Abstract
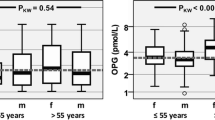
Abnormalities in blood inflammatory markers have been associated with clinical manifestations and the pathogenesis of the fibromyalgia syndrome (FMS); a relationship between inherited alpha1-antitrypsin deficiency (AATD) and FMS has also been recently raised. In this study, plasma levels of inflammatory markers in FMS patients with and without AATD have been investigated. Blood samples from 138 age-matched females (79 FMS) and 59 general population (GP), with normal MM [n = 82 (59.4%)] and with MS, MZ, SZ, and ZZ AATD genotypes [n = 56 (40.6%)], were analyzed by ELISA for monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor-alpha (TNFα), soluble TNFα receptors I and II, interleukin-8, and vascular endothelial growth factor (VEGF). Plasma levels of MCP-1, VEGF, and TNFα were significantly lower in FMS and GP subjects with AATD compared with those with normal MM-AAT genotypes. Moreover, plasma levels of MCP-1, VEGF, and TNFα were lower in AATD subjects with FMS than in those without FMS (P = 0.000, 0.000, and 0.046, respectively). No statistical differences were found for the other substances measured. Furthermore, a logistic regression model based on plasma MCP-1 cutoff value of ≤130 pg/ml allowed us to discriminate between FMS and GP subjects with a sensitivity of about 93% and a specificity of 79%. Low plasma levels of MCP-1, VEGF, and TNFα are related to AATD, although more markedly in FMS patients. Thus, hypotheses considering FMS as an inflammatory condition related to high plasma levels of inflammatory biomarkers cannot be supported.

Similar content being viewed by others
References
Wolfe F, Smythe HA, Yunus MB, Bennett RM, Bombardier C, Goldenberg DL, Tugwell P, Campbell SM, Abeles M, Clark P, Fam AG, Farber SJ, Fiechtner JJ, Franklin CM, Gatter RA, Hamaty D, Lessard J, Lichtbroun AS, Masi AT, McCain GA, Reynolds WJ, Romano TJ, Russell IJ, Sheon RP, The American College of Rheumatology (1990) Criteria for the classification of fibromyalgia. Arthritis Rheum 33:160–172
Wallace DJ, Linker-Israeli M, Hallegua D, Silverman S, Silver D, Weisman MH (2001) Cytokines play an aetiopathogenetic role in fibromyalgia: a hypothesis and pilot study. Rheumathology (Oxford) 40(7):743–749
Staud R, Pierce DD, Robinson ME, Mauderli AP, Vierck CJ (2004) Maintenance of windup of second pain requires less frequent stimulation in fibromyalgia patients compared to normal controls. Pain 110(3):689–696
Maes M, Libbrecht I, Van Hunsel F, Lin AH, De Clerck L, Stevens W, Kenis G, de Jongh R, Bosmans E, Neels H (1999) The immune-inflammatory pathophysiology of fibromyalgia: increased serum soluble gp130, the common signal transducer protein of various neurotrophic cytokines. Psychoneuroendocrinology 24(4):371–383
Gur A, Karakoc M, Nas K, Remzi C, Denli A, Sarac J (2002) Cytokines and depression in cases with fibromyalgia. J Rheumatol 29(2):358–361
Hein G, Franke S (2002) Are advanced glycation end-product-modified proteins of pathogenetic importance in fibromyalgia? Rheumatology (Oxford) 41(10):1163–1167
Pache M, Ochs J, Genth E, Mierau R, Kube T, Flammer J (2003) Increased plasma endothelin-1 levels in fibromyalgia syndrome. Rheumathology 42:493–494
Bagis S, Tamer L, Sahin G, Sahin G, Bilgin R, Guler H, Ercan B, Erdogan C (2003) Free radicals and antioxidants in primary fibromyalgia: an oxidative stress disorder? Rheumatol Int 28:188–190
Ozgocmen S, Ozyurt H, Sogut S, Akyol O, Ardicoglu O, Yildizhan H (2006) Antioxidant status, lipid peroxidation and nitric oxide in fibromyalgia: etiologic and therapeutic concerns. Rheumatol Int 26(7):598–603
