Abstract
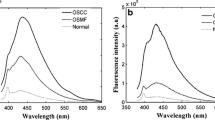
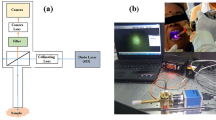
In vivo detection of oral precancer has been carried out by a fluorescence-based, in-house-developed handheld probe on three groups: oral squamous cell carcinoma (OSCC), dysplastic (precancer), and control (normal). Measurements have been performed on a total of 141 patients and volunteers of different age groups. Excitation wavelength of 405 nm was used and fluorescence emission spectra were recorded in the scan range of 450.14 to 763.41 nm at very low incident power (122 μW) from different oral sites buccal mucosa (BM), lateral boarder of tongue (LBT), and dorsal surface of tongue (DST). Spectral profiles are found to vary among the three groups as well as among the different oral sites. Major and minor bands of flavin adenine dinucleotide (FAD) and porphyrins near 500, 634, 676, 689, and 703 nm have been obtained. Porphyrin contribution is found to be more dominant than the FAD in OSCC and dysplastic groups as compared to the control group. A better classification has been observed using the entire spectral range rather than restricting to individual bands, by application of principal component analysis (PCA), Mahalanobis distance model, and receiver operating characteristic analysis (ROC). ROC on Mahalanobis distance differentiates OSCC to normal, dysplastic to normal, and OSCC to dysplastic with sensitivities from 71% to 98%, 92% to 94% and 81% to 93% and specificities 91% to 100%, 86% to 100% and 79% to 97% for oral sites BM, LBT and DST. LBT and DST appear to be more sensitive to dysplasia detection as compared to BM.






Similar content being viewed by others
References
Omar E (2015) Current concepts and future of noninvasive procedures for diagnosing oral squamous cell carcinoma--a systematic review. Head Face Med 11: 6
Varshitha A (2015) Prevalence of oral cancer in India. J Pharm Sci Res 7:845–848
Thomas J, Vineet DA, Rasheena PM, Krishnan JK (2013) Early detection of precancerous and cancerous lesions: an overview. JIAOMR 25:35–39
Yardimci G, Kutlubay Z, Engin B, Tuzun Y (2014) Precancer lesions of oral mucosa. World J Clin Cases 2:866–872
Dikshit R, Gupta PC, Ramasundarahettige C, Gajalakshmi V, Aleksandrowicz L, Badwe R, Kumar R, Roy S, Suraweera W, Bray F, Mallath M, Singh PK, Sinha DN, Shet AS, Gelband H, Jha P, Million Death Study Collaborators (2012) Cancer mortality in India: a nationally representative survey. Lancet 379:1807–1816
Scully C, Bagan JV, Hopper C, Epstein JB (2008) Oral cancer: current and future diagnostic techniques. Am J Dent 21:199–209
Koenig K, Schneckenburger H (1994) Laser-induced autofluorescence for medical diagnosis. J Fluoresc 4:17–40
de Oliveira Silva FR, Bellini MH, Tristão VR, Schor N, Vieira ND Jr, Courrol LC (2010) Intrinsic fluorescence of protoporphyrin IX from blood samples can yield information on the growth of prostate tumours. J Fluoresc 20:1159–1165
Inaguma M, Hashimoto K (1999) Porphyrin-like fluorescence in oral cancer: in vivo fluorescence spectral characterization of lesions by use of a near-ultraviolet excited autofluorescence diagnosis system and separation of fluorescent extracts by capillary electrophoresis. Cancer 86:2201–2211
Alfano R, Tang G, Pradhan A, Lam W, Choy D, Opher E (1987) Fluorescence spectra from cancerous and normal human breast and lung tissues. IEEE J Quantum Electron 23:1806–1811
Alfano R, Yang Y (2003) Stokes shift emission spectroscopy of human tissue and key biomolecules. IEEE J Sel Top Quantum Electron 9:148–153
Ramanujam N, Mitchell MF, Mahadevan-Jansen A, Thomson SL, Staerkel G, Malpica A, Wright T, Atkinson N, Richardas-Kortum R (1996) Cervical precancer detection using a multivariate statistical algorithm based on laser-induced fluorescence spectra at multiple excitation wavelengths. Photochem Photobiol 64:720–735
de Veld DC, Bakker Schut TC, Skurichina M, Witjes MJ, Van der Wal JE, Roodenburg JL, Sterenborg HJ (2005) Autofluorescence and Raman microspectroscopy of tissue sections of oral lesions. Lasers Med Sci 19:203–209
Hillemanns P, Reiff J, Stepp H, Soergel P (2007) Lymph node metastasis detection of ovarian cancer by porphyrin fluorescence photodetection: case report. Lasers Med Sci 22:131–135
Devi S, Panigrahi PK, Pradhan A (2014) Detecting cervical cancer progression through extracted intrinsic fluorescence and principal component analysis. J Biomed Opt 19:127003
