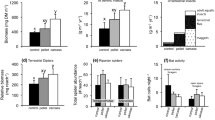
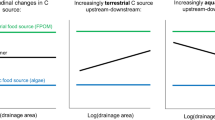
Abstract
The term “keystone species” is used to describe organisms that exert a disproportionately important influence on the ecosystems in which they live. Analogous concepts such as “keystone mutualism” and “mobile links” illustrate how, in many cases, the interactions of two or more species produce an effect greater than that of any one species individually. Because of their role in transporting nutrients from the ocean to river and riparian ecosystems, Pacific salmon (Oncorhynchus spp.) and brown bear (Ursus arctos) have been described as keystone species and mobile links, although few data are available to quantify the importance of this interaction relative to other nutrient vectors. Application of a mass balance model to data from a southwestern Alaskan stream suggests that nitrogen (N) influx to the riparian forest is significantly increased in the presence of both salmon and bear, but not by either species individually. The interactions of salmon and bear may provide up to 24% of riparian N budgets, but this percentage varies in time and space according to variations in salmon escapement, channel morphology and watershed vegetation characteristics, suggesting interdependence and functional redundancy among N sources. These findings illustrate the complexity of interspecific interactions, the importance of linkages across ecosystem boundaries and the necessity of examining the processes and interactions that shape ecological communities, rather than their specific component parts.




Similar content being viewed by others
References
Alexander V, Billington MM. 1986. Nitrogen fixation in the Alaskan taiga. In: Van Cleve K, Chapin FS III, Flanagan PW, Viereck LA, Dyrness CT, eds. Forest ecosystems in the Alaskan taiga. New York: Springer. p 112–20
Bartz KK, Naiman RJ. 2005. Impacts of salmon-borne nutrients on riparian soils and vegetation in southeast Alaska. Ecosystems (in press)
Bencala KE, Walters RW. 1983. Simulation of solute transport in a mountain pool-and-riffle stream: a transient storage model. Water Resour Res 20:1797–803
Ben-David M, Hanley TA, Schell DM. 1998. Fertilization of terrestrial vegetation by spawning Pacific salmon: the role of flooding and predator activity. Oikos 83:47–55
Ben-David M, Hanley TA, Schell DM. 1997a. Seasonal changes in diets of coastal and riverine mink: the role of spawning Pacific salmon. Can J Zool 75:803–11
Ben-David M, Flynn RW, Schell DM. 1997b. Annual and seasonal changes in diets of martens: evidence from stable isotope analysis. Oecologia 111:280–91
Bertness MD, Leonard GH. 1997. The role of positive interactions in communities: lessons from intertidal habitats. Ecology 78:1976–89
Bertness MD, Shumway SW. 1993. Competition and facilitation in marsh plants. Am Nat 142:718–24
Bilby RE, Bisson PA. 1998. Function and distribution of large woody debris. In: Naiman RJ, Bilby RE, eds. River ecology and management. New York: Springer. p 324–46
Bilby RE, Fransen BR, Bisson PA, Walter JK. 1998. Response of juvenile coho salmon (Oncorhynchus kisutch) and steelhead (Oncorhynchus mykiss) to the addition of salmon carcasses to two streams in southwestern Washington, U.S.A. Can J Fish Aquat Sci 55:1909–18
Bilby RE, Fransen BR, Bisson PA. 1996. Incorporation of nitrogen and carbon from spawning coho salmon into the trophic system of small streams: evidence from stable isotopes. Can J of Fish Aquat Sci 53:164–73
Binkley D. 1983. Ecosystem production in Douglas-fir plantations: interactions of red alder and site fertility. For Ecol Manage 5:215–7
Binkley D. 1986. Forest nutrition management. New York: Wiley. P 304
Binkley D, Sollins P, McGill WB. 1985. Natural abundance of Nitrogen-15 as a tool for tracing alder-fixed nitrogen. Soil Sci Soc Am J 49:444–7
Binkley D, Sollins P, Bell R, Sachs D, Myrold D. 1992. Biogeochemistry of adjacent conifer and alder-conifer stands. Ecology 73:2022–33