Uceyler N, Valenza R, Stock M, Schedel R, Sprotte G, Sommer C (2006) Reduced levels of antiinflammatory cytokines in patients with chronic widespread pain. Arthritis Rheum 54(8):2656–2664
Bazzichi L, Rossi A, Massimetti G, Giannaccini G, Giuliano T, De Feo F, Ciapparelli A, Dell'Osso L, Bombardieri S (2007) Cytokine patterns in fibromyalgia and their correlation with clinical manifestations. Clin Exp Rheumatol 25(2):225–230
Altindag O, Gur A, Calgan N, Soran N, Celik H, Selek S (2007) Paraoxonase and arylesterase activities in fibromyalgia. Redox Rep 12(3):134–138
Wang H, Moser M, Schiltenwolf M, Buchner M (2008) Circulating cytokine levels compared to pain in patients with fibromyalgia—a prospective longitudinal study over 6 months. J Rheumatol 35:7–11
Zhang Z, Cherryholmes G, Mao A, Marek C, Longmate J, Kalos M, St Amand RP, Shively JE (2008) High plasma levels of MCP-1 and eotaxin provide evidence for an immunological basis of fibromyalgia. Exp Biol Med 233(9):1171–1180
Chung CP, Titova D, Oeser A, Randels M, Avalos I, Milne GL, Morrow JD, Stein CM (2008) Oxidative stress in fibromyalgia and its relationship to symptoms. Clin Rheumatol 28(4):435–438
Cordero MD, Moreno-Fernández AM, Demiguel M, Bonal P, Campa F, Jiménez-Jiménez LM, Ruiz-Losada A, Sánchez-Domínguez B, Sánchez Alcázar JA, Salviati L, Navas P (2008) Coenzyme Q10 distribution in blood is altered in patients with Fibromyalgia. Clin Biochem 42(7–8):732–735
Ablin J, Neumann L, Buskila D (2008) Pathogenesis of fibromyalgia—a review. Joint Bone Spine 75(3):273–279
Blanco I, Canto H, de Serres FJ, Bustillo EF, Rodríguez MC (2004) Alpha-1-antitrypsin replacement therapy efficiently controls fibromyalgia symptoms in two PI ZZ alpha-1-antitrypsin deficiency patients. J Rheumatol 31(10):2082–2085
Blanco I, Arbesú D, Al Kassam D, De Serres F, Fernández-Bustillo E, Rodríguez Menéndez C (2006) Alpha1-antitrypsin polymorphism in fibromyalgia patients from the Asturias Province in Northern Spain: a significantly higher prevalence of the PI*Z deficiency allele in patients than in the general population. J Musculoske Pain 14(3):5–12
Blanco I, de Serres FJ, Janciauskiene S, Arbesú D, Bustillo EF, Cárcaba V, Nita I, Astudillo A (2007) Estimates of the prevalence and number of fibromyalgia syndrome patients and their alpha-1 antitrypsin phenotypic distribution in ten countries. J Musculoske Pain 15(4):9–23
Blanco I, Astudillo A, Domínguez F, Janciauskiene S, Cárcaba V, Gallo C, Canto H, de Serres F, Fernández-Bustillo E (2009) Intravenous Infusions of Purified Alpha 1-Antitrypsin Effectively Controls Symptoms and Reverts Muscle Biopsy Changes in an MZ-Alpha-1 Antitripsyn Deficiency and Fibromyalgia Syndrome Patient. J Musculoske Pain (in press)
Russell IJ (2006) Alpha1-antitrypsin deficiency in the fibromyalgia syndrome. J Musculoske Pain 14(3):1–3
Russell IJ (2007) Pathogenesis of the fibromyalgia syndrome. J Musculoske Pain 15(4):5–8
Blanco I, de Serres FJ, Fernández-Bustillo E et al (2005) Alpha 1-antitrypsin and fibromyalgia: new data in favour of the inflammatory hypothesis of fibromyalgia. Med Hypotheses 64(4):759–769
American Thoracic Society (2003) European Respiratory Society Statement: standards for the diagnosis and management of individuals with alpha1-antitrypsin deficiency. Am J Respir Crit Care Med 168:818–900
Janciauskiene S, Stevens T, Blanco I (2007) New insights into the biology of alpha1-antitrypsin and its role in chronic obstructive pulmonary disease. Curr Respir Med Rev 3:147–158
