Abstract
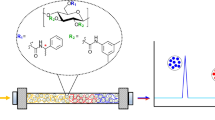
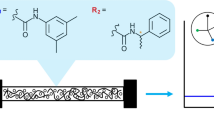
A series of cellulose 3,5-dimethylphenylcarbamates (CDMPCs) with different degrees of substitution (DS) and degrees of polymerization (DP) were homogeneously synthesized in 1-allyl-3-methylimidazolium chloride (AmimCl). Then, the CDMPCs were coated on silica gel and used as chiral stationary phases (CSPs), and their chiral recognition abilities for seven racemates were evaluated by high performance liquid chromatography. The results showed that DS and DP of CDMPCs had a great influence on chiral recognition abilities of the CSPs. The CSPs with the DS ≈ 1 gives a low chiral recognition to most racemates. On the contrast, the CSPs with the DS ≥ 2 exhibited high chiral separation abilities. For example, six racemates could be separated on the CSP with CDMPC of DS ≈ 2 (CSP-2). Especially, for the enantioseparation of 1-(2-naphthyl) ethanol and Tröger’s base, CSP-2 gave the highest separation ability in all of CSPs. On the other hand, when the DP of cellulose was in a range from 39 to 220, the chiral separation abilities of CDMPCs increased as the DP increased. This work demonstrates that the structure of cellulose esters such as DS and DP has important effect on their chiral separation ability, and therefore provides a practical method to design and prepare desirable CSPs for different racemates.
Similar content being viewed by others
References
Kubota, T., Yamamoto, C. and Okamoto, Y., J. Am. Chem. Soc., 2000, 122: 4056
Ikai, T. and Okamoto, Y., Chem. Rev., 2009, 109: 6077
Chen, L.R. “Chiral separation in liquid chromatography” (in Chinese), Science Press, Beijing, 2007, p. 58
Okamoto, Y., in “Chiral recognition in separation methods”, ed. by Berthod, A., Springer, Berlin Heidelberg New York, 2010, p. 33
Grieb, S.J., Matlin, S.A., Belenguer, A.M. and Ritchie, H.J., J. Chromatogr. A, 1995, 697: 271
Yashima, E., Sahavattanapong, P. and Okamoto, Y., Chirality, 1996, 8: 446
Hearon, W.M., Hiatt, G.D. and Fordyce, C.R., J. Am. Chem. Soc., 1943, 65: 2449
Erler, U., Klemm, D. and Nehls, I., Macromol. Rapid Commun., 1992, 13: 195
Erdmenger, T., Haensch, C., Hoogenboom, R. and Schubert, U.S., Macromol. Biosci., 2007, 7: 440
Toga, Y., Hioki, K., Namikoshi, H., and Shibata, T., Cellulose, 2004, 11: 65
Enomoto, N., Furukawa, S., Ogasawara, Y., Akano, H., Kawamura, Y., Yashima, E. and Okamoto, Y., Anal. Chem., 1996, 68: 2798
Swatloski, R.P., Spear, S.K., Holbrey, J.D. and Rogers, R.D., J. Am. Chem. Soc., 2002, 124: 4974
Pinkert, A., Marsh, K.N., Pang, S. and Staiger, M.P., Chem. Rev., 2009, 109: 6712
Wu, J., Zhang, J., Zhang, H., He, J.S., Ren, Q. and Guo, M., Biomacromolecules, 2004, 5: 266
Barthel, S. and Heinze, T., Green Chem., 2006, 8: 301
Cao, Y., Wu, J., Zhang, J., Li, H.Q., Zhang, Y. and He, J.S., Chem. Eng. J., 2009, 147: 13
Zhang, J.M., Wu, J., Cao, Y., Sang, S.M., Zhang, J. and He, J.S., Cellulose, 2009, 16: 299
Chen, J., Zhang, J.M, Feng, Y., He, J.S. and Zhang, J., Chinese J. Polym. Sci., 2014, 32(1): 1
Zhang, H., Wu, J., Zhang, J. and He, J.S., Macromolecules, 2005, 38: 8272
Meiland, M., Liebert, T. and Heinze, T., Macromol. Mater. Eng., 2011, 296: 802
GB 5888-86
Goodlett, V.W., Doughert. Jt and Patton, H.W., J. Polym. Sci., Part A-1: Polym. Chem., 1971, 9: 155
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was financially supported by the National Natural Science Foundation of China (Nos. 51103167, 21174151 and 51425307).
Rights and permissions
About this article
Cite this article
Chen, Ww., Ding, Mc., Zhang, M. et al. Chiral separation abilities of homogeneously synthesized cellulose 3,5-dimethylphenylcarbamates: Influences of degree of substitution and molecular weight. Chin J Polym Sci 33, 1633–1639 (2015). https://doi.org/10.1007/s10118-015-1695-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-015-1695-y