Blundell GM, Ben-David M, Bowyer RT. 2003. Sociality in river otters: cooperative foraging or reproductive stategies? Behav Ecol 13:134–41
Bond WJ. 1993. Keystone species. In: Schulze E-D, Mooney HA, eds. Biodiversity and ecosystem function. New York: Springer. p 237–253
Bruno JF, Stachowicz JJ, Bertness MD. 2003. Inclusion of facilitation into ecological theory. Trends Ecol Evol 18:119–25
Bryant JP. 1987. Feltleaf willow-snowshoe hare interactions: plant carbon/nutrient balance and floodplain succession. Ecology 68:1319–27
Carroll CR. 1992. Ecological management of sensitive natural areas. In: Fiedler PL, Jain SK, eds. Conservation biology: the theory and practice of nature conservation, preservation and management. New York: Chapman & Hall. P 347–72
Cederholm CJ, Peterson NP. 1985. The retention of coho salmon (Oncorhynchus kisutch) carcasses by organic debris in small streams. Can J Fish Aquat Sci 42:1222–5
Cederholm CJ, Houston DB, Cole DL, Scarlett WJ. 1989. Fate of coho salmon (Oncorhynchus kisutch) carcasses in spawning streams. Can J Fish Aquat Sci 46:1347–1355
Christian CE. 2001. Consequences of a biological invasion reveal the importance of mutualism for plant communities. Nature 413:635–9
Conway WG. 1989. The prospects for sustaining species and their evolution. In: Western D, Pearl MC, eds. Conservation for the twenty-first century. New York: Oxford University Press. p 199–209
Daly GT. 1966. Nitrogen fixation by nodulated Alnus rugosa. Can J Bot 44:1607–21
Deluca TH, Zackrisson O, Nilsson M-C, Sellstedt A. 2002. Quantifying nitrogen-fixation in feather moss carpets of boreal forests. Nature 419:917–20
Edwards RT. 1998. The hyporheic zone. In: Naiman RJ, Bilby RE, ed. River ecology and management. New York: Springer. p 399–429
Fensham RJ, Fairfax RJ, Holman JE, Whitehead PJ. 2002. Quantitative assessment of vegetation structural attributes from aerial photography. Int J Remote Sens 23:2293–317
Galloway JN, Cowling EB. 2002. Reactive nitrogen and the world: 200 years of change. Ambio 31:64–71
Gende SM, Quinn TP, Willson MF. 2001. Consumption choice by bears feeding on salmon. Oecologia 127:372–82
Gilbert LE. 1980. Food web organization and the conservation of neotropical diversity. In: Soule ME, Wilcox BA, eds. Conservation biology: an evolutionary-ecological perspective. Sunderland, MA: Sinauer. p 11–33
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Linkaemper GW, Cromack K Jr., Cummins KW. 1986. Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302
Hartman CW, Johnson PR. 1984. Environmental atlas of Alaska. Fairbanks (AK): Institute of Water Resources/Engineering Experiment Station, University of Alaska
Helfield JM, Naiman RJ. 2001. Effects of salmon-derived nitrogen on riparian forest growth and implications for stream productivity. Ecology 82:2403–9
Helfield JM, Naiman RJ. 2002. Salmon and alder as nitrogen sources to riparian forests in a boreal Alaskan watershed. Oecologia 133:573–82
Hilderbrand GV, Hanley TA, Robbins CT, Schwartz CC. 1999a. Role of brown bears (Ursus arctos) in the flow of marine nitrogen into a terrestrial ecosystem. Oecologia 121:546–50
Hilderbrand GV, Schwartz CC, Robbins CT, Jacoby ME, Hanley TA, Arthur SM, Servheen C. 1999b. The importance of meat, particularly salmon, to body size, population productivity, and conservation of North American brown bears. Can J Zool 77:132–8
Hilderbrand GV, Farley SD, Robbins CT, Hanley TA, Titus K, Servheen C. 1996. Use of stable isotopes to determine diets of living and extinct bears. Can J Zool 74:2080–88
Honeggar R. 1991. Functional aspects of the lichen symbiosis. Annu Rev Plant Physiol Plant Mol Biol 42:553–78
Hurlbert SH. 1997. Functional importance vs. keystoneness: reformulating some questions in theoretical biocenology. Aust J Ecol 22:369–82
Jackson JK, Fisher SG. 1986. Secondary production, emergence and export of aquatic insects of a Sonoran Desert stream. Ecology 67:629–38