Cordelier P, Strayer DS (2003) Mechanisms of alpha1-antitrypsin inhibition of cellular serine proteases and HIV-1 protease that are essential for HIV-1 morphogenesis. Biochim Biophys Acta 1638(3):197–207
Shapiro L, Pott GB, Ralston AH (2001) Alpha-1-antitrypsin inhibits human immunodeficiency virus type 1. FASEB J 15(1):115–122
Chan ED, Kaminska AM, Gill W, Chmura K, Feldman NE, Bai X, Floyd CM, Fulton KE, Huitt GA, Strand MJ, Iseman MD, Shapiro L (2007) Alpha-1-antitrypsin (AAT) anomalies are associated with lung disease due to rapidly growing mycobacteria and AAT inhibits Mycobacterium abscessus infection of macrophages. Scand J Infect Dis 39(8):690–696
Knappstein S, Ide T, Schmidt MA, Heusipp G (2004) Alpha 1-antitrypsin binds to and interferes with functionality of EspB from atypical and typical enteropathogenic Escherichia coli strains. Infect Immun 72(8):4344–4350
Cantin AM, Woods DE (1999) Aerosolized prolastin suppresses bacterial proliferation in a model of chronic Pseudomonas aeruginosa lung infection. Am J Respir Crit Care Med 160(4):1130–1135
Lushbaugh WB, Kairalla AB, Hofbauer AF, Arnaud P, Cantey JR, Pittman FE (1981) Inhibition of Entamoeba histolytica cytotoxin by alpha 1 antiprotease and alpha 2 macroglobulin. Am J Trop Med Hyg 30(3):575–585
Forney JR, Yang S, Healey MC (1997) Synergistic anticryptosporidial potential of the combination alpha-1-antitrypsin and paromomycin. Antimicrob Agents Chemother 41(9):2006–2008
De Serres FJ, Blanco I, Fernández-Bustillo E (2007) PI S and PI Z alpha-1 antitrypsin deficiency worldwide. A review of existing genetic epidemiological data. Monaldi Arch Chest Dis 67(4):184–208
Gooptu B, Lomas DA (2009) Conformational pathology of the serpins: themes, variations, and therapeutic strategies. Annu Rev Biochem 78:147–176
Blanco I, Fernández E, Rodríguez MC, Fernández A (1999) Allelic frequency of the gene of alpha-1-antitrypsin in the general population in a county in Asturias. Med Clin (Barc) 113(10):366–370
Blanco I, Fernández E (2001) Alpha1-antitrypsin Pi phenotypes S and Z in Spain: an analysis of the published surveys. Respir Med 95(2):109–114
Aldonyte R, Eriksson S, Piitulainen E, Wallmark A, Janciauskiene S (2004) Analysis of systemic biomarkers in COPD patients. COPD 1(2):155–164
Amel Kashipaz MR, Swinden D, Todd I, Powell RJ (2003) Normal production of inflammatory cytokines in chronic fatigue and fibromyalgia syndromes determined by intracellular cytokine staining in short-term cultured blood mononuclear cells. Clin Exp Immunol 132(2):360–365
Kaufmann I, Eisner C, Richter P, Huge V, Beyer A, Chouker A, Schelling G, Thiel M (2007) Lymphocyte subsets and the role of TH1/TH2 balance in stressed chronic pain patients. Neuroimmunomodulation 14(5):272–280
Kaufmann I, Schelling G, Eisner C, Richter HP, Beyer A, Krauseneck T, Chouker A, Thiel M (2008) Decrease in adhesion molecules on polymorphonuclear leukocytes of patients with fibromyalgia. Rheumatol Int 29:1109–1111
Aldonyte R, Jansson L, Piitulainen E, Janciauskiene S (2003) Circulating monocytes from healthy individuals and COPD patients. Respir Res 4:11
Zisman DA, Kunkel SL, Strieter RM, Tsai WC, Bucknell K, Wilkowski J, Standiford TJ (1997) MCP-1 protects mice in lethal endotoxemia. J Clin Invest 99(12):2832–2836
Gomes RN, Figueiredo RT, Bozza FA, Pacheco P, Amâncio RT, Laranjeira AP, Castro-Faria-Neto HC, Bozza PT, Bozza MT (2006) Increased susceptibility to septic and endotoxic shock in monocyte chemoattractant protein 1/cc chemokine ligand 2-deficient mice correlates with reduced interleukin 10 and enhanced macrophage migration inhibitory factor production. Shock 26(5):457–463
