Abstract
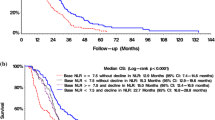
Standard treatment for patients with primary glioblastoma (GBM) includes surgery, radiotherapy, and concomitant and adjuvant temozolomide (TMZ). Recent reports have demonstrated that TMZ-induced myelosuppression correlates with survival in patients with GBM. However, those results were evaluated before the 2016 revision of the World Health Organization classification. This study examined whether myelosuppression during concomitant TMZ phase correlates with prognosis in GBM, IDH-wildtype patients. We examined circulating blood cell counts in 50 patients with GBM, IDH-wildtype who received the standard treatment protocol between August 2005 and November 2015. We assessed relationships between rates of decrease in blood cells (white blood cells (WBC), neutrophils, lymphocytes, red blood cells, and platelets) during the concomitant TMZ phase and overall survival (OS) using univariate and multivariate analyses including other clinicopathological factors (age, sex, Karnofsky Performance Status (KPS), extent of resection, O6-methylguanine-DNA methyltransferase (MGMT) status). Log-rank testing revealed that age, KPS, extent of resection, MGMT status, and decrease rates of WBC, neutrophils, and platelets correlated significantly with OS. On multivariate analysis, age, MGMT status, and decrease rate of neutrophils correlated significantly with OS. Patients with a ≥ 40% decrease in neutrophils showed significantly longer OS than those with < 40% (hazard ratio = 2.815; 95% confidence interval = 1.177–7.038; P = 0.0196). A decrease of ≥ 40% in neutrophils represents a predictor of good prognosis for GBM, IDH-wildtype. Blood cell counts during the concomitant TMZ phase can help predict OS in patients with GBM, IDH-wildtype receiving the standard treatment protocol.



Similar content being viewed by others
References
Akobeng AK (2007) Understanding diagnostic tests 3: receiver operating characteristic curves. Acta Paediatr 96:644–647. https://doi.org/10.1111/j.1651-2227.2006.00178.x
Athanassiou H, Synodinou M, Maragoudakis E, Paraskevaidis M, Verigos C, Misailidou D, Antonadou D, Saris G, Beroukas K, Karageorgis P (2005) Randomized phase II study of temozolomide and radiotherapy compared with radiotherapy alone in newly diagnosed glioblastoma multiforme. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 23:2372–2377. https://doi.org/10.1200/JCO.2005.00.331
Brandes AA, Tosoni A, Cavallo G, Bertorelle R, Gioia V, Franceschi E, Biscuola M, Blatt V, Crino L, Ermani M, GICNO (2006) Temozolomide 3 weeks on and 1 week off as first-line therapy for recurrent glioblastoma: phase II study from gruppo italiano cooperativo di neuro-oncologia (GICNO). Br J Cancer 95:1155–1160. https://doi.org/10.1038/sj.bjc.6603376
Brandes AA, Tosoni A, Cavallo G, Scopece L, Gardiman M, Berti F, Pasetto LM, Ferrarese F, Blatt V, Ermani M (2006) Temozolomide (TMZ) 3 weeks on/1 week off in the treatment of progressive low grade gliomas: a phase II GICNO study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 24:1514
Chen Z, Chen W, Wang J, Zhu M, Zhuang Z (2015) Pretreated baseline neutrophil count and chemotherapy-induced neutropenia may be conveniently available as prognostic biomarkers in advanced gastric cancer. Intern Med J 45:854–859. https://doi.org/10.1111/imj.12786
Donskov F, Michaelson MD, Puzanov I, Davis MP, Bjarnason GA, Motzer RJ, Goldstein D, Lin X, Cohen DP, Wiltshire R, Rini BI (2015) Sunitinib-associated hypertension and neutropenia as efficacy biomarkers in metastatic renal cell carcinoma patients. Br J Cancer 113:1571–1580. https://doi.org/10.1038/bjc.2015.368
Gao B, Klumpen HJ, Gurney H (2008) Dose calculation of anticancer drugs. Expert Opin Drug Metab Toxicol 4:1307–1319. https://doi.org/10.1517/17425255.4.10.1307
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003. https://doi.org/10.1056/NEJMoa043331
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Mellai M, Piazzi A, Caldera V, Monzeglio O, Cassoni P, Valente G, Schiffer D (2011) IDH1 and IDH2 mutations, immunohistochemistry and associations in a series of brain tumors. J Neuro-Oncol 105:345–357. https://doi.org/10.1007/s11060-011-0596-3
