Abstract
Nitrogen availability in the Arctic strongly influences plant productivity and distribution, and in permafrost systems with patterned ground, ecosystem carbon and nutrient cycling can vary substantially over short distances. Improved understanding of fine-scale spatial and temporal variation in soil N availability is needed to better predict tundra responses to a warming climate. We quantified plant-available inorganic nitrogen at multiple soil depths in 12 microhabitats associated with a gradient from low-center ice-wedge polygons to high-center polygons in coastal tundra at Utqiaġvik (formerly Barrow), Alaska. We measured vegetation composition, biomass, N content, and rooting depth distribution, as well as soil temperature, moisture, pH, and thaw depth to determine relationships between the spatial and temporal variability in N availability and environmental and vegetation drivers. Soil moisture varied across the microhabitats of the polygonal terrain and was the most important variable linked to distribution of both ammonium and nitrate, with ammonium predominating in wetter areas and nitrate predominating in drier areas. Total inorganic N availability increased as the soil in the active layer thawed, but the newly available N near the permafrost boundary late in the season was apparently not available to roots and did not contribute to plant N content. Nitrate in the drier sites also was not associated with plant N content, raising the possibility of N losses from this N-limited ecosystem. The strong relationship between soil moisture, inorganic N availability, and plant N content implies that understanding hydrological changes that may occur in a warming climate is key to determining nutrient cycling responses in complex polygonal tundra landscapes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In arctic tundra, plant production is limited by low soil nutrient availability resulting from slow mineralization rates in cold, acidic, and often anoxic soils with low organic matter quality (Shaver and Chapin 1995; Jonasson and Shaver 1999; van Wijk and others 2004).
An improved understanding of the spatial and temporal variability and controls on soil nutrient availability is needed to predict how a rapidly warming arctic climate will alter ecosystem structure and processes, such as plant community composition (for example, Chapin and others 1995; Jonasson and others 1999; Graglia and others 2001; Zamin and others 2014), and thus carbon balance and albedo (Hobbie and others 2002; Sturm and others 2005; Weintraub and Schimel 2005a; Loranty and others 2011). Overall, warming is anticipated to increase nutrient availability in arctic soils (Rustad and others 2001; Schmidt and others 2002; Biasi and others 2008; Lamb and others 2011), both through increasing mineralization rates in surface soils and through release of previously unavailable N from near-surface permafrost soils as a result of increased thaw depth (Keuper and others 2012). However, in arctic landscapes underlain by permafrost, determining trajectories of ecosystem change in response to global warming is a major challenge, as freeze–thaw processes frequently result in the formation of patterned-ground features, such as polygons, circles, nets, steps, and stripes (Washburn 1979). These fine-scale features result in spatial heterogeneity in abiotic conditions and plant communities, and thus significant spatial variation in ecosystem processes (Webber 1978; Bockheim and others 1999; Walker and others 2008; Zona and others 2011; Kelley and others 2012).
The mosaic of physical and biotic conditions in patterned-ground landscapes provides an opportunity to explore relationships between the structure and function of tundra plant communities and environmental factors (for example, water, nutrients) that may change in response to a warming climate. Across patterned-ground features, significant differences may exist in both the form of plant-available nutrients and in their spatial and temporal dynamics. Nitrogen (N) is often the most limiting nutrient in arctic ecosystems, especially in wetter sites, though co-limitation with phosphorus (P) is also widespread (Shaver and Chapin 1980; Zamin and Grogan 2012). Differences in forms of available N (for example, nitrate, ammonium, or organic N) are an important determinant of species distribution (McKane and others 2002), and thus an influence on vegetation community nutrient content and chemistry (Aerts and Chapin 2000; Welker and others 2005), and also may determine the potential for N losses from ecosystems through leaching (Harms and Jones 2012).
Arctic ecosystems exhibit strong seasonal variability in plant nutrient availability (Weintraub and Schimel 2005b), so that relatively short periods may be critical in determining total plant nutrient uptake at the annual scale (compare Sullivan and Welker 2005), and the peak in nutrient availability may not correspond to when plants have the capacity to acquire nutrients (Koven and others 2015). Further, tundra underlain by permafrost has the unique feature that as the thaw depth increases throughout the growing season, the volume of soil available to plant roots increases. Across contrasting microtopographic positions, maximum thaw depth, soil moisture, soil temperatures, and soil type (Brown 1967; Webber 1978), as well as the vertical distributions of soil nutrients and nutrient cycling (Wild and others 2015), are known to vary. Our objective was to investigate how these factors influence time-integrated measures of nutrient availability and their interaction with plant productivity.
We investigated the nature and controls on fine-scale spatial and temporal variability in inorganic soil N availability in multiple microhabitats in polygonal tundra at the Next Generation Ecosystem Experiment (NGEE) Arctic site in the coastal tundra landscape of Barrow Environmental Observatory (BEO), Alaska. We hypothesized that plant-available inorganic N would vary across a gradient from low-center ice-wedge polygons to dry, high-center polygons, and that the increase in N available for plant acquisition in deeper soil as the thaw depth increases throughout the growing season would be important to quantify to fully understand the soil N availability. The variation of inorganic nitrogen across the landscape and with soil depth has implications for the responses of this ecosystem to increased thaw depth projected to occur with climatic warming. Our measurement objectives were to address two questions focusing on spatial (in 2012) and temporal (in 2013) variability:
-
1.
How do the form and availability of inorganic N and the relationships between plant-available N and vegetation, environmental, and soil variables vary in polygonal microhabitats across a gradient of polygonal degradation?
-
2.
How do plants respond to seasonal patterns of N availability associated with increasing thaw depth in contrasting types of polygons at two end points of the soil moisture spectrum?
Methods
Field Site
We carried out this study at the Next Generation Ecosystem Experiment (NGEE)-Arctic site on the Barrow Environmental Observatory (BEO), 6 km east of the coastal village of Utqiaġvik (formerly Barrow) on the North Slope of Alaska (71°17′N, 156°47′W). This area is underlain by continuous, ice-rich permafrost to greater than 400 m and has an active-layer thickness less than 1 m (Bockheim and others 1999). The landscape of the BEO is a mosaic of thaw lake basins, drained thaw lake basins, and interstitial or matrix tundra areas (Hinkel and others 2003), with approximately 65% of the terrestrial surface covered by polygonal ground (Brown 1967). Soils are classified as Gelisols and comprise an organic-rich surface horizon overlying mineral material. Over the period 1981–2010, annual mean temperature at Barrow was − 11.2 °C, July mean temperature was + 4.9 °C, and January mean temperature was − 25.2 °C. Mean annual precipitation was 115 mm, with 77% occurring as rainfall between June and October (NOAA 2018).