Kaye JP, Binkley D, Rhoades C. 2003. Stable soil nitrogen accumulation and flexible organic matter stochiometry during primary floodplain succession. Biogeochemistry 63:1–22
Kielland K., Bryant JP. 1998. Moose herbivory in taiga: effects on biogeochemistry and vegetation dynamics in primary succession. Oikos 82:377–83
Kline TC Jr., Goering JJ, Mathisen OA, Poe PH, Parker PL. 1990. Recycling of elements transported upstream by runs of Pacific salmon: I. δ15N and δ13C evidence in Sashin Creek, southeastern Alaska. Can J Fish Aquat Sci 47:136–144
Kline TC Jr., Goering JJ, Mathisen OA, Poe PH, Parker PL, Scanlan RS. 1993. Recycling of elements transported upstream by runs of Pacific salmon: II. δ15N and δ13C evidence in the Kvichak river watershed, Bristol Bay, southwestern Alaska. Can J Fish Aquat Sci 50:2350–65
Klingensmith KM, Van Cleve K. 1993. Denitrification and nitrogen fixation in floodplain successional soils along the Tanana River, interior Alaska. Can J For Res 23:956–63
Kotliar NB. 2000. Application of the new keystone-species concept to prairie dogs: how well does it work? Conserv Biol 14:1715–21
Larkin GA, Slaney PA. 1997. Implications of trends in marine-derived nutrient influx to south coastal British Columbia salmonid production. Fisheries 22:16–24
Lawrence DB. 1958. Glaciers and vegetation in southeastern Alaska. Am Sci 46:89–122
Lawton JH, Jones CG. 1995. Linking species and ecosystems: organisms as ecosystem engineers. In: Jones CG, Lawton JH, eds. Linking species and ecosystems. New York: Chapman and Hall. p 141–50
Leopold LB, Ed. 1953. Round river: from the journals of Aldo Leopold. New York: Oxford University Press, p 286
Lundberg J, Moberg F. 2003. Mobile link organisms and ecosystem functioning: implications for ecosystem resilience and management. Ecosystems 6:87–98
Macarthur R. 1972. Strong, or weak, interactions? Trans Conn Acad Arts Sci 44:177–88
Mathisen OA, Parker PL, Goering JJ, Kline TC, Poe PH, Scalan RS. 1988. Recycling of marine elements transported into freshwater by anadromous salmon. Verhandlungen der Internationalen Vereinigung fur Theoretische und Angewandte Limnologie 23:2249–58
Meehan EP. 2000. The role of flies (Diptera: Calliphoridae) in salmon decomposition: implications for nutrient movement through intermediate trophic levels. Senior Thesis. Seattle (WA): University of Washington
Meehan WR, Swanson FJ, Sedell JR. 1977. Influences of riparian vegetation on aquatic ecosystems with particular reference to salmonid fishes and their food supply. In: Johnson RR, Jones DA, Eds. Importance, preservation, and management of riparian habitat: a symposium. Fort Collins (CO): USDA Forest Service, General Technical Report RM-43, Rocky Mountain Forest and Range Experiment Station, p 137–45
Michael JH. 1995. Enhancement effects of spawning pink salmon on stream rearing juvenile coho salmon: managing one resource to benefit another. Northwest Sci 69:228–233
Mills LS, Soule ME, Doak DF. 1993. The keystone-species concept in ecology and conservation. BioScience 43:219–24
Minakawa N. 1997. The dynamics of aquatic insect communities associated with salmon spawning. PhD Dissertation, Seattle (WA): University of Washington
Minakawa N, Gara RI. 1999. Ecological effects of a chum salmon (Oncorhynchus keta) spawning run in a small stream of the Pacific Northwest. J Freshw Ecol 14:327–35
Mulder CPH, Uliassi DD, Doak DF. 2001. Physical stress and diversity-productivity relationships: the role of positive interactions. Proc National Academy of Sciences of the United States of America 98: 6704–6708
Mynemi RB, Dong J, Tucker CJ, Kaufmann RK, Kauppi PE, Liski J, Zhou L, Alexeyev V, Hughes MK. 2001. A large carbon sink in the woody biomass of northern forests. Proc Nat Acad Sci USA 98:14784–9
Naiman RJ, Décamps H. 1997. The ecology of interfaces: riparian zones. Ann Rev Ecol Syst 28:621–58
Naiman RJ, Rogers KH. 1997. Large animals and system - level characteristic in river corridors. BioScience. 47:521–529