Perry JR, Belanger K, Mason WP, Fulton D, Kavan P, Easaw J, Shields C, Kirby S, Macdonald DR, Eisenstat DD, Thiessen B, Forsyth P, Pouliot JF (2010) Phase II trial of continuous dose-intense temozolomide in recurrent malignant glioma: RESCUE study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 28:2051–2057. https://doi.org/10.1200/JCO.2009.26.5520
Quillien V, Lavenu A, Karayan-Tapon L, Carpentier C, Labussiere M, Lesimple T, Chinot O, Wager M, Honnorat J, Saikali S, Fina F, Sanson M, Figarella-Branger D (2012) Comparative assessment of 5 methods (methylation-specific polymerase chain reaction, MethyLight, pyrosequencing, methylation-sensitive high-resolution melting, and immunohistochemistry) to analyze O6-methylguanine-DNA-methyltranferase in a series of 100 glioblastoma patients. Cancer 118:4201–4211. https://doi.org/10.1002/cncr.27392
Saito T, Sugiyama K, Ikawa F, Yamasaki F, Ishifuro M, Takayasu T, Nosaka R, Muragaki Y, Kawamata T, Kurisu K (2016) Permeability surface area product using perfusion CT is a valuable prognostic factor in glioblastomas treated with radiotherapy plus concomitant and adjuvant temozolomide. World neurosurgery. https://doi.org/10.1016/j.wneu.2016.09.072
Sawyer M, Ratain MJ (2001) Body surface area as a determinant of pharmacokinetics and drug dosing. Investig New Drugs 19:171–177
Shah GD, Silver JS, Rosenfeld SS, Gavrilovic IT, Abrey LE, Lassman AB (2007) Myelosuppression in patients benefiting from imatinib with hydroxyurea for recurrent malignant gliomas. J Neuro-Oncol 85:217–222. https://doi.org/10.1007/s11060-007-9408-1
Shen M, Hu P, Donskov F, Wang G, Liu Q, Du J (2014) Tumor-associated neutrophils as a new prognostic factor in cancer: a systematic review and meta-analysis. PLoS One 9:e98259. https://doi.org/10.1371/journal.pone.0098259
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
Tazzyman S, Niaz H, Murdoch C (2013) Neutrophil-mediated tumour angiogenesis: subversion of immune responses to promote tumour growth. Semin Cancer Biol 23:149–158. https://doi.org/10.1016/j.semcancer.2013.02.003
Vaios EJ, Nahed BV, Muzikansky A, Fathi AT, Dietrich J (2016) Bone marrow response as a potential biomarker of outcomes in glioblastoma patients. J Neurosurg:1–7. https://doi.org/10.3171/2016.7.JNS16609
Verhoeff JJ, Lavini C, van Linde ME, Stalpers LJ, Majoie CB, Reijneveld JC, van Furth WR, Richel DJ (2010) Bevacizumab and dose-intense temozolomide in recurrent high-grade glioma. Annals of oncology : official journal of the European Society for Medical Oncology 21:1723–1727. https://doi.org/10.1093/annonc/mdp591
Williams M, Liu ZW, Woolf D, Hargreaves S, Michalarea V, Menashy R, Kooner I, Wilson E (2012) Change in platelet levels during radiotherapy with concurrent and adjuvant temozolomide for the treatment of glioblastoma: a novel prognostic factor for survival. J Cancer Res Clin Oncol 138:1683–1688. https://doi.org/10.1007/s00432-012-1243-x
Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W, Kos I, Batinic-Haberle I, Jones S, Riggins GJ, Friedman H, Friedman A, Reardon D, Herndon J, Kinzler KW, Velculescu VE, Vogelstein B, Bigner DD (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773. https://doi.org/10.1056/NEJMoa0808710
Acknowledgements
We thank Masako Yoshihiro, Department of Neurosurgery, Hiroshima University, Graduate School of Biomedical and Health Science, Hiroshima, Japan, for the immunohistochemical staining.
Funding
This study was funded by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 26462183.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Ethical approval was obtained from the institutional review board of Hiroshima University (No. E-730).
Informed consent
Given the retrospective design, the institutional review board waived the requirement for informed consent. We took the opt-out method. To protect patient privacy, we removed all identifiers from our records upon completion of our analyses.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Saito, T., Sugiyama, K., Hama, S. et al. Prognostic importance of temozolomide-induced neutropenia in glioblastoma, IDH-wildtype patients. Neurosurg Rev 41, 621–628 (2018). https://doi.org/10.1007/s10143-017-0903-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-017-0903-3