The NGEE-Arctic site comprises a 0.5 × 0.5 km area of polygonal tundra immediately west of a vegetated thaw lake basin (Heikoop and others 2015; Langford and others 2016). Within this site, we selected four dominant polygon types indicating differing stages of permafrost degradation (Lara and others 2015) to investigate spatial variability in plant-available inorganic N. These comprised (i) low-centered polygons with low-lying rims and limited trough development, (ii) low-centered polygons with raised rims, (iii) transitional polygons with flat centers, and (iv) high-centered polygons with sloping rims. Within four polygons of each of these types, we established 1 × 1 m permanent plots in the center (low pond, flat or high), rim (raised or sloping) and trough microhabitats, making a total of 48 plots. Photographs of the plots are available in the NGEE data archive (Sloan and others 2014a).
Plant Community Nitrogen and Phosphorus Content
In late July to early August, 2012, when aboveground biomass was at its peak, we estimated fractional cover of all vascular and non-vascular species within each 1 × 1 m plot and then harvested aboveground biomass in an adjacent 0.25 × 0.25 m plot. We separated plant material by species and plant part, dried it at 70°C for 2–3 days, and weighed it. We determined the N concentration of ground plant material using a Model 4010 Elemental Combustion System (Costech Analytical Technologies, Valencia, CA, USA) and P concentrations in Kjeldahl digests on a QuikChem 8500 analyzer (Lachat Instruments, Loveland, CO, USA) using Lachat QuikChem Method 13-115-01-1-B. We calculated N and P contents of each species by multiplying their concentrations by biomass for each plant part. Total aboveground plant community N and P contents (g m−2) of the 0.25 × 0.25 m plots are the sum of all components. Plant community foliar N and P concentrations (mg g−1) are the sum of leaf N or P content across all species divided by total foliar mass. More details on these methods are available in the NGEE data archive (Sloan and others 2014a, 2018).
Root Distribution
Immediately after harvest of aboveground vascular vegetation, we collected 5-cm-diameter soil cores to the depth of resistance (usually 20–30 cm), which was often the permafrost boundary, for determination of root biomass. In some locations (for example, the very wet troughs and wet centers), saturated soil precluded the use of a soil corer, and in these locations, we cut a 5 cm × 10 cm block with a serrated knife. We measured depth to the base of the organic soil horizon and then sectioned the soil cores or blocks into 5-cm-depth increments in the field and shipped them frozen to Oak Ridge National Laboratory. In the laboratory, we removed all live fine roots (determined by color and elasticity) from a thawed subsample of each soil depth and oven-dried and weighed them (see Sloan and others 2018 for more details).
Environmental and Soil Characteristics
We used a handheld probe (SM300 Soil Moisture Sensor, HH2 Moisture Meter, Delta-T, Cambridge, UK) to measure soil moisture at 0–5 cm depth weekly during the 2012 and 2013 growing seasons (late-June–September). We also measured depth of the active layer with a metal probe. We used HOBO Tidbit v2 loggers (Onset, Bourne, MA, USA) to log soil temperature at 5 cm depth at 30-min intervals throughout the study period. In 2013, we added additional loggers to the low-centered and high-centered polygons at 15 cm (10 July) and 25 cm (8 August) depth. We measured soil pH in subsamples of the 0–5 cm depth increment using a 4:1 ratio of distilled water to soil (see Sloan and others 2014b for more details).
Plant-Available Inorganic Nitrogen
In 2012, we used Plant Root Simulator (PRS®) probes (Western Ag Innovations Inc., Saskatoon, SK Canada) to estimate plant-available inorganic N, or N supply rate, as ammonium (NH4+–N) and nitrate (NO3−–N). The probes consist of a plastic casing containing a 55 mm × 16 mm ion exchange resin membrane (two-sided area of 17.5 cm2), which is intended to mimic the action of a plant root, acting as an “infinite” sink of labile nutrients until membrane saturation (Western Ag 2018). The probes provide a dynamic measure of nutrient supply rate to the resin surface, which is more likely than static measures of nutrient concentrations to correlate with plant nutrient uptake rate. The PRS® probes have advantages over traditional soil sampling as they cause minimal disturbance and permit remeasurement in the same location over time (Johnson and others 2005), and compared to resins in bead form, the membranes facilitate direct soil–resin contact (Harrison and Maynard 2014). On June 23, 2012, we inserted four pairs of probes (where each pair consisted of one anion and one cation probe) adjacent to the 1 × 1 m permanent plot. The probes were oriented vertically with the membranes at 0–5.5 cm within the soil profile. We recovered the first set of probes on July 27, 2012, and replaced them with new probes in the same slits. We recovered these probes on August 28, 2012. On each recovery date, we combined the four anion probes and four cation probes from each plot, cleaned them of soil in the laboratory according to manufacturer instructions, and shipped them in insulated containers for further analysis (Western Ag 2018). There the probes were eluted for 1 h in 0.5 M HCl, and colorimetry using an automated flow injection analysis system was used to analyze the eluate for nitrate and ammonium concentrations. The detection limit was 0.1 mg ml−1 for both N types. As values from the early season (June–July) and late season (July–August) PRS® probes were strongly correlated at the plot level, we summed them to give total growing season values. Phosphorus adsorption onto the resins also was analyzed in a similar manner, but the majority of values were less than the detection limit, precluding further analysis (see Sloan and others 2014c for more details).