Naiman RJ, Fetherston KL, McKay SJ, Chen J. 1998. Riparian forests. In: Naiman RJ, Bilby RE, eds. River ecology and management. New York: Springer. p 289–323
Naiman RJ, Melillo JM, Lock MA, Ford TE, Reice SR. 1987. Longitudinal patterns of ecosystem processes and community structure in a subarctic river continuum. Ecology 68:1139–56
National Atmospheric Deposition Program/National Trends Network (NADP). 2003. Annual deposition data, sites AK01 and AK03. NADP Program Office, Illinois State Water Survey, 2204 Griffith Dr., Champaign, IL 61820
Nienstaedt H, Zasada JC. 1990. White spruce. Burns RM, Honkala BH, technical coordinators. Silvics of North America: 1. Conifers. Agriculture Handbook 654, Washington (DC): USDA Forest Service, p 204–26
O’Keefe TC, Edwards RT. 2003. Evidence for hyporheic transfer and removal of marine-derived nutrients in a sockeye stream in southwest Alaska. Am Fish Soc Symp 34:99–107
Orwell G. 1945. Animal farm. New York: Harcourt, Brace and Company. p 118
Paine RT. 1992. Food-web analysis through field measurement of per capita interaction strength. Nature 355:73–5
Paine RT. 1969. A note on trophic complexity and community stability. Am Nat 103:91–3
Paine RT. 1966. Food web complexity and species diversity. Am Nat 100:65–75
Pastor JP, Naiman RJ, Dewey B, McInnes P. 1988. Moose, microbes and the boreal forest. BioScience 38:770–7
Polis GA, Anderson WB, Holt RD. 1997. Toward an integration of landscape and food web ecology: the dynamics of spatially subsidized food webs. Annu Rev Ecol Syst 28:289–316
Power ME, Tilman D, Estes JA, Menge BA, Bond WJ, Mills LS, Daily G, Castilla JC, Lubchenco J, Paine RT. 1996. Challenges in the quest for keystones. BioScience 46:609–20
Quinn TP, Buck GB. 2000. Scavenging by brown bears, Ursus arctos, and glaucous-winged gulls, Larus glaucescens, on adult sockeye salmon, Oncorhynchus nerka. Can Field-Nat 114:217–23
Quinn TP, Kinnison MT. 1999. Size-selective predation by brown bears on sockeye salmon. Oecologia 121:273–82
Quinn TP, Wetzel L, Bishop S, Overberg K, Rogers DE. 2001. Influence of breeding habitat on bear predation and age at maturity and sexual dimorphism of sockeye salmon populations. Can J Zool 79:1782–93
Rai AN. 1988. Nitrogen metabolism. In: Galun M, ed. CRC Handbook of Lichenology, volume I. Boca Raton (FL): CRC. p 201–37
Rai AN, Rowell P, Stewart WDP. 1983. Interactions between cyanobacterium and fungus during 15N2-incorporation and metabolism in the lichen Peltigerna canina. Arch Microbiol 137:241–51
Reimchen TE. 2000. Some ecological and evolutionary aspects of bear-salmon interactions in coastal British Columbia. Can J Zool 78:448–57
Rhoades C, Oskarsson H, Binkley D, Stottlemyer B. 2001. Alder (Alnus crispa) effects on soils in ecosystems of the Agashashok River valley, northwest Alaska. Ecoscience 8:89–95
Rogers DE, Rogers BJ. 1998. Spawning ground surveys in the Wood River Lakes. Seattle (WA): University of Washington Alaska Salmon Program Technical Report No. FRI-UW-9803
Rohlf DJ. 1991. Six biological reasons why the Endangered Species Act doesn’t work – and what to do about it. Conserv Biol 5:273–82
Ruggerone GT, Hanson R, Rogers DE. 2000. Selective predation by brown bears (Ursus arctos) foraging on spawning sockeye salmon (Oncorhynchus nerka). Can J Zool 78:974–81
Schulze ED, Chapin FS III, Gebauer G. 1994. Nitrogen nutrition and isotope differences among life forms at the northern treeline of Alaska. Oecologia 100:406–12
Schuldt JA, Hershey AE. 1995. Effect of salmon carcass decomposition on Lake Superior tributary streams. J N Am Benthol Soc 14:259–68
Soulé ME, Estes JA, Berger J, Martínez Del Rio C. 2003. Ecological effectiveness: conservation goals for interactive species. Conserv Biol 17:1238–50
Suominen O, Danell K, Bryant JP. 1999. Indirect effects of mammalian browsers on vegetation and ground-dwelling insects in an Alaskan floodplain. Ecoscience 6:505–10