Depaolo RW, Lathan R, Rollins BJ, Karpus WJ (2005) The chemokine CCL2 is required for control of murine gastric Salmonella enterica infection. Infect Immun 73(10):6514–6522
Speyer CL, Speyer CL, Gao H, Rancilio NJ, Neff TA, Huffnagle GB, Sarma JV, Ward PA (2004) Novel chemokine responsiveness and mobilization of neutrophils during sepsis. Am J Pathol 165(6):2187–2196
Ramnath RD, Ng SW, Guglielmotti A, Bhatia M (2008) Role of MCP-1 in endotoxemia and sepsis. Int Immunopharmacol 8(6):810–818
Kalehua AN, Nagel JE, Whelchel LM, Gides JJ, Pyle RS, Smith RJ, Kusiak JW, Taub DD (2004) Monocyte chemoattractant protein-1 and macrophage inflammatory protein-2 are involved in both excitotoxin-induced neurodegeneration and regeneration. Exp Cell Res 297(1):197–211
Lund E, Kendall SA, Janerot-Sjoberg B, Bengtson A (2003) Muscle metabolism in fibromyalgia studied by P-31 magnetic resonance spectroscopy during aerobic and anaerobic exercise. Scand J Rheumatol 32:138–145
Jeschonneck M, Graohman G, Hein H, Sprott H (2000) Abnormal microcirculation and temperature in skin above tender points in patients with fibromyalgia. Rheumatology (Oxford) 39(8):917–921
Kasahara Y, Tuder RM, Cool CD, Lynch DA, Flores SC, Voelkel NF (2001) Endothelial cell death and decreased expression of vascular endothelial growth factor and vascular endothelial growth factor receptor 2 in emphysema. Am J Respir Crit Care Med 163(3 Pt 1):737–744
Richter AG, Maughan EO, Perkins GD, Nathani N, Thickett DR (2005) VEGF levels in pulmonary fibrosis. Thorax 60(2):171
Gavin TP, Stallings HW 3rd, Zwetsloot KA, Westerkamp LM, Ryan NA, Moore RA, Pofahl WE, Hickner RC (2005) Lower capillary density but no difference in VEGF expression in obese vs. lean young skeletal muscle in humans. J Appl Physiol 98(1):315–321
Croley AN, Zwetsloot KA, Westerkamp LM, Ryan NA, Pendergast AM, Hickner RC, Pofahl WE, Gavin TP (2005) Lower capillarization, VEGF protein, and VEGF mRNA response to acute exercise in the vastus lateralis muscle of aged vs. young women. J Appl Physiol 99(5):1872–1879
Togo F, Natelson BH, Adler GH, Ottenweller JE, Goldenberg DL, Struzik ZR, Yamamoto Y (2008) Plasma cytokine fluctuations over time in healthy controls and patients with fibromyalgia. Exp Biol Med (Maywood) 234:232–240
Silva RA, Gomes MS, Appelberg R (2000) Minor role played by type I tumour necrosis factor receptor in the control of Mycobacterium avium proliferation in infected mice. Immunology 99:203–207
Campbell IK, O’Donnell K, Lawlor KE, Ian P (2001) Wicks severe inflammatory arthritis and lymphadenopathy in the absence of TNF. J Clin Invest 107(12):1519–1527
Acknowledgments
The authors acknowledge the expert editorial assistance of Ms. Jimena Blanco Fueyo (MA University of Geneva, CH). The authors are also indebted to Ms. Sue Edelstein from the NIEHS Arts and Photography for displaying figures to summarize the laboratory findings.
Grant supporters
This study has been endorsed by the Spanish National Health Institute Carlos III, the Biohealth Research Office [OIB] of the Principado de Asturias, Spain (IB and VC), and by the Crafoords and Lundström Foundations (SJ).
Disclosures
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blanco, I., Janciauskiene, S., Nita, I. et al. Low plasma levels of monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor-alpha (TNFα), and vascular endothelial growth factor (VEGF) in patients with alpha1-antitrypsin deficiency-related fibromyalgia. Clin Rheumatol 29, 189–197 (2010). https://doi.org/10.1007/s10067-009-1318-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-009-1318-5