Whereas the measurements in 2012 focused on spatial variability across the contrasting polygonal microhabitats, the focus of investigations in 2013 was on seasonal variation in N availability within the vertical soil profile and in plant community N content. We installed PRS® probes in new locations around the center, rim, and trough of low- and high-centered polygons only (24 plots), as these plots represented the end points of the soil moisture spectrum. We replaced these probes at approximately 14-day intervals throughout the growing season, making a total of six sampling periods between June 18 and September 13, 2013. We also installed deeper probes when thaw permitted, at 11–16.5 cm depth for the final four intervals (July 16–September 13, 2013) and at 22–27.5 cm depth for the final two intervals (August 13–September 13, 2013). To produce seasonal totals of NH4+–N at multiple soil depths, we used thaw depth data to estimate the number of days of thaw during the growing season at each probe depth increment for each plot and multiplied these values by the average NH4+–N availability per day at that depth. We harvested and measured plant biomass and foliar N content in 10 × 10 cm subplots adjacent to the probes at 2-week intervals from June 22 to August 29, 2013.
Statistics
We analyzed differences in total inorganic N availability in the 0–5.5 cm horizon in 2012 between microhabitats using one-way ANOVA, and within microhabitats, we used paired t tests to test differences between NH4+–N and NO3––N. We checked all data for normality and heteroscedasticity and log transformed where appropriate. We used linear mixed models to test the importance of vegetation and soil variables in determining ammonium and nitrate availability using polygon as a random effect to account for the spatial relationships between plots. First, we fit all explanatory variables and their quadratic terms, retaining only significant quadratic terms. We then identified significant interactions using a series of models containing main effects and all possible two-way interactions, and then simplified the models by removing nonsignificant terms. We used the methods of Nakagawa and Schielzeth (2013) to calculate the proportion of the data explained by fixed and random effects after examining the model fits visually to ensure assumptions were met. Relationships between plant-available N and vascular plant N content were analyzed using linear regression. We used R 3.1.0 (R Core Team 2014) for all statistical analyses.
Results
Vegetation and Environmental Characteristics of Microhabitats
We investigated changes in vegetation and environmental characteristics across microhabitats that encompassed low-centered polygons with low-lying rims and limited trough development, low-centered polygons with raised rims, transitional polygons with flat centers, and high-centered polygons with sloping rims.
Troughs
Vegetation and environmental characteristics of trough microhabitats were similar across polygon types (Table 1; Figures S1–S3). Trough soils were at or near saturation throughout the growing season and had thick organic horizons (12–15 cm). Average soil temperatures at 5 cm depth were approximately 1 °C higher than in the drier microhabitats (Table 1). Soil pH (top 5 cm) was higher in the troughs (5.20–5.54) of all four polygon types than in the rims and centers (4.39–5.10) (Table 1). Troughs had high vascular plant biomass (90 g m−2 on average across polygon types) and consistently high moss cover. Vascular plant communities were dominated by the sedge Carex aquatilis, with lesser amounts of the grass Dupontia fisheri and the forb Petasites frigidus.
Rims
The low-lying rims of polygons on the margins of the drained thaw lake had similar vegetation composition and biomass to low-center ponds, but soils did not remain saturated throughout the growing season, and average soil temperatures at 5 cm depth were lower in the rims than in the ponds (Table 1). Raised and sloping rim soils had intermediate moisture content and temperatures and thinner organic horizons than adjacent troughs (Table 1). Raised and sloping rims in low-, flat- and high-center polygons differed from other locations in the composition of the vegetation community, which included the sedges Eriophorum angustifolium and E. russeolum, Luzula arctica (rush), and greater than 15% lichen cover. Vascular biomass averaged 43 g m−2 in rims, with the lowest values (24.7 g m−2) on the sloping rims of high-centered polygons.
Spatial Variability of Plant-Available Inorganic Nitrogen
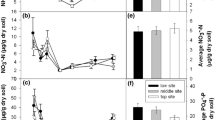
Growing season total plant-available inorganic N at 0–5.5 cm depth (NO3−–N + NH4+–N; Figure 1) did not differ significantly between microhabitats (one-way ANOVA, F11, 36 = 1.55, P = 0.155). Nevertheless, there was more than twofold variation in plant-available inorganic N across the landscape (19.5 ± 2.8 μg N in low-lying rims vs. 44.6 ± 9.4 μg N in flat centers, per 10 cm2 of resin during a 67-day burial period). Soil moisture explained 76% of the variability in NH4+–N availability across the landscape, with the greatest values of NH4+–N availability occurring in the wettest sites (Figure 2A). Other soil factors, including temperature, pH, or organic layer thickness, did not improve the relationship. NO3––N availability within flat centers, high centers, and raised, sloping, or low-lying rims was generally similar to NH4+–N availability, averaging 55% of total availability, but in troughs of all polygon types and in low-center ponds, NH4+–N availability was significantly greater than NO3––N availability (Figure 1), averaging 83% of total availability. The fraction of total inorganic N availability attributed to NO3––N declined linearly with increasing soil moisture (Figure 2B).

Data source: Sloan and others (2014c).
Plant-available inorganic N (NH4+ and NO3−) at 0–5 cm depth during the growing season (June 23–August 28, 2012) in contrasting microhabitats in polygonal tundra. Data are the mean ± SE of four polygons of the amount on N absorbed onto 10 cm2 of resin over 67 days.
Relationship between soil moisture measured at 0–5 cm depth and A plant-available NH4+–N, and B the fraction of total inorganic N attributed to NO3−–N. Data are the mean ± SE of four polygons per polygon type. N availability is reported as the amount absorbed onto 10 cm2 of resin over a 67-day incubation period. Triangles = low-centered polygons (drained thaw lake), circles = low-centered polygons, squares = flat-centered polygons, diamond = high-centered polygons, gray = center, white = rim, black = trough.
Temporal Variability in Plant-Available Inorganic Nitrogen
Ammonium availability at 0–5.5 cm depth varied little during the 2013 growing season in any microhabitat in the low- and high-centered polygons (the wetter and drier endpoints of the soil moisture gradient, respectively) where temporal variability was quantified (Figure 3). As in 2012, ammonium availability in 2013 was consistently higher in troughs and low centers than in high centers, raised and sloping rims. As thaw depth increased, measurements in deeper soil horizons showed greater ammonium availability at depth, except in the troughs of high-centered polygons (Figure 3). Despite the much shorter duration that deeper soil was thawed relative to surface soil, deeper soil contributed a significant fraction of total ammonium availability integrated over the growing season (Figure 4).

Data source: Sloan and others (2014c).