Triska FJ, Jackman AP, Duff JH, Avanzino RJ. 1994. Ammonium sorption to channel and riparian sediments: a transient storage pool for dissolved inorganic nitrogen. Biogeochemistry 26:67–83
U.S. Geological Survey (USGS). 1979. 1:63 360 series topographic map, Dillingham (B-8 and C-8) quadrangle, Alaska
Van Cleve K, Alexander V. 1981. Nitrogen cycling in tundra and boreal ecosystems. In: Clarke FE, Rosswall T, eds. Terrestrial nitrogen cycles, Ecological Bulletin, Vol. 33. Stockholm: Swedish Natural Science Research Council. P 375–404
Van Cleve K, Viereck LA. 1981. Forest succession in relation to nutrient cycling in the boreal forest of Alaska. In: West DC, Shugart HH, Botkin DB, eds. Forest succession: concepts and application. New York: Springer. p 179–211
Van Cleve K, Yarie J. 1986. Interaction of temperature, moisture, and soil chemistry in controlling nutrient cycling and ecosystem development in the taiga of Alaska. In: Van Cleve K, Chapin FS III, Flanagan PW, Viereck LA, Dyrness CT, eds. Forest ecosystems in the Alaskan taiga. New York: Springer. p 160–89
Van Cleve K, Oliver LK, Schlentner R, Viereck LA, Dyrness CT. 1983. Productivity and nutrient cycling in taiga forest ecosystems. Can J For Res 13:747–66
Van Cleve K, Viereck LA, Schlentner RL. 1971. Accumulation of nitrogen in alder (Alnus) ecosystems near Fairbanks, Alaska. Arct Alp Res 3:101–14
Van Daele LJ. 1998. Game management unit 17 brown bear survey and inventory report. In: Hicks MV, editor, Federal aid in wildlife restoration, management report of survey-inventory activities: brown bear. Juneau (AK): Alaska Department of Fish and Game. P 147–57
Van Daele LJ, Morgart JR, Hinkes MT, Kovach SD, Denton JW, Kaycon RH. 2001. Grizzlies, eskimos, and biologists: cross-cultural bear management in southwest Alaska. Ursus 12:141–52
Vogel JG, Gower ST. 1998. Carbon and nitrogen dynamics of boreal jack pine stands with and without a green alder understory. Ecosystems 1:386–400
Ward JV. 1989. The four dimensional nature of lotic ecosystems. J N Am Benthol Soc 8:2–8
Willson MF, Halupka KC. 1995. Anadromous fish as keystone species in vertebrate communities. Conserv Biol 9:489–97
Willson MF, Gende SM, Marston BH. 1998. Fishes and the forest: expanding perspectives on fish-wildlife interactions. BioScience 48:455–62
Wilson BF, Patterson WA III, O’Keefe JF. 1985. Longevity and persistence of alder west of the tree line on the Seward Peninsula, Alaska. Can J Bot 63:1870–5
Wipfli MS, Hudson J, Caouette J. 1998. Influence of salmon carcasses on stream productivity: response of biofilm and benthic macroinvertebrates in southeastern Alaska, USA. Can J Fish Aquat Sci 55:1503–11
Woodruff DS. 1989. The problems of conserving genes and species. In: Western D, Pearl MC, eds. Conservation for the twenty-first century. New York: Oxford University Press. p 76–88
Wurtz TL. 1995. Understory alder in three boreal forests of Alaska: local distribution and effects on soil fertility. Can J For Res 25:987–96
Wurtz TL. 2000. Interactions between white spruce and shrubby alders at three boreal forest sites in Alaska. Portland (OR): USDA Forest Service Pacific Northwest Research Station, General Technical Report PNW-GTR-481
Acknowledgements
We thank M. Ben-David, R. E. Bilby, R. T. Edwards, J. Elser, M. C. Liermann, T. C. O’Keefe, R. T. Paine, D. L. Peterson, T. P. Quinn, C. A. Reidy, D. E. Schindler and two anonymous reviewers for their valuable contributions to this paper. We also thank L. Ström for illustrating Figure 4 and the University of Washington Alaska Salmon Program for logistic support in Alaska. Financial support was provided by the National Science Foundation (DEB 98−06575), the United States Forest Service’s Pacific Northwest Research Station and the University of Washington.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Helfield, J.M., Naiman, R.J. Keystone Interactions: Salmon and Bear in Riparian Forests of Alaska. Ecosystems 9, 167–180 (2006). https://doi.org/10.1007/s10021-004-0063-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-004-0063-5