Bergholt MS, Zheng W, Lin K, Ho KY, Teh M, Yeoh KG, So JB, Huang Z (2011) Combining near-infrared-excited autofluorescence and Raman spectroscopy improves in vivo diagnosis of gastric cancer. Biosens Bioelectron 26:4104–4110
Tromberg BJ, Shah N, Lanning R, Cerussi A, Espinoza J, Pham T, Svaasand L, Butler J (2000) Non-invasive in vivo characterization of breast tumours using photo migration spectroscopy. Neoplasia 2:26–40
Farwell DG, Meier JD, Park J, Sun Y, Coffman H, Poirier B, Phipps J, Tinling S, Enepekides DJ, Marcu L (2010) Time- resolved fluorescence spectroscopy as a diagnostic technique of oral carcinoma. Arch Otolaryngol Head Neck Surg 136:126–133
Amelink A, Sterenborg HJ, Roodenburg JL, Witjes MJ (2011) Non-invasive measurement of the microvascular properties of non-dysplastic and dysplastic oral leukoplakias by use of optical spectroscopy. Oral Oncol 47:1165–1170
Singh SP, Deshmukh A, Chaturvedi P, Murali KC (2012) In vivo Raman spectroscopic identification of premalignant lesions in oral buccal mucosa. J Biomed Opt 17:105002
Pichardo-Molina JL, Frausto-Reyes C, Barbosa-García O, Huerta-Franco R, González-Trujillo JL, Ramírez-Alvarado CA, Gutiérrez-Juárez G, Medina-Gutiérrez C (2007) Raman spectroscopy and multivariate analysis of serum samples from breast cancer patients. Lasers Med Sci 22:229–236
Yuvaraj M, Aruna P, Koteeswaran D, Tamilkumar P, Ganesh S (2015) Rapid fluorescence spectroscopic characterization of salivary DNA of normal subjects and OSCC patients using ethidium bromide. J Fluoresc 25:79–85
Kumar P, Singh A, Kumar Kanaujia S, Pradhan A (2018) Human saliva for oral precancer detection: a comparison of fluorescence and Stoke shift spectroscopy. J Fluoresc 28:419–426
Gillenwater A, Jacob R, Ganeshappa R, Kemp B, El-Naggar AK, Palmer JL, Clayman G, Mitchell MF, Richarda_Kortum R (1998) Noninvasive diagnosis of oral neoplasia based on fluorescence spectroscopy and native tissue autofluorescence. Arch Otolaryngol Head Neck Surg 124:1251–1258
Majumder SK, Mahanty SK, Ghosh N, Gupta PK, Jain DK, Khan F (2000) A pilot study on the use of autofluorescence for diagnosis of the cancer of human oral cavity. Curr Sci 79:1089–1094
Lane PM, Gilhuly T, Whitehead P, Zeng H, Poh CF, Ng S, Williams PM, Zhang L, Rosin MP, MacAulay CE (2006) Simple device for the direct visualization of oral-cavity tissue fluorescence. J Biomed Opt 11:024006
De Veld DC, Sterenborg HJ, Roodenburg JL, Witjes MJ (2004) Effect of individual characteristics on healthy oral mucosa autofluorescence spectra. Oral Oncol 40:815–823
Jayanthi JL, Subhash N, Stephen M, Philip EK, Beena VT (2011) Comparative evaluation of the diagnostic performance of autofluorescence and diffuse reflectance in oral cancer detection: a clinical study. J Biophotonics 4:696–706
Nazeer SS, Asish R, Venugopal C, Anita B, Gupta AK, Jayasree RS (2014) Noninvasive assessment of the risk of tobacco abuse in oral mucosa using fluorescence spectroscopy: a clinical approach. J Biomed Opt 19:057013
Abdi H, Williams LJ (2010) Principal component analysis. Wiley Interdiscp Rev: Comput Stat 2:433–459
De Maesschalac R, Jouan-Rimbaud D, Massart DL (2000) The Mahalanobis distance. Chemom Intell Lab Syst 50:1–18
Brereton RG (2015) The Mahalanobis distance and its relationship to principal component scores. J Chemom 29:143–145
Fawcett T (2006) An introduction to ROC analysis. Pattern Recogn Lett 27:861–874
Akobeng AK (2007) Understanding diagnostic test 3: receiver operating characteristic curves. Acta Paediatr 90:644–647
Acknowledgements
We would like to acknowledge IMPRINT India, MHRD government, for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Role of funding source
Facilities of Biophotonics lab at IIT Kanpur has been utilized in the study.
Ethics approval
The study was approved by the Ethics Committee of GSVM Medical College Kanpur and IIT Kanpur and performed in accordance with the Declaration of Helsinki.
Informed consent
Informed consent form was taken from all participating patients and volunteers.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumar, P., Kanaujia, S.K., Singh, A. et al. In vivo detection of oral precancer using a fluorescence-based, in-house-fabricated device: a Mahalanobis distance-based classification. Lasers Med Sci 34, 1243–1251 (2019). https://doi.org/10.1007/s10103-019-02720-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-019-02720-9