Plant-available NH4+-N at multiple depths in microhabitats of low- and high-centered polygons during the 2013 growing season. Data are the mean ± SE of four polygons from each of the six microhabitats. Available N is expressed as the amount of NH4+–N absorbed onto 10 cm2 of resin over a 14-day incubation period. No data were collected on 4 September from two of the microhabitats. Solid symbols = 5.5 cm depth, gray symbols = 16.5 cm depth; white symbols = 27.5 cm depth.
Upper panels: NH4+-N availability integrated over the 2013 growing season at three depths in microhabitats of low- and high-centered polygons. Data are the mean ± SE of four polygons per microhabitat, calculated as the amount of N absorbed per day onto 10 cm2 of resin, averaged over multiple measurement periods, and multiplied by the number of days the soil was thawed at the given depth. Lower panels: fine-root biomass in soil cores or soil blocks collected July 31–August 4, 2012, in 5 cm increments.
The availability of nitrate remained low (near detection limit) at all locations and depths throughout the 2013 growing season, with the exception of the centers of high-centered polygons. In high-center microhabitats, “hot spots” of high nitrate occurred (Figure S4). Hot spots were of greater magnitude deepest in the soil profile and did not correlate with any changes in measured environmental conditions.
Fine-Root Biomass
The vertical distribution of fine roots at the end of July–early August in 2012 was strikingly different from the seasonally integrated distribution of available ammonium (Figure 4). Total fine-root biomass in rims and centers of low-centered polygons was three times that of high-centered polygons, but the depth distribution was shallower: More than 80% of fine-root biomass was in the top 10 cm of low-centered polygons rims and centers compared to 60–72% in high-centered polygons. No more than 2% of root biomass was deeper than 20 cm in any location. Both ammonium availability and root mass were greater in troughs than in rims and centers in both low- and high-centered polygons. There was 2–4 times as much fine-root mass in troughs relative to rims and centers of the same polygon type; troughs of low-centered polygons had twice the fine-root biomass as troughs of high-centered polygon troughs.
Aboveground Plant N Concentration and Content
Foliar N concentration in August 2012 ranged from 10 to 49 mg g−1; 70% of 209 individual observations ranged from 18 to 28 mg g−1, with a biomass-weighted mean value of 24 ± 2 mg g−1 (Table 2; Figure 5). Species or PFT was not a significant source of variation with the exception of the only evergreen shrub Vaccinium vitis-idaea, which had significantly lower N concentration (10 mg g−1). The influence of landscape position on foliar N concentration, independent of differences in plant community composition, can be looked at only in Carex aquilfolium, the only species present in all plots. N concentration in Carex was 25.6 ± 0.4 mg g−1 and varied little (± 12%) with landscape position. N concentration of the entire vascular plant community also did not vary with landscape position, with the notable exception of a 16% lower concentration in the centers of high-centered polygons, attributable to the presence of the evergreen, low-N Vaccinium vitis-idaea. Given the general constancy of foliar N concentration across landscape position, total aboveground N content (g m−2 ground area) of the vascular plant community reflected the spatial trends in plant community biomass (Figure 5B). Total plant N content was the greatest in troughs and generally declined with soil moisture, with the least amount in the centers and rims of high-centered polygons. Soil moisture fraction explained 85% of the variation in plant N content (Figure 6A).

Data source: Sloan and others (2018).
A Foliar N and P concentrations of the plant community calculated as the total N or P content of leaves in a 10 cm × 10 cm plot divided by total leaf mass. B Total aboveground N and P content per m2 ground area. Data are the mean ± SE of four polygons per microhabitat. Solid bars: N; striped bars: P.
Relationship between plant community aboveground N content (g N per m2 ground area) and A soil moisture measured at 0–5 cm depth, and B and plant-available NH4+–N. Data are the mean ± SE of four polygons per polygon type. NH4+–N availability is reported as the amount absorbed onto 10 cm2 of resin over a 67-day incubation period. Triangles = low-centered polygons (drained thaw lake); circles = low-centered polygons; squares = flat-centered polygons; diamond = high-centered polygons. Gray symbols = center; white symbols = rim; black symbols = trough.
When we assessed the temporal dynamics of aboveground plant community N content in 2013, we found that foliar N concentrations generally declined throughout the growing season, similar to the observations of Chapin and others (1975), except in the center of high-centered polygons. At these locations, the plant community began the growing season with a lesser N concentration than observed elsewhere and remained relatively constant (Figure S5A). Total N content, reflecting both foliar N concentration and plant biomass, peaked in early August in the low-centered polygon and the troughs of high-centered polygons (Figure S5B), also similar to the pattern observed by Chapin and others (1975).
Foliar P concentrations averaged 1.75 mg g−1 (range 0.86–3.24 mg g−1). Spatial variation in P concentration was greater than that of N (Figure 5); plant community P concentration tended to increase with plant biomass. The lowest concentration was in the centers of high-centered polygons, and the highest concentrations were in troughs. The spatial pattern in total aboveground P content was similar to that of N except that differences were larger among polygon types and microhabitats. Plant community N:P ratio ranged from 10.5 to 16.8. The N:P ratio in Carex aquatilis varied from 10.9 in troughs of wet low-centered polygons to 20.9 in the dry rims of flat-centered and high-centered polygons (data not shown). Taking an N:P ratio above 20 as a rough indicator of P limitation and N:P below 10 of N limitation (Güsewell 2004), most of our observations can be considered indicative of co-limitation.
Plant–Soil Interactions
The spatial pattern in plant community N content in 2012 was correlated with NH4+–N availability (Figure 6B). However, it also is noted that both NH4+–N availability and plant N content were correlated with soil moisture (Figures 2A, 6A), and it is likely that the microtopographic gradients in soil moisture are the dominant influence on plant communities and soil nutrient status at this site. There was no correlation between NO3––N availability and plant N content.
Discussion
The microhabitats created by polygonal tundra and the seasonal variation in conditions created by thawing permafrost lead to a unique set of interactions between vegetation and plant-available N. Here, we discuss how our observations address the two primary objectives: (1) the form and availability of inorganic N and the relationships between vegetation, soil variables, and plant-available N and (2) the spatial and temporal dynamics of plant-available N and plant response to those dynamics. We then consider the implications for these observations to predictions of how this ecosystem will respond to changing environmental conditions.
