 9:698
Mechulam A, Chernov KG, Mucher E, Hamon L, Curmi PA, Pastré D (2009) Polyamine sharing between tubulin dimers favours microtubule nucleation and elongation via facilitated diffusion. PLoS Comput Biol 5:e1000255
Millanes AM, Fontaniella B, Legaz ME, Vicente C (2005) Glycoproteins from sugarcane plants regulate cell polarity of Ustilago scitaminea teliospores. J Plant Physiol 162:253–265
Piñón D, de Armas R, Vicente C, Legaz ME (1999) Role of polyamines in the infection of sugarcane buds by Ustilago scitaminea spores. Plant Physiol Biochem 37:57–64
Pohjanpelto P, Virtanen I, Hölttä E (1981) Polyamine starvation causes disappearance of actin filaments and microtubules in polyamine-auxotrophic CHO cells. Nature 293:475–477
Que Y, Xu L, Wu Q, Liu Y, Ling H, Liu Y, Zhang Y, Guo J, Su Y, Chen J, Wang S, Zhang C (2014) Genome sequencing of Sporisorium scitamineum provides insights into the pathogenic mechanisms of sugarcane smut. BMC genom 15:1
Rajam MV, Galston AW (1985) The effects of some polyamine biosynthetic inhibitors on growth and morphology of phytopathogenic fungi. Plant Cell Physiol 26:683–692
Rajam MV, Weinstein LH, Galston AW (1985) Prevention of a plant disease by specific inhibition of fungal polyamine biosynthesis. Proc Nat Acad Sci 82:6874–6878
Sánchez-Elordi E, de los Ríos LM, Vicente C, Legaz ME (2015) Sugarcane arginase competes with the same fungal enzyme as a false quorum signal against smut teliospores. Phytochem Lett 14:115–122
Sánchez-Elordi E, Baluska F, Echevarría C, Vicente C, Legaz ME (2016a) Defence sugarcane glycoproteins disorganize microtubules and prevent nuclear polarization and germination of Sporisorium scitamineum teliospores. J Plant Physiol 200:111–123
Sánchez-Elordi E, Vicente-Manzanares M, Díaz E, Legaz ME, Vicente C (2016b) Plant-pathogen interactions: sugarcane glycoproteins induce chemotaxis of smut teliospores by cyclic contraction and relaxation of the cytoskeleton. South Afr J Bot 105:66–78
Sánchez-Elordi E, Contreras R, de Armas R, Benito MC, Alarcón B, de Oliveira E, del Mazo C, Díaz-Peña EM, Santiago R, Vicente C, Legaz ME (2108) Differential expression of SofDIR16 and SofCAD genes in smut resistant and susceptible sugarcane cultivars in response to Sporisorium scitamineum. J Plant Physiol 226:103–113
Savarin P, Barbet A, Delga S, Joshi V, Hamon L, Lefevre J, Nakib S, de Bandt JP, Moinard C, Curmi PA, Pastré D (2010) A central role for polyamines in microtubule assembly in cells. Biochem J 430:151–159
Takao K, Rickhag M, Hegardt C, Oredsson S, Persson L (2006) Induction of apoptotic cell death by putrescine. Int J Biochem Cell Biol 38:621–628
Trione EJ (1990) Growth and sporulation of Ustilago scitaminea, in vivo and in vitro. Mycol Res 94:489–493
Valdés-Santiago L, Cervantes-Chávez JA, Ruiz-Herrera J (2009) Ustilago maydis spermidine synthase is encoded by a chimeric gene, required for morphogenesis, and indispensable for survival in the host. FEMS Yeast Res 9:923–935
Valdés-Santiago L, Guzmán de Peña D, Ruiz-Herrera J (2010) Life without putrescine: disruption of the gene-encoding polyamine oxidase in Ustilago maydis odc mutants. FEMS Yeast Res 10:928–940
Valdés-Santiago L, Cervantes-Chávez JA, Geraldine León-Ramírez C, Ruiz-Herrera J (2012) Polyamine metabolism in fungi with emphasis on phytopathogenic species. J Amino Acids. https://doi.org/10.1155/2012/837932
Waller JM (1970) Sugarcane smut (Ustilago scitaminea) in Kenya: infection and resistance. Trans Br Mycol Soc 54:405–414
Walters DR (1995) Inhibition of polyamine biosynthesis in fungi. Mycol Res 199:129–139
Walters D (2003) Research review: Resistance to plant pathogens: possible roles for free polyamines and polyamine catabolism. New Phytol 159:109–115
West HM, Walters DR (1989) Effects of polyamine biosynthesis inhibitors on growth of Pyrenophora teres, Gaeumannomyces graminis, Fusarium culmorum and Septoria nodorum in vitro. Mycol Res 92:453–457
Wojtasik W, Kulma A, Namysł K, Preisner M, Szopa J (2015) Polyamine metabolism in flax in response to treatment with pathogenic and non-pathogenic Fusarium strains. Front Plant Sci 6:291
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sánchez-Elordi, E., de los Ríos, L.M., Vicente, C. et al. Polyamines levels increase in smut teliospores after contact with sugarcane glycoproteins as a plant defensive mechanism. J Plant Res 132, 405–417 (2019). https://doi.org/10.1007/s10265-019-01098-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-019-01098-7