Controls on Plant-Available Inorganic Nitrogen
We showed that in the polygonal tundra of the Barrow Environmental Observatory, microhabitat primarily influenced the form rather than amount of available inorganic N occurring in the upper soil horizon. While some caution may be needed in resin assessments of NO3−–N relative to NH4+–N, given potentially higher resin affinity for NH4+–N (Giblin and others 1994), we certainly found that the resins accumulated large amounts of NO3––N in microhabitats such as flat and high centers. The lack of variability in total inorganic N availability in 2012 across microhabitats, coupled with the uniformity in aboveground plant N concentration, was unexpected given the substantial variation in peak vascular biomass and coverage of plant functional types, as well as the known differences in soil moisture and soil texture driven by differences in microtopography across the polygons. Total root-available inorganic N may not be the best metric for evaluating plant–soil N interactions in this system. Resin-based approaches to N availability are especially sensitive to soil moisture, as would be expected for a plant root as well, and high values of soil mineral N do not necessarily translate into greater N to plants (or greater PRS® values) if roots are not present or the soil is too dry to facilitate transport of N to roots (Johnson and others 2005). We also note that our data on plant-available N should not be interpreted as a proxy for net N mineralization as the correlation between resin-based approaches to N availability and net mineralization has been quite variable in a number of studies (Giblin and others 1994; Johnson and others 2005; Harrison and Maynard 2014; and references therein). Also, we did not measure organic N (for example, amino acids) availability, which is known to be a potentially important N source for some arctic species (Chapin and others 1993; Kielland 1994; Schimel and Chapin 1996; McKane and others 2002). McKane and others (2002) concluded from observations in the tussock tundra at Toolik Lake, Alaska, that species dominance (aboveground productivity) was related to species’ ability to exploit the most abundant resource, which varied with form of N, seasonal variability, and location in the soil profile. In our study, plant community N content primarily reflected midseason ammonium availability in the upper soil horizon, and ammonium availability was strongly correlated with soil moisture.
Relationships between site factors, inorganic N availability, and plant N were obscured by the fraction of inorganic N availability attributed to NO3––N, which declined significantly with soil moisture. The relationship between NO3––N fraction and soil moisture is consistent with metagenomics analyses demonstrating high nitrification potential of microbial populations in drier polygon features (Heikoop and others 2015). In contrast to the observations of Liu and others (2018) that soil nitrate can contribute about one-third of the N used by tundra plants in northern Alaska, we saw no relationship between NO3––N availability and plant productivity or N content, suggesting that nitrate, which comprised a substantial component of total available inorganic N in drier microhabitats (Figure 3B), may not be taken up by the vascular species present. In particular, V. vitis-idaea, which was present only on the dry, nitrate-rich centers of high-centered polygons, had the lowest foliar N concentration at the site, and was shown previously in the tussock tundra study of McKane and others (2002) that this species relied on organic N and ammonium and not on nitrate (but see Liu and others 2018).
Seasonal Variation in N Availability
A unique feature of the permafrost environment is that the soil volume that can be exploited by plant roots is not fixed but increases throughout the growing season as the thaw depth increases. Keuper and others (2012) demonstrated that soil near the permafrost boundary in subarctic peatlands can release a biologically relevant amount of plant-available N as it thaws and through continued microbial mineralization of organically bound N. Likewise, Salmon and others (2016) reported a strong positive relationship between depth of permafrost thaw and N availability in a tundra ecosystem. A key question is whether plants can exploit the increasing supply of available N as the soil thaws (Koven and others 2015).
In our study, there was little temporal variation in ammonium availability in the upper horizon of the active layer of soil within individual microhabitats despite seasonal variation in soil moisture and temperature. In contrast to ammonium, nitrate availability occurred as “hotspots” with wide spatial variability. There was, however, substantial seasonal variation in total ammonium availability in the total volume of thawed soil. Plant-available ammonium increased with soil depth in the majority of microhabitats, but the N made available in deeper, newly thawed soil was present for a relatively small fraction of the growing season. Furthermore, the rooting depth distributions across the polygons were extremely shallow; 98% of fine-root biomass across the polygonal tundra was in the top 20 cm of the soil (Table 1). This fits the pattern observed across the tundra, where shallow tundra roots are primarily confined to the organic horizon (Iversen and other 2015). The low density of plant roots at depth and the colder soils presumably contributed to limited plant uptake. Limited plant N uptake, combined with seasonal variation in the balance between N mineralization and microbial immobilization in cold tundra soils (Schimel and Mikan 2005), resulted in consistently higher levels of available ammonium in deeper soil horizons as the soil thawed later in the year. The increase in N supply was not reflected in aboveground plant biomass or N content, which had already peaked before the deeper soil had thawed (Figure S5B). If not utilized by plants or microbes, the potential exists for loss of N from the ecosystem through lateral transport or denitrification (Heikoop and others 2015; Treat and others 2016).
Implications for Arctic Ecosystem Function in a Changing Climate
The arctic environment is very different from the temperate biomes, and Earth system models often have not accounted for many processes unique to the Arctic (Koven and others 2015). For reliable predictions of responses of arctic ecosystems to climatic warming, models will need to incorporate some of the unusual aspects of N dynamics and plant–soil interactions in permafrost systems, including the increase in soil depth and nutrient capital during the growing season and the relationships between root density profiles and N preferences of different plant species (Iversen and others 2015).
It has been proposed that increasing thaw depth in permafrost regions, as is projected to occur with climatic warming, will increase the amount of N available for plant uptake and stimulate plant productivity in the N-limited arctic environment (Keuper and others 2012). Our data support the premise that N supply rate is substantial in deeper soils and at the permafrost boundary, consistent with observations in Alaskan tussock tundra (Sistla and others 2013; Harms and Jones 2012) and Siberia (Wild and others 2015; Beermann and others 2017), and therefore total N supply increases as the thaw depth increases during the growing season. Furthermore, near-surface permafrost soil can contain significantly more plant-available N than active-layer soil (Keuper and others 2012). However, N in near-surface permafrost soil will become available to plants only late in the growing season, and our observations show that it is incorrect to assume that plants will necessarily access the additional N that becomes available as the growing season progresses, as this will be contingent on roots being present and active in the newly thawed and cold soil near the permafrost boundary. The increased N supply at depth occurred after peak plant aboveground biomass had been attained, and there were few roots in the cold, deeper soil. Even if roots were present in deeper soil, 15N labeling studies at this site with three species with contrasting rooting behavior demonstrated that plant N uptake was not consistently related to root biomass density profiles as is commonly assumed by many models, but rather on species-specific physiological root traits and competition between roots and microbes (Zhu and others 2016). Keuper and others (2017), however, demonstrated that some species that do have roots at the thaw front have the capacity to take up N late in the season and store it for use the next spring. Nevertheless, we conclude that in evaluating potential responses of arctic ecosystems to climatic warming, it should not be assumed that a warming-induced thickening of the active layer and release of additional N will support increased plant productivity, as this will depend on characteristics of the species present. With permafrost features and a full N cycle included in the Community Land Model version 4.5, Koven and others (2015) showed the impact of deep N on the carbon budget was small because of the seasonal offset between deep N availability and plant N demand. Although they did not account for the possibility of late-season N uptake and storage in roots for use in the following spring (Keuper and others 2017), they concluded that future climate change effects on carbon balance would depend more on the rate and extent of permafrost thaw and soil decomposition than on enhanced N availability for vegetation growth resulting from permafrost thaw. Indeed, in our study, increased plant N content and biomass was associated with N availability in surface soils. The possibility that a warmer atmosphere will stimulate N mineralization and N availability in surface soils and thereby increase plant productivity remains a viable hypothesis.
Soil moisture had the dominant control on the plant and soil N economy, with the driest microsites associated with less ammonium availability, a greater fraction of total inorganic N present as nitrate, and a lesser amount of N in the aboveground plant community. Plants were not taking advantage of the greater nitrate availability—the one woody plant on the site (Vaccinium vitis-idaea) occurred only on dry, high-centered polygons where the relative amount of nitrate was greatest, and it had lower foliar N than any other species on site. If climatic warming leads to a general drying of the tundra, our data suggest that a greater proportion of the inorganic N pool will be present as nitrate. Nitrate may be of less utility to plants such as Vaccinium vitis-idaea that have a physiological preference for ammonium, although isotopic evidence indicated that Vaccinium sp. in the Alaskan tundra can take up and assimilate nitrate (Liu and others 2018). Alternatively, although nitrate availability may increase with drying soils, water limitation may limit plant production instead of nitrogen availability. Even in a putatively N-limited system, increased nitrification that is not coupled to plant uptake could lead to N losses from the system and adverse consequences for water quality—responses that are an indication of ecosystem degradation.
Conclusions
Two primary conclusions that emerged from this analysis are: (1) Nitrogen dynamics are strongly influenced by spatial patterns in soil moisture. Drier habitats have a greater proportion of their N economy as nitrate, but the plant species present on the drier sites cannot necessarily take advantage of the nitrate supply, perhaps because of increasing moisture limitation. (2) Although increasing thaw depth increases the total availability of inorganic N, the additional N at depth is not necessarily taken up by plants and is not associated with increased plant productivity. Both of these conclusions should be considered when projecting responses of this unique arctic environment to future warmer, drier conditions.
References
Aerts R, Chapin FS. 2000. The mineral nutrition of wild plants revisited: A re-evaluation of processes and patterns. Adv Ecol Res 30:1–67.
Beermann F, Langer M, Wetterich S, Strauss J, Boike J, Fiencke C, Schirrmeister L, Pfeiffer EM, Kutzbach L. 2017. Permafrost thaw and liberation of inorganic nitrogen in eastern Siberia. Permafr Periglac Process 28:605–18.
Biasi C, Meyer H, Rusalimova O, Hammerle R, Kaiser C, Baranyi C, Daims H, Lashchinsky N, Barsukov P, Richter A. 2008. Initial effects of experimental warming on carbon exchange rates, plant growth and microbial dynamics of a lichen-rich dwarf shrub tundra in Siberia. Plant Soil 307:191–205.
Bockheim JG, Everett LR, NelsonFE Hinkel KM, Brown J. 1999. Soil organic carbon storage and distribution in Arctic Tundra, Barrow, Alaska. Soil Sci Soc Am J 63:934–40.
Brown J. 1967. Tundra soils formed over ice wedges northern Alaska. Soil Sci Soc Am Proc 31:686.
Chapin FS, Van Cleve K, Tieszen LL. 1975. Seasonal nutrient dynamics of tundra vegetation at Barrow, Alaska. Arct Alp Res 7:209–26.
Chapin FS, Moilanen L, Kielland K. 1993. Preferential use of organic nitrogen for growth by a nonmycorrhizal arctic sedge. Nature 361:150–3.
Chapin FS, Shaver GR, Giblin AE, Nadelhoffer KJ, Laundre JA. 1995. Responses of arctic tundra to experimental and observed changes in climate. Ecology 76:694–711.
Giblin AE, Laundre JA, Nadelhoffer KJ, Shaver GR. 1994. Measuring nutrient availability in arctic soils using ion-exchange resins—a field-test. Soil Sci Soc Am J 58:1154–62.
Graglia E, Jonasson S, Michelsen A, Schmidt IK, Havstrom M, Gustavsson L. 2001. Effects of environmental perturbations on abundance of subarctic plants after three, seven and ten years of treatments. Ecography 24:5–12.
Güsewell S. 2004. N: P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–66.
Harms TK, Jones JB Jr. 2012. Thaw depth determines reaction and transport of inorganic nitrogen in valley bottom permafrost soils. Glob Change Biol 18:2958–68.
Harrison DJ, Maynard DG. 2014. Nitrogen mineralization assessment using PRS™ probes (ion-exchange membranes) and soil extractions in fertilized and unfertilized pine and spruce soils. Can J Soil Sci 94:21–34.
Heikoop JM, Throckmorton HM, Newman BD and others 2015. Isotopic identification of soil and permafrost nitrate sources in an Arctic tundra ecosystem. J Geophys Res-Biogeosci 120:1000–17.
Hinkel KM, Eisner WR, Bockheim JG, Nelson FE, Peterson KM, Dai XY. 2003. Spatial extent, age, and carbon stocks in drained thaw lake basins on the Barrow Peninsula, Alaska. Arct Antarct Alp Res 35:291–300.
Hobbie SE, Nadelhoffer KJ, Hogberg P. 2002. A synthesis: The role of nutrients as constraints on carbon balances in boreal and arctic regions. Plant Soil 242:163–70.
Iversen CM, Sloan VL, Sullivan PF, Euskirchen ES, McGuire AD, Norby RJ, Walker AP, Warren JM, Wullschleger SD. 2015. The unseen iceberg: plant roots in arctic tundra. New Phytol 205:34–58.
Johnson DW, Verburg PSJ, Arnone JA. 2005. Soil extraction, ion exchange resin, and ion exchange membrane measures of soil mineral nitrogen during incubation of a tallgrass prairie soil. Soil Sci Soc Am J 69:260–5.
Jonasson S, Michelsen A, Schmidt IK, Nielsen EV. 1999. Responses in microbes and plants to changed temperature, nutrient, and light regimes in the arctic. Ecology 80:1828–43.
Jonasson S, Shaver GR. 1999. Within-stand nutrient cycling in arctic and boreal wetlands. Ecology 80:2139–50.
Kelley AM, Epstein HE, Ping C-L, Walker DA. 2012. Soil nitrogen transformations associated with small patterned ground features along a North American Arctic transect. Permaf Periglac Process 23:196–206.
Keuper F, van Bodegom PM, Dorrepaal E, Weedon JT, van Hal J, van Logtestijn RSP, Aerts R. 2012. A frozen feast: thawing permafrost increases plant-available nitrogen in subarctic peatlands. Glob Change Biol 18:1998–2007.
Keuper F, Dorrepaal E, van Bodegom PM, van Logtestijn R, Venhuizen G, van Hal J, Aerts R. 2017. Experimentally increased nutrient availability at the permafrost thaw front selectively enhances biomass production of deep-rooting subarctic peatland species. Glob Change Biol 23:4257–66.
Kielland K. 1994. Amino-acid-absorption by arctic plants—implications for plant nutrition and nitrogen cycling. Ecology 75:2373–83.
Koven CD, Lawrence DM, Riley WJ. 2015. Permafrost carbon-climate feedback is sensitive to deep soil carbon decomposability but not deep soil nitrogen dynamics. Proc Natl Acad Sci USA 112:3752–7.
Lamb EG, Han S, Lanoil BD, Henry GHR, Brummell ME, Banerjee S, Siciliano SD. 2011. A High Arctic soil ecosystem resists long-term environmental manipulations. Glob Change Biol 17:3187–94.
Langford Z, Kumar J, Hoffman FM, Norby RJ, Wullschleger SD, Sloan VL, Iversen CM. 2016. Mapping arctic plant functional type distributions in the Barrow Environmental Observatory using WorldView-2 and LiDAR datasets. Remote Sens 8:733.
Lara MJ, McGuire AD, Euskirchen ES, Tweedie CE, Hinkel KM, Skurikhin AN, Romanovsky VE, Grosse G, Bolton WR, Genet H. 2015. Polygonal tundra geomorphological change in response to warming alters future CO2 and CH4 flux on the Barrow Peninsula. Glob Change Biol 21:1634–51.
Liu XY, Koba K, Koyama LA, Hobbie SE, Weiss MS, Inagaki Y, Shaver GR, Giblin AE, Hobara S, Nadelhoffer KJ, Sommerkorn M, Rastetter EB, Kling GW, Laundre JA, Yano Y, Makabe A, Yano M, Liu CQ. 2018. Nitrate is an important nitrogen source for Arctic tundra plants. Proc Natl Acad Sci 115:3398–403.
Loranty MM, Goetz SJ, Beck PSA. 2011. Tundra vegetation effects on pan-Arctic albedo. Environ Res Lett 6:024014.
McKane RB, Johnson LC, Shaver GR, Nadelhoffer KJ, Rastetter EB, Fry B, Giblin AE, Kielland K, Kwiatkowski BL, Laundre JA, Murray G. 2002. Resource-based niches provide a basis for plant species diversity and dominance in arctic tundra. Nature 415:68–71.
Nakagawa S, Schielzeth H. 2013. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–42.
NOAA. 2018. Data tools: 1981-2010 normals. https://www.ncdc.noaa.gov/cdo-web/datatools/normals. Accessed March 11, 2018.
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, Cornelissen JHC, Gurevitch J, GCTE NEWS. 2001. A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–62.
Salmon VG, Soucy P, Mauritz M, Celis G, Natali SM, Mack MC, Schuur EAG. 2016. Nitrogen availability increases in a tundra ecosystem during five years of experimental permafrost thaw. Glob Change Biol 22:1927–41.
Schimel JP, Chapin FS. 1996. Tundra plant uptake of amino acid and NH4 + nitrogen in situ: Plants compete well for amino acid N. Ecology 77:2142–7.
Schimel JP, Mikan C. 2005. Changing microbial substrate use in Arctic tundra soils through a freeze-thaw cycle. Soil Biol Biochem 37:1411–18.
Schmidt IK, Jonasson S, Shaver GR, Michelsen A, Nordin A. 2002. Mineralization and distribution of nutrients in plants and microbes in four arctic ecosystems: responses to warming. Plant Soil 242:93–106.
Shaver GR, Chapin FS. 1980. Response to fertilization by various plant-growth forms in an Alaskan tundra—nutrient accumulation and growth. Ecology 61:662–75.
Shaver GR, Chapin FS. 1995. Long-term responses to factorial, NPK fertilizer treatment by Alaskan wet and moist tundra sedge species. Ecography 18:259–75.
Sistla SA, Moore JC, Simpson RT, Gough L, Shaver GR, Schimel JP. 2013. Long-term warming restructures Arctic tundra without changing net soil carbon storage. Nature 497:615–19.
Sloan VL, Brooks JD, Wood SJ, Liebig JA, Siegrist J, Iversen CM, Norby RJ. 2014a. Plant community composition and vegetation height, Barrow, Alaska, Ver. 1. Next Generation Ecosystem Experiments Arctic Data Collection, Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA. Data set accessed at http://dx.doi.org/10.5440/1129476.
Sloan VL, Liebig JA, Hahn MS, Curtis JB, Brooks JD, Rogers A, Iversen CM, Norby RJ. 2014b. Soil temperature, soil moisture and thaw depth, Barrow, Alaska, Ver. 1. Next Generation Ecosystem Experiments Arctic Data Collection, Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA. Data set accessed at http://dx.doi.org/10.5440/1121134.
Sloan VL, Iversen CM, Liebig JA, Curtis JB, Hahn MS, Siegrist J, Norby RJ. 2014c. Plant available nutrients, Barrow, Alaska Ver. 1. Next Generation Ecosystem Experiments Arctic Data Collection, Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA. Data set accessed at http://dx.doi.org/10.5440/1120920.
Sloan VL, McGuire AD, Euskirchen ES, Slette IJ, Carter KR, Wood SJ, Childs J, Iversen CM, Norby RJ. 2018. Leaf area, vegetation biomass and nutrient content, Barrow, Alaska, 2012-2013. Next Generation Ecosystem Experiments Arctic Data Collection, Oak Ridge National Laboratory, U.S. Department of Energy, Oak Ridge, Tennessee, USA. Data set accessed at https://dx.doi.org/10.5440/1375316.
Sturm M, Schimel J, Michaelson G, Welker JM, Oberbauer SF, Liston GE, Fahnestock J, Romanovsky VE. 2005. Winter biological processes could help convert arctic tundra to shrubland. Bioscience 55:17–26.
Sullivan PF, Welker JM. 2005. Warming chambers stimulate early season growth of an arctic sedge: results of a minirhizotron field study. Oecologia 142:616–26.
Treat CC, Wollheim WM, Varner RK, Bowden WB. 2016. Longer thaw seasons increase nitrogen availability for leaching during fall in tundra soils. Environ Res Lett 11:6.
van Wijk MT, Clemmensen KE, Shaver GR, Williams M, Callaghan TV, Chapin FS, Cornelissen JHC, Gough L, Hobbie SE, Jonasson S, Lee JA, Michelsen A, Press MC, Richardson SJ, Rueth H. 2004. Long-term ecosystem level experiments at Toolik Lake, Alaska, and at Abisko, Northern Sweden: generalizations and differences in ecosystem and plant type responses to global change. Glob Change Biol 10:105–23.
Walker DA, Epstein HE, Romanovsky VE, Ping CL, Michaelson GJ, Daanen RP, Shur Y, Peterson RA, Krantz WB, Raynolds MK, Gould WA, Gonzalez G, Nicolsky DJ, Vonlanthen CM, Kade AN, Kuss P, Kelley AM, Munger CA, Tamocai CT, Matveyeva NV, Daniels FJA. 2008. Arctic patterned-ground ecosystems: A synthesis of field studies and models along a North American Arctic Transect. J Geophys Res-Biogeosci 113: G3S01.
Washburn AL. 1979. Geocryology. A survey of periglacial processes and environments. London: Edward Arnold.
Webber PJ. 1978. Spatial and temporal variation of the vegetation and its production, Barrow, Alaska. In: Tieszen LL, Ed. Vegetation and production ecology of an Alaskan arctic tundra. New York: Springer. p 37–112.
Weintraub MN, Schimel JP. 2005a. Nitrogen cycling and the spread of shrubs control changes in the carbon balance of arctic tundra ecosystems. Bioscience 55:408–15.
Weintraub MN, Schimel JP. 2005b. The seasonal dynamics of amino acids and other nutrients in Alaskan Arctic tundra soils. Biogeochemistry 73:359–80.
Welker JM, Fahnestock JT, Sullivan PF, Chimner RA. 2005. Leaf mineral nutrition of Arctic plants in response to warming and deeper snow in northern Alaska. Oikos 109:167–77.
Western Ag. 2018. Plant root simulator (PRS®) probes. https://www.westernag.ca/innov. Accessed March 11, 2018.
Wild B, Schnecker J, Knoltsch A, Takriti M, Mooshammer M, Gentsch N, Mikutta R, Alves RJE, Gittel A, Lashchinskiy N, Richter A. 2015. Microbial nitrogen dynamics in organic and mineral soil horizons along a latitudinal transect in western Siberia. Glob Biogeochem Cycles 29:567–82.
Zamin TJ, Bret-Harte MS, Grogan P. 2014. Evergreen shrubs dominate responses to experimental summer warming and fertilization in Canadian mesic low arctic tundra. J Ecol 102:749–66.
Zamin TJ, Grogan P. 2012. Birch shrub growth in the low Arctic: the relative importance of experimental warming, enhanced nutrient availability, snow depth and caribou exclusion. Environ Res Lett 7:034027.
Zhu Q, Iversen CM, Riley WJ, Slette IJ, Vander Stel HM. 2016. Root traits explain observed tundra vegetation nitrogen uptake patterns: Implications for trait-based land models. J Geophys Res-Biogeosci 121:3101–12.
Zona D, Lipson DA, Zulueta RC, Oberbauer SF, Oechel WC. 2011. Microtopographic controls on ecosystem functioning in the Arctic Coastal Plain. J Geophys Res-Biogeosci 116: G00I08.
Acknowledgements
We thank Deanne Brice, Ingrid Slette, and Kelsey Carter for their help in the field and laboratory, and Verity Salmon and two anonymous reviewers for their helpful comments on an earlier version of this paper. The Next-Generation Ecosystem Experiments (NGEE Arctic) project is supported by the US Department of Energy, Office of Science, Biological and Environmental Research Program. Oak Ridge National Laboratory is managed by UT-Battelle, LLC, for the US Department of Energy under contract DE-AC05-00OR22725.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Contributions
RJN, VLS, and CMI conceived and designed the study. RJN, VLS, CMI, and JC performed field research and laboratory analyses. RJN and VLS analyzed the data and wrote the paper with substantial input from CMI.
This manuscript has been authored by UT-Battelle, LLC under Contract No. DE-AC05-00OR22725 with the US Department of Energy. The United States Government retains and the publisher, by accepting the article for publication, acknowledges that the United States Government retains a non-exclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this manuscript, or allow others to do so, for United States Government purposes. The Department of Energy will provide public access to these results of federally sponsored research in accordance with the DOE Public Access Plan (http://energy.gov/downloads/doe-public-access-plan).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Norby, R.J., Sloan, V.L., Iversen, C.M. et al. Controls on Fine-Scale Spatial and Temporal Variability of Plant-Available Inorganic Nitrogen in a Polygonal Tundra Landscape. Ecosystems 22, 528–543 (2019). https://doi.org/10.1007/s10021-018-0285-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-